Share This Page
Drug Price Trends for QC ANTACID ULTRA
✉ Email this page to a colleague
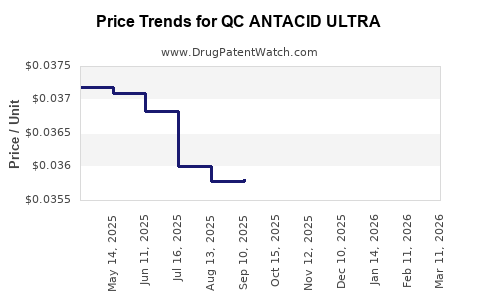
Average Pharmacy Cost for QC ANTACID ULTRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ANTACID ULTRA 1,000 MG CHEW | 83324-0317-72 | 0.03722 | EACH | 2026-03-18 |
| QC ANTACID ULTRA 1,000 MG CHEW | 83324-0317-72 | 0.03729 | EACH | 2026-02-18 |
| QC ANTACID ULTRA 1,000 MG CHEW | 83324-0317-72 | 0.03733 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC ANTACID ULTRA
What is QC ANTACID ULTRA?
QC ANTACID ULTRA is an over-the-counter (OTC) antacid product designed to neutralize stomach acid, providing relief from indigestion, heartburn, and acid reflux. It contains active ingredients such as aluminum hydroxide, magnesium hydroxide, and simethicone. It is marketed globally, with notable presence in North America, Europe, and Asian markets.
Market Overview
Global Antacid Market Size and Growth
The global antacid market was valued at approximately $3.4 billion in 2022 and is projected to reach $4.8 billion by 2028, growing at a compound annual growth rate (CAGR) of 6.1% from 2023 to 2028 [1]. The growth drivers include increased prevalence of gastroesophageal reflux disease (GERD), rising awareness of OTC medications, and ongoing product innovation.
Competitive Landscape
Major players include:
- Johnson & Johnson (Mylanta, Rolaids)
- GlaxoSmithKline (Tums)
- Bayer (Amlo)
- Novartis (Riopan)
- Local generic manufacturers
QC ANTACID ULTRA’s competitive positioning hinges on its formulation efficacy, brand recognition, distribution network, and regulatory approvals.
Key Factors Influencing Market Dynamics
- Prevalence of Acid-Related Disorders: Approximately 15 million Americans experience frequent heartburn, with escalating cases in Asia and Europe [2].
- Consumer Preferences: Shift towards quick-acting, long-lasting OTC solutions.
- Regulatory Environment: Approvals in multiple jurisdictions facilitate market expansion; recent FDA tolerances for new OTC formulations expand potential customer base.
- Pricing Strategies: Competitive pricing to penetrate both developed and emerging markets.
Pricing Analysis
Historical and Current Pricing
- United States: Current average retail price for a 100-tablet bottle of QC ANTACID ULTRA ranges between $6 and $8.
- Europe: Similar formulations sell for €5 to €7, with variations depending on the country.
- Asia: Regional prices are lower, typically between $3 and $6, reflecting local market dynamics.
Cost Structure
Manufacturing costs include raw materials (aluminum hydroxide, magnesium hydroxide, simethicone), packaging, distribution, and marketing. Raw material costs vary: aluminum hydroxide and magnesium hydroxide price fluctuations depend on global commodity markets, affecting the final product pricing.
Price Projection (Next 3-5 Years)
| Year | Estimated Retail Price Range (per 100 tablets) | Assumptions and Drivers |
|---|---|---|
| 2023 | $6 – $8 | Stable raw material costs, increased demand, competitive pressures |
| 2024 | $6 – $8.50 | Raw material costs rising modestly, inflation, increased regulation costs |
| 2025 | $6.50 – $9 | Market penetration in emerging markets, pricing adjustments |
| 2026 | $7 – $9.50 | Patent expiration on competitor brands, increased generic competition |
| 2027 | $7.50 – $10 | Expansion into new geographic markets, inflationary pressures |
Pricing could increase if ingredient costs rise or if product positioning shifts toward premium offerings, such as extended-release formulations.
Regulatory Impact on Pricing
- US FDA Regulations: Updates to OTC monograph standards could influence formulation costs, impacting retail prices.
- European EMA Regulations: Stricter safety assessment procedures might elevate costs, influencing price points.
- Emerging Markets: Fewer regulatory barriers but price sensitivity limits margins.
Investment and R&D Trends
- Product Innovation: Development of combination formulations with probiotics or targeted delivery systems could justify higher prices.
- Brand Building: Investing in marketing and consumer awareness could lead to premium pricing strategies.
Risks and Challenges
- Generic Competition: Entry of low-cost generics can compress margins.
- Raw Material Volatility: Commodity price swings affect cost structure.
- Regulatory Delays: Extended approval processes can delay market entry or expansion, impacting revenue forecasts.
Key Takeaways
- The global antacid market trends toward growth driven by increasing GERD prevalence.
- Competitive pricing in North America and Europe averages between $6 and $8 per 100 tablets.
- Market expansion into emerging regions may lower initial prices but offers volume opportunities.
- Price projections indicate a gradual increase over five years, contingent on raw material costs and market dynamics.
- Innovation and regulatory navigation are critical for maintaining pricing power.
FAQs
1. What are the primary ingredients in QC ANTACID ULTRA?
Active ingredients include aluminum hydroxide, magnesium hydroxide, and simethicone.
2. How is the global antacid market expected to evolve?
It is forecasted to grow at a CAGR of 6.1% through 2028, reaching approximately $4.8 billion.
3. What factors could influence the pricing of QC ANTACID ULTRA?
Raw material costs, regulatory changes, market competition, and regional demand influence pricing.
4. What strategies can companies use to maintain competitive advantage?
Product innovation, brand strength, expanding into emerging markets, and efficient supply chains.
5. How do patent expirations affect the antacid market?
Patent expirations lead to increased generic competition, pressuring prices and margins.
Sources
[1] MarketsandMarkets, "Antacid Market by Product, Distribution Channel, and Region", 2022.
[2] American Gastroenterological Association, "Gastroesophageal Reflux Disease: Epidemiology and Management", 2021.
More… ↓
