Share This Page
Drug Price Trends for SELENIUM SULFIDE
✉ Email this page to a colleague
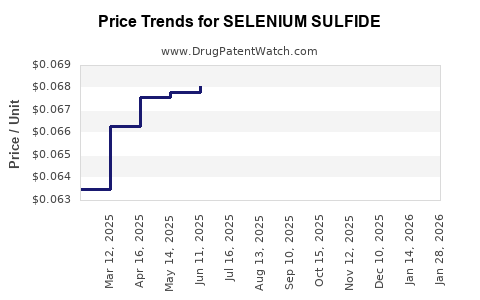
Average Pharmacy Cost for SELENIUM SULFIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SELENIUM SULFIDE 2.5% LOTION | 45802-0040-64 | 0.07852 | ML | 2026-05-20 |
| SELENIUM SULFIDE 2.25% SHAMPOO | 69367-0229-18 | 0.17035 | ML | 2026-05-20 |
| SELENIUM SULFIDE 2.25% SHAMPOO | 42192-0152-06 | 0.17035 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Selenium Sulfide: Market Dynamics and Price Projections
Selenium sulfide (SeS2) is an inorganic compound primarily utilized as an active pharmaceutical ingredient (API) in topical antifungal and antiseborrheic treatments. Its efficacy in managing conditions such as dandruff, seborrheic dermatitis, and tinea versicolor underpins its consistent demand in the pharmaceutical and personal care sectors. Market growth is influenced by the prevalence of these dermatological conditions, advancements in formulation technologies, and regulatory approvals for new indications or improved delivery systems.
What is the Current Global Market Size and Projected Growth for Selenium Sulfide?
The global selenium sulfide market was valued at approximately $500 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.5% over the next five years, reaching an estimated $625 million by 2028. This growth is primarily driven by the increasing incidence of fungal infections and scalp disorders, coupled with a rising consumer awareness regarding dermatological health and a growing preference for over-the-counter (OTC) treatments. The Asia-Pacific region is anticipated to exhibit the highest growth rate due to a large population base, improving healthcare infrastructure, and increasing disposable incomes.
What are the Key Therapeutic Applications and End-User Segments for Selenium Sulfide?
Selenium sulfide's primary therapeutic applications are concentrated in dermatology.
- Antifungal Agent: It effectively targets dermatophytes, the fungi responsible for conditions like tinea versicolor (pityriasis versicolor).
- Antiseborrheic Agent: It reduces the rate of skin cell turnover and exhibits fungistatic properties, making it a cornerstone treatment for dandruff and seborrheic dermatitis.
The principal end-user segments are:
- Pharmaceutical Manufacturers: Companies producing prescription and OTC topical medications for dermatological conditions. This segment represents the largest share of the market.
- Cosmetic and Personal Care Companies: Manufacturers of shampoos, conditioners, and scalp treatments formulated for anti-dandruff and scalp health.
- Contract Manufacturing Organizations (CMOs): Entities that produce selenium sulfide API or finished formulations on behalf of other companies.
A breakdown of market share by application in 2023 shows:
- Dandruff and Seborrheic Dermatitis: 75%
- Tinea Versicolor: 20%
- Other Dermatological Applications: 5%
What are the Dominant Product Forms and Strengths in the Selenium Sulfide Market?
Selenium sulfide is predominantly available in two primary product forms:
- Suspensions: Typically 1% or 2.5% concentrations, these are the most common formulations for shampoos and topical lotions. The 1% concentration is widely available as an OTC product.
- Powder: Used as an API for compounding pharmacies and for the synthesis of more complex formulations.
Market share by concentration in 2023:
- 1% Suspension: 65%
- 2.5% Suspension: 30%
- Powder/Other Concentrations: 5%
The 1% suspension formulation holds the largest market share due to its broad availability and established efficacy in OTC dandruff shampoos. The 2.5% concentration is generally available by prescription or in specialized OTC products.
What is the Competitive Landscape and Key Manufacturers of Selenium Sulfide?
The selenium sulfide market is moderately consolidated, with a few key players holding significant market shares. Competition is based on product quality, API purity, manufacturing capacity, regulatory compliance, and pricing. Key manufacturers include:
- Bio-Care Multiventures Pvt. Ltd.: A prominent Indian manufacturer with a strong export presence.
- Hubei Yuanhua Chemical Group Co., Ltd.: A major Chinese producer of chemical APIs.
- Hunan Jiujiang Pharmaceutical Co., Ltd.: Another significant Chinese supplier.
- Alfa Aesar (Thermo Fisher Scientific): Offers selenium sulfide for research and specialized applications.
- Sigma-Aldrich (Merck KGaA): Provides high-purity selenium sulfide for various industrial and research uses.
These companies supply API to formulators, and some also produce finished dosage forms. The market is also influenced by generic manufacturers who produce bioequivalent versions of branded selenium sulfide products.
What Factors Influence Selenium Sulfide Pricing?
Selenium sulfide pricing is influenced by several factors:
- Raw Material Costs: The cost of selenium metal and sulfur, the primary raw materials, directly impacts production costs. Fluctuations in the global supply and demand for these commodities can lead to price volatility.
- Manufacturing Costs: Energy consumption, labor, regulatory compliance (cGMP standards), and quality control processes contribute significantly to the overall cost of production.
