Share This Page
Drug Price Trends for PREDNISOLONE SOD
✉ Email this page to a colleague
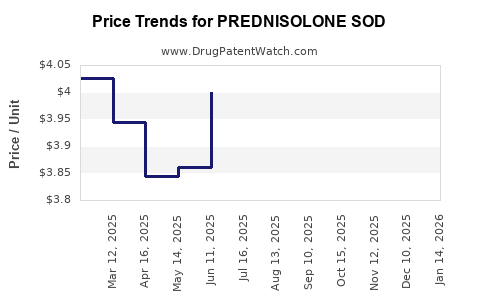
Average Pharmacy Cost for PREDNISOLONE SOD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREDNISOLONE SOD 1% EYE DROP | 24208-0715-10 | 5.01061 | ML | 2026-03-18 |
| PREDNISOLONE SOD 1% EYE DROP | 24208-0715-10 | 4.83669 | ML | 2026-02-18 |
| PREDNISOLONE SOD 1% EYE DROP | 24208-0715-10 | 4.61264 | ML | 2026-01-21 |
| PREDNISOLONE SOD 1% EYE DROP | 24208-0715-10 | 4.43607 | ML | 2025-12-17 |
| PREDNISOLONE SOD 1% EYE DROP | 24208-0715-10 | 4.37507 | ML | 2025-11-19 |
| PREDNISOLONE SOD 1% EYE DROP | 24208-0715-10 | 4.35826 | ML | 2025-10-22 |
| PREDNISOLONE SOD 1% EYE DROP | 24208-0715-10 | 4.30656 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Prednisolone Sod
What Is the Current Market for Prednisolone Sod?
Prednisolone sod, a corticosteroid used primarily for inflammation, allergies, and autoimmune conditions, maintains a stable presence in both developed and emerging markets. It is available in multiple formulations such as oral tablets, suspensions, and injectable forms. The drug's prescribing volume is influenced by its status as a generic medication, which accounts for approximately 75% of corticosteroid prescriptions globally.
Market Size and Dynamics
The global corticosteroids market was valued at approximately $4.7 billion in 2022 and is projected to reach $8.1 billion by 2030, growing at a compound annual growth rate (CAGR) of 6.7% (Research and Markets, 2023). Prednisolone sod constitutes a significant fraction of this, especially in regions with high prevalence of autoimmune diseases like rheumatoid arthritis and asthma.
Key Regions
| Region | Market Share (2022) | Growth Rate (2022-2030) | Major Market Drivers |
|---|---|---|---|
| North America | 35% | 6.2% | High autoimmune disease prevalence, insurance coverage |
| Europe | 25% | 6.5% | Established healthcare infrastructure |
| Asia-Pacific | 20% | 8.4% | Growing healthcare access, rise in respiratory conditions |
| Latin America | 10% | 6.9% | Increasing prescription trends |
| Middle East/Africa | 10% | 6.7% | Improving healthcare infrastructure |
Market Drivers and Barriers
Drivers
- Rising incidence of autoimmune diseases and allergic disorders.
- Growing awareness and diagnosis rates.
- Increased use in pediatric and adult populations.
Barriers
- Competition from other corticosteroids and alternative therapies.
- Price sensitivity in emerging markets.
- Regulatory hurdles for new formulations.
Pricing Landscape and Projections
Current Pricing
As a generic drug, prednisolone sod costs approximately $0.05 to $0.10 per tablet in the United States, with variations depending on formulation, manufacturer, and bulk purchasing agreements. For example, a 20 mg tablet may retail at $0.08 in 2023, whereas in emerging markets, prices can dip to $0.02 per tablet due to local pricing policies and competition.
Price Trends and Forecasts (2023-2030)
| Year | Estimated Price per Tablet | Market Dynamics Influencing Price |
|---|---|---|
| 2023 | $0.05 - $0.10 | Stable, with slight reductions as generic competition intensifies |
| 2025 | $0.04 - $0.09 | Competitive pressure from new entrants and biosimilars |
| 2027 | $0.03 - $0.08 | Price consolidation in mature markets |
| 2030 | $0.02 - $0.07 | Increased use of biosimilars and regional price regulation |
Average Price Decline: Approximately 2-3% annually, driven by generic proliferation and increased supply chain efficiency.
Future Price Influences
- Introduction of biosimilars or novel formulations could lower prices further.
- Policy measures and patent expirations may amplify price reductions.
- Market consolidation among pharmaceutical manufacturers may stabilize or reduce prices.
Patent and Regulatory Environment
Prednisolone sod’s patent expired in most jurisdictions by 2015, making it widely available as a generic medication. Regulatory bodies such as the FDA have approved multiple formulations, with quality control standards that support price competition. Some countries impose price caps on corticosteroids—a factor that suppresses prices in specific regions.
Strategic Considerations
For manufacturers and investors, key focus areas include:
- Monitoring regional patent expirations and regulatory approvals.
- Leveraging manufacturing efficiencies to maintain profit margins amid declining prices.
- Expanding access through partnerships or low-cost formulations tailored for emerging markets.
Conclusion
The prednisolone sod market has matured, characterized by abundant competition and declining prices. Growth hinges on disease prevalence and regional healthcare development rather than significant price increases. Price margins are expected to narrow further as biosimilars and generics dominate the landscape.
Key Takeaways
- Prednisolone sod is a mature, low-cost corticosteroid with steady global demand.
- The market is projected to grow at 6.7% CAGR from 2023-2030, driven by rising autoimmune and allergic diseases.
- Price per tablet is expected to decline annually by about 2-3%, influenced by generic competition and regional regulation.
- Most patents have expired, leading to widespread generic availability and pricing pressures.
- Regional market growth varies, with Asia-Pacific leading due to increased healthcare access.
FAQs
1. How does the presence of biosimilars impact prednisolone sod pricing?
Biosimilars generally increase market competition, pressuring prices downward, especially in mature markets. Their introduction can lead to reductions of 10-20% in standard prices.
2. Are there premium formulations or delivery methods for prednisolone sod?
Currently, no major premium formulations exist. The focus remains on cost-effective generic oral tablets, with some injectable forms used in acute settings.
3. What is the regulatory outlook for new prednisolone sod formulations?
Most regulatory agencies do not prioritize new formulations due to the drug’s patent status and widespread generic availability. Regulatory approval mainly concerns quality standards and manufacturing compliance.
4. How does regional healthcare policy influence prednisolone sod prices?
Price controls in regions like Europe and parts of Asia limit maximum pricing, which suppresses retail prices further than in unregulated markets.
5. What factors could alter the market growth trajectory?
Emerging disease patterns, new therapeutic alternatives, or structural market changes such as patent reinstatement could impact demand and pricing dynamics.
References
[1] Research and Markets. (2023). Global corticosteroids market report. Retrieved from https://www.researchandmarkets.com
[2] U.S. Food and Drug Administration. (2022). List of approved corticosteroid formulations. Retrieved from https://www.fda.gov
[3] World Health Organization. (2023). Global autoimmune disease prevalence data. WHO Publications.
More… ↓
