Drug Price Trends for PIOGLITAZONE-GLIMEPIRIDE
✉ Email this page to a colleague
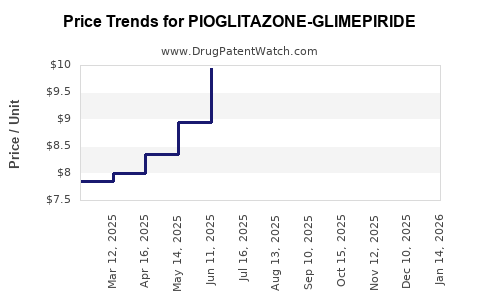
Average Pharmacy Cost for PIOGLITAZONE-GLIMEPIRIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PIOGLITAZONE-GLIMEPIRIDE 30-2 | 66993-0821-30 | 7.69783 | EACH | 2024-11-20 |
| PIOGLITAZONE-GLIMEPIRIDE 30-4 | 66993-0822-30 | 7.78960 | EACH | 2024-11-20 |
| PIOGLITAZONE-GLIMEPIRIDE 30-4 | 66993-0822-30 | 8.07933 | EACH | 2024-10-23 |
| PIOGLITAZONE-GLIMEPIRIDE 30-2 | 66993-0821-30 | 7.20222 | EACH | 2024-09-18 |
| PIOGLITAZONE-GLIMEPIRIDE 30-4 | 66993-0822-30 | 8.80860 | EACH | 2024-09-18 |
| PIOGLITAZONE-GLIMEPIRIDE 30-2 | 66993-0821-30 | 7.20222 | EACH | 2024-08-21 |
| PIOGLITAZONE-GLIMEPIRIDE 30-4 | 66993-0822-30 | 8.78163 | EACH | 2024-08-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |


