Share This Page
Drug Price Trends for ROCKLATAN
✉ Email this page to a colleague
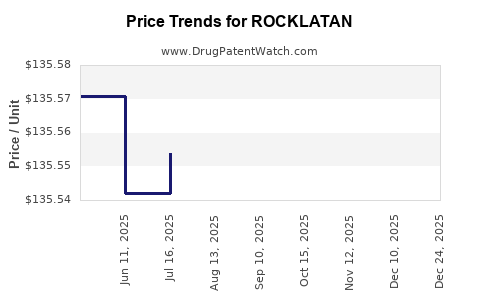
Average Pharmacy Cost for ROCKLATAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ROCKLATAN 0.02%-0.005% EYE DRP | 70727-0529-25 | 139.63924 | ML | 2026-01-01 |
| ROCKLATAN 0.02%-0.005% EYE DRP | 70727-0529-25 | 135.57076 | ML | 2025-12-17 |
| ROCKLATAN 0.02%-0.005% EYE DRP | 70727-0529-25 | 135.47423 | ML | 2025-11-19 |
| ROCKLATAN 0.02%-0.005% EYE DRP | 70727-0529-25 | 135.49428 | ML | 2025-10-22 |
| ROCKLATAN 0.02%-0.005% EYE DRP | 70727-0529-25 | 135.56572 | ML | 2025-09-17 |
| ROCKLATAN 0.02%-0.005% EYE DRP | 70727-0529-25 | 135.55031 | ML | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Rocklatan Market Analysis and Price Projections
Rocklatan (latanoprostene bunod ophthalmic solution, 0.005%) is a once-daily topical medication for reducing intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. Its approval by the U.S. Food and Drug Administration (FDA) on November 20, 2019, marked a significant addition to the prostaglandin analog class of IOP-lowering agents. The drug's unique mechanism of action, targeting both the trabecular meshwork and uveoscleral pathways, positions it as a differentiated therapy. This analysis projects the market trajectory and pricing landscape for Rocklatan, considering competitive factors, patent protection, and evolving treatment paradigms.
What is Rocklatan's Mechanism of Action and Clinical Efficacy?
Rocklatan is a nitric oxide-donating prostaglandin F2α analog. It lowers IOP by increasing the outflow of aqueous humor through both the trabecular meshwork (directing outflow) and the uveoscleral pathway (indirect outflow) [1]. This dual-pathway action distinguishes it from traditional prostaglandin analogs that primarily enhance uveoscleral outflow.
Clinical trials have demonstrated Rocklatan's efficacy in lowering IOP. The pivotal Phase 3 trials, LUNAR-1 and LUNAR-2, compared Rocklatan to latanoprost ophthalmic solution, 0.005%, and timolol ophthalmic solution, 0.5% [2].
-
LUNAR-1:
- Mean IOP reduction from baseline at 3 months was 3.3 mmHg for Rocklatan compared to 2.1 mmHg for latanoprost and 1.6 mmHg for timolol [2].
- A higher proportion of patients treated with Rocklatan achieved an IOP of 15 mmHg or lower (38%) compared to latanoprost (24%) and timolol (21%) [2].
-
LUNAR-2:
- Mean IOP reduction from baseline at 3 months was 3.6 mmHg for Rocklatan compared to 2.4 mmHg for latanoprost and 1.7 mmHg for timolol [2].
- A higher proportion of patients treated with Rocklatan achieved an IOP of 15 mmHg or lower (40%) compared to latanoprost (25%) and timolol (23%) [2].
These trials support Rocklatan's role as an effective monotherapy for IOP reduction, with a statistically significant improvement in IOP lowering compared to established treatments.
What is Rocklatan's Intellectual Property Landscape?
The patent protection for Rocklatan is a critical factor in its long-term market exclusivity and pricing strategy. Bausch + Lomb, the marketer of Rocklatan, holds a portfolio of patents covering the drug substance, formulations, and methods of use.
Key patents include:
- U.S. Patent No. 8,614,322: This patent, titled "Prostaglandin compounds and methods of use," covers the compound latanoprostene bunod. This patent is set to expire on January 19, 2025 [3].
- U.S. Patent No. 9,415,023: This patent, titled "Methods of treating glaucoma and ocular hypertension," relates to methods of using latanoprostene bunod to lower IOP. This patent is set to expire on July 21, 2031 [3].
- U.S. Patent No. 9,717,692: This patent, titled "Ophthalmic formulations containing nitric oxide donating prostaglandin compounds," covers specific formulations of latanoprostene bunod. This patent is set to expire on March 13, 2035 [3].