- Purity and Grade: Pharmaceutical-grade selenium sulfide commands a higher price than technical-grade material due to stringent purity requirements and quality assurance processes.
- Supply Chain Dynamics: Availability of key intermediates, logistics, and geopolitical factors affecting trade can influence pricing.
- Regulatory Landscape: Compliance with FDA, EMA, and other national regulatory body requirements adds to manufacturing overhead and can affect market entry for new producers.
- Demand from End-User Industries: Increased demand from pharmaceutical and cosmetic sectors can drive prices up, especially for high-purity grades.
Average wholesale prices for pharmaceutical-grade selenium sulfide API (per kilogram) in early 2024:
- 99% Purity: $350 - $450
- 98% Purity: $300 - $380
These prices can vary based on order volume, supplier, and prevailing market conditions.
What are the Emerging Trends and Future Outlook for Selenium Sulfide?
Emerging trends in the selenium sulfide market include:
- Advancements in Formulation Technology: Research into novel delivery systems, such as microencapsulation or sustained-release formulations, aims to improve efficacy, reduce side effects, and enhance patient compliance.
- Exploration of New Indications: While established for dermatological conditions, ongoing research may explore selenium sulfide's potential in other therapeutic areas, though this is a nascent trend.
- Increasing Demand for OTC Products: The growing self-care trend and the convenience of OTC availability for common scalp conditions will continue to fuel demand for selenium sulfide-containing shampoos and lotions.
- Sustainable Manufacturing Practices: A growing emphasis on environmentally friendly production processes and the sourcing of raw materials is likely to influence manufacturing strategies.
The future outlook for selenium sulfide remains stable to positive. The established efficacy of selenium sulfide in treating prevalent dermatological conditions ensures continued demand. Growth will be steady, driven by the personal care market and the accessibility of OTC treatments. While significant disruptive technological advancements are not immediately apparent, incremental improvements in formulation and manufacturing efficiency are expected.
What are the Regulatory Considerations for Selenium Sulfide?
Selenium sulfide is subject to rigorous regulatory oversight globally. In the United States, it is regulated by the Food and Drug Administration (FDA) as an active ingredient in both OTC and prescription drugs.
- OTC Monograph System: For dandruff and seborrheic dermatitis treatments, 1% selenium sulfide is recognized under the FDA's OTC monograph system (21 CFR Part 358), allowing its use without specific new drug approval if manufacturers adhere to the monograph's conditions.
- New Drug Applications (NDAs): For 2.5% concentrations or for new indications, manufacturers must submit an NDA, which involves extensive clinical trials demonstrating safety and efficacy.
- Good Manufacturing Practices (cGMP): Manufacturers of selenium sulfide API and finished products must comply with cGMP guidelines to ensure product quality, safety, and consistency.
- International Regulations: Similar regulatory frameworks exist in Europe (European Medicines Agency - EMA), Canada (Health Canada), and other major markets, requiring compliance with local drug and cosmetic regulations.
Key Takeaways
- The global selenium sulfide market is projected to grow at a CAGR of 4.5%, reaching an estimated $625 million by 2028, driven by its use in treating common dermatological conditions.
- Dandruff and seborrheic dermatitis constitute the largest application segment, with 1% selenium sulfide suspensions being the dominant product form.
- Key manufacturers are primarily based in China and India, with a consolidated competitive landscape.
- Pricing is influenced by raw material costs (selenium, sulfur), manufacturing expenses, purity levels, and regulatory compliance.
- Emerging trends include formulation advancements and the continued demand for accessible OTC products.
- Selenium sulfide is regulated by bodies like the FDA and EMA, with adherence to cGMP and monograph requirements being critical for market access.
Frequently Asked Questions
-
What is the primary reason for the steady demand for selenium sulfide? The primary reason is its established efficacy as an antifungal and antiseborrheic agent in treating common dermatological conditions like dandruff, seborrheic dermatitis, and tinea versicolor.
-
Are there any significant new therapeutic areas being explored for selenium sulfide beyond dermatology? While research may explore other areas, the current market and significant growth drivers are firmly rooted in its dermatological applications. Exploration of new indications is a nascent trend and not a primary market driver presently.
-
How does the purity of selenium sulfide API affect its price? Higher purity grades, particularly pharmaceutical-grade selenium sulfide (e.g., 99%), command higher prices due to more rigorous manufacturing and quality control processes required to meet stringent regulatory standards for drug formulation.
-
What are the main challenges faced by selenium sulfide manufacturers? Challenges include volatility in raw material prices (selenium and sulfur), stringent regulatory compliance requirements (cGMP), and maintaining consistent product quality across different batches and suppliers.
-
Will the rise of natural or alternative ingredients impact the selenium sulfide market? While the personal care market sees interest in natural ingredients, selenium sulfide's established clinical efficacy for specific medical conditions provides it with a defensible market position, particularly in therapeutic shampoos and treatments where scientific validation is paramount.
Citations
[1] Market Research Report on Selenium Sulfide. (2023). Global Market Insights. [2] FDA Drug Approvals and Monograph System. (2023). U.S. Food and Drug Administration. [3] Chemical Supplier Data. (2024). Various Chemical Distributors and Manufacturers. [4] Industry Analysis of Pharmaceutical APIs. (2023). Grand View Research. [5] EMA Regulatory Guidelines for APIs. (2023). European Medicines Agency.
More… ↓
