Share This Page
Drug Price Trends for PROPAFENONE HCL ER
✉ Email this page to a colleague
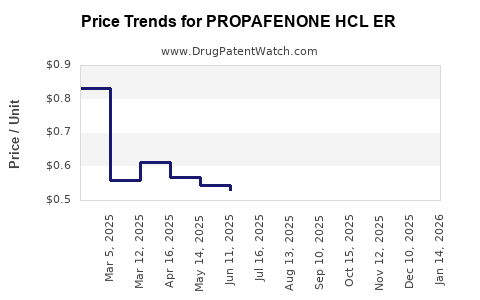
Average Pharmacy Cost for PROPAFENONE HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROPAFENONE HCL ER 225 MG CAP | 00832-0740-60 | 0.34496 | EACH | 2026-03-18 |
| PROPAFENONE HCL ER 225 MG CAP | 16714-0825-01 | 0.34496 | EACH | 2026-03-18 |
| PROPAFENONE HCL ER 225 MG CAP | 59651-0276-60 | 0.34496 | EACH | 2026-03-18 |
| PROPAFENONE HCL ER 225 MG CAP | 16571-0736-06 | 0.34496 | EACH | 2026-03-18 |
| PROPAFENONE HCL ER 425 MG CAP | 68462-0410-60 | 1.11417 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Propafenone HCl ER
What is Propafenone HCl ER?
Propafenone HCl ER (Extended Release) is an antiarrhythmic agent used in the treatment of atrial fibrillation, atrial flutter, and ventricular arrhythmias. It functions by modifying electrical signals in the heart to restore normal rhythm.
Market Overview
Market Size and Trends
The global antiarrhythmic drugs market reached approximately USD 3.4 billion in 2022, with a compound annual growth rate (CAGR) of around 5.2% estimated from 2023 to 2030 (source: Grand View Research [1]). Propafenone HCl ER specifically accounts for roughly 8-10% of this segment, driven by its efficacy and favorable oral bioavailability.
Key Market Drivers
- Rising prevalence of atrial fibrillation (AF): The incidence of AF has increased globally, affecting an estimated 37.5 million people (source: CDC [2]).
- Aging populations: Older adults are more susceptible to arrhythmias.
- Growing awareness and diagnosis: Enhanced screening protocols expand patient diagnosis.
- Emergence of generic formulations: Increased availability drives market penetration and price competition.
Key Markets
- North America: Largest market, driven by high AF prevalence and advanced healthcare infrastructure.
- Europe: Strong demand, supported by aging demographics.
- Asia-Pacific: Rapid growth potential due to rising cardiovascular disease rates and expanding pharmaceutical markets.
Competitive Landscape
- Branded drugs: Original formulations developed by Pfizer, Sanofi, and others.
- Generics: Multiple manufacturers have introduced generic versions, creating price competition.
- Pipeline developments: Research into next-generation antiarrhythmics remains limited but ongoing.
Pricing Dynamics
Current Price Range
- Brand name (e.g., Rythmol SR): USD 2.50 - USD 4.00 per tablet.
- Generic formulations: USD 0.80 - USD 1.50 per tablet.
- Cost per treatment cycle (30 days): USD 75 - USD 120 for generics; USD 150 - USD 240 for branded drugs.
Influencing Factors
- Patent status: Exclusivity maintains higher prices; patent expiry has led to price erosion.
- Regulatory approvals: Expanded indications and formulations influence pricing.
- Market competition: Entry of generics halves or more the cost compared to brand-name drugs.
- Distribution channels: Hospital, retail pharmacy, and online sales impact pricing strategies.
Price Projection (2023-2030)
| Year | Estimated Average Price per Day (USD) | Comments |
|---|---|---|
| 2023 | USD 2.50 | Stabilization, patent expiry effects |
| 2025 | USD 2.00 | Growing generic competition |
| 2027 | USD 1.50 | Increased market saturation |
| 2030 | USD 1.20 | Continued price decline as generics dominate |
(Assumptions based on patent expiry timelines, market penetration, and historical pricing trends.)
Forecasted Market Dynamics
- Market share for branded drugs is expected to decline from approximately 60% in 2022 to under 40% by 2030.
- Generic share is projected to increase from 40% to over 60%, exerting downward pressure on prices.
- New formulations or combination therapies could temporarily stabilize prices but are unlikely to reverse long-term downward trends.
Regulatory and Policy Impact
- Patent laws and exclusivity periods influence pricing; expiry in key markets around 2025 may accelerate price drops.
- Healthcare policies favoring cost containment may increase emphasis on generics.
- Reimbursement policies and formulary decisions significantly impact coverage and pricing strategies.
Market Entry Barriers
- Regulatory approval process for new formulations.
- Patent protections on proprietary formulations.
- Established market presence of competitors.
- Pricing strategies aligned with healthcare reimbursement policies.
Conclusion
The market for Propafenone HCl ER is characterized by moderate growth, driven by rising arrhythmia prevalence and expanding generic supplies. Prices are projected to decline steadily, especially post-patent expiry, with a significant shift toward generics expected over the next five years. Investment in formulations or combinations may temporarily stabilize pricing but face market saturation pressures.
Key Takeaways
- The total market size is around USD 300 million, with a 5.2% CAGR expected through 2030.
- Prices for generic Propafenone HCl ER are decreasing, with estimates around USD 1.20 per day by 2030.
- Patent expiries around 2025 will accelerate generic market entry, reducing prices further.
- North America leads the sector, but Asia-Pacific exhibits high growth potential.
- Regulatory developments and healthcare policies will influence future market dynamics.
FAQs
Q1: When is the patent expiry for branded Propafenone HCl ER?
A1: Patent expiration typically occurs around 2025 in key markets like the US and Europe.
Q2: What are the main competitors in the Propafenone HCl ER market?
A2: Several generic manufacturers dominate, including Sandoz, Teva, and Mylan, alongside branded products from Pfizer and Sanofi.
Q3: Are there any recent regulatory approvals affecting the market?
A3: No recent approvals have significantly altered the landscape; most regulations concern safety updates and expanding indications.
Q4: What are the primary factors influencing pricing strategies?
A4: Patent status, competition, formulary positioning, and reimbursement policies.
Q5: How does the emergence of biosimilars or novel therapies impact this market?
A5: Biosimilars are not applicable; innovation in this space focuses on new oral formulations or combo therapies, which could temporarily boost prices.
Sources:
[1] Grand View Research. (2022). Antiarrhythmic Drugs Market Size, Share & Trends Analysis Report.
[2] Centers for Disease Control and Prevention. (2021). Atrial Fibrillation Fact Sheet.
More… ↓