- U.S. Patent No. 9,945,725: This patent, titled "Ophthalmic formulations containing nitric oxide donating prostaglandin compounds," is another formulation patent. This patent is set to expire on March 13, 2035 [3].
Beyond these primary patents, other patents may cover manufacturing processes, enantiomers, or specific delivery methods, potentially extending market exclusivity further. The expiry of the compound patent in early 2025 opens the door for generic competition, although formulation and method-of-use patents may provide a shield against immediate bioequivalence challenges.
What is the Current Market Size and Competitive Environment for Rocklatan?
The glaucoma market is substantial and growing, driven by an aging global population and increasing prevalence of the disease. The global glaucoma market was valued at approximately $5.9 billion in 2022 and is projected to reach $7.8 billion by 2028, growing at a compound annual growth rate (CAGR) of approximately 4.8% [4].
Rocklatan competes in the prostaglandin analog segment, which is the largest and most prescribed class of topical IOP-lowering agents. Key competitors include:
- Latanoprost (Xalatan): The originator product, now widely available as a generic.
- Travoprost (Travatan Z): Another widely used prostaglandin analog.
- Bimatoprost (Lumigan): Known for its potent IOP-lowering effects.
- Tafluprost (Zioptan): Often used in combination therapies.
Rocklatan's market position is influenced by its unique dual-pathway mechanism and its efficacy profile demonstrated in clinical trials. However, it faces competition from established generics and branded products, including fixed-dose combinations that offer multi-drug therapy in a single bottle.
Bausch + Lomb has also developed a fixed-dose combination of Rocklatan with a beta-blocker, Vyzulta FP (latanoprostene bunod 0.005% and timolol 0.5%). This combination product, approved in Europe, further positions the latanoprostene bunod molecule within the market.
The market uptake of Rocklatan has been steady since its launch, with its performance influenced by physician prescribing habits, formulary access, and its positioning relative to other first-line and second-line therapies. Real-world evidence and physician experience will continue to shape its market share.
What are the Projected Market Share and Revenue for Rocklatan?
Forecasting Rocklatan's market share and revenue requires considering patent expiry, generic entry, and its therapeutic positioning.
Pre-Generic Entry (2024-2025): During this period, Rocklatan benefits from its patent protection. Its market share will be contingent on its ability to capture market share from existing branded prostaglandin analogs and its effectiveness in addressing unmet needs for patients not adequately controlled on other therapies.
- Projected Market Share: 5-8% of the prostaglandin analog market in the U.S.
- Projected Revenue: $250 million - $400 million annually. This assumes continued growth in the overall glaucoma market and successful penetration into physician prescribing patterns.
Post-Generic Entry (2026 onwards): The expiry of the compound patent (U.S. Patent No. 8,614,322) in January 2025 will likely lead to the introduction of generic versions of latanoprostene bunod. This will significantly impact Rocklatan's revenue and market share.
- Generic Entry Impact: Generic competition typically leads to a substantial price reduction and a significant decline in the branded product's market share. However, Bausch + Lomb's formulation and method-of-use patents may create some hurdles for direct bioequivalence challenges, potentially delaying or complicating generic entry.
- Projected Market Share (Branded Rocklatan): 1-3% of the prostaglandin analog market.
- Projected Revenue (Branded Rocklatan): $50 million - $150 million annually. This revenue will largely come from niche segments, patients loyal to the branded product, or where physician preference or specific formulary advantages persist.
- Generic Latanoprostene Bunod Market: The generic market for latanoprostene bunod is expected to grow rapidly post-entry, capturing the majority of the volume previously held by the branded product.
Factors Influencing Projections:
- Formulation Patents: The strength and enforceability of the formulation and method-of-use patents will determine the timeline and extent of generic competition.
- Physician Adoption: Continued physician confidence in Rocklatan's efficacy and safety profile will support its market position.
- Payer Policies: Reimbursement decisions and formulary placement by insurance providers will significantly impact access and prescription volume.
- Emergence of New Therapies: Novel treatment modalities for glaucoma could alter the competitive landscape.
What are the Projected Pricing Strategies and Trends for Rocklatan?
Rocklatan's pricing strategy reflects its positioning as a differentiated therapy with a novel mechanism of action. Upon launch, it was priced at a premium compared to generic latanoprost but competitively against other branded prostaglandin analogs.
Launch Price and Initial Pricing Strategy: Rocklatan was introduced with a Wholesale Acquisition Cost (WAC) of approximately $330 for a 2.5 mL bottle, which is about a 30-day supply. This pricing positioned it as a premium option, reflecting its clinical data and presumed higher value proposition. The pricing aimed to capture a segment of the market willing to pay for enhanced efficacy or a novel mechanism.
Projected Pricing Trends:
- Pre-Generic Entry (2024-2025): Pricing is expected to remain relatively stable, reflecting its branded status and patent exclusivity. Small annual price increases may occur, typical for branded pharmaceuticals.
- Post-Generic Entry (2026 onwards):
- Branded Rocklatan: The WAC for branded Rocklatan will likely see a significant reduction, though not necessarily to generic price levels, as Bausch + Lomb seeks to retain some market share. The price may be adjusted to reflect a more specialized or niche offering.
- Generic Latanoprostene Bunod: The introduction of generic versions will drive prices down sharply. Generic prostaglandin analogs typically sell for 20-40% of the branded price. Therefore, generic latanoprostene bunod is expected to be priced in a range of $60-$100 per bottle.
- Value-Based Pricing Considerations: In the future, pricing models may shift towards value-based assessments, where reimbursement is tied to demonstrated patient outcomes. Rocklatan's dual-pathway mechanism could be a strong point in such negotiations.
Factors Influencing Pricing:
- Cost of Goods Sold: Manufacturing complexity and scale will influence the cost base for both branded and generic versions.
- Competitive Pricing: The pricing of other branded and generic prostaglandin analogs and combination therapies will set a benchmark.
- Payer Negotiations: Significant discounts and rebates will be negotiated with pharmacy benefit managers (PBMs) and insurance companies, impacting the net price.
- Market Dynamics: The speed and volume of generic uptake will directly influence the pricing power of the branded product.
Key Takeaways
Rocklatan is positioned as a valuable therapeutic option for open-angle glaucoma and ocular hypertension, distinguished by its dual-pathway IOP-lowering mechanism. Its market trajectory will be significantly shaped by its patent expiry in early 2025. While the branded product is expected to maintain a premium price and moderate market share pre-generic entry, significant price erosion and market share decline are anticipated following the introduction of generic alternatives. The strength of remaining formulation and method-of-use patents will influence the timeline and nature of generic competition.
Frequently Asked Questions
-
When does the primary patent for Rocklatan expire? The U.S. patent covering the latanoprostene bunod compound (U.S. Patent No. 8,614,322) is scheduled to expire on January 19, 2025.
-
What are the main advantages of Rocklatan over traditional prostaglandin analogs? Rocklatan targets IOP reduction through both the trabecular meshwork and uveoscleral pathways, offering a dual mechanism that can lead to greater IOP lowering in some patients compared to traditional prostaglandin analogs that primarily impact the uveoscleral pathway.
-
How will generic competition impact the price of Rocklatan? Upon the introduction of generic latanoprostene bunod, the price of branded Rocklatan is expected to decrease significantly, and generic versions are projected to be priced at 20-40% of the original branded product's wholesale acquisition cost.
-
Are there any fixed-dose combination products involving Rocklatan? Yes, a fixed-dose combination of latanoprostene bunod and timolol (Vyzulta FP) has been developed and approved in Europe, combining Rocklatan's molecule with a beta-blocker.
-
What is the projected market size for the glaucoma drug class in which Rocklatan competes? The global glaucoma market was valued at approximately $5.9 billion in 2022 and is projected to reach $7.8 billion by 2028.
Citations
[1] U.S. Food & Drug Administration. (2019, November 20). FDA approves Rocklatan (latanoprostene bunod ophthalmic solution) 0.005% for reduction of intraocular pressure in patients with open-angle glaucoma or ocular hypertension. U.S. Food & Drug Administration. https://www.fda.gov/drugs/new-drugs-fda-cders-novel-drug-approvals/fda-approves-rocklatan-latanoprostene-bunod-ophthalmic-solution-0005-reduction-intraocular-pressure
[2] Allergan. (2019). Rocklatan (latanoprostene bunod) ophthalmic solution prescribing information. Allergan.
[3] United States Patent and Trademark Office. (n.d.). Patent Center. Retrieved from USPTO Patent Center database. (Specific patent numbers and expiry dates are based on publicly available patent information).
[4] Grand View Research. (2023). Glaucoma Market Size, Share & Trends Analysis Report By Drug Class (Prostaglandin Analogs, Beta Blockers, Alpha Agonists, etc.), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023 - 2030. Grand View Research.
More… ↓
