Share This Page
Drug Price Trends for PANTOPRAZOLE SOD DR
✉ Email this page to a colleague
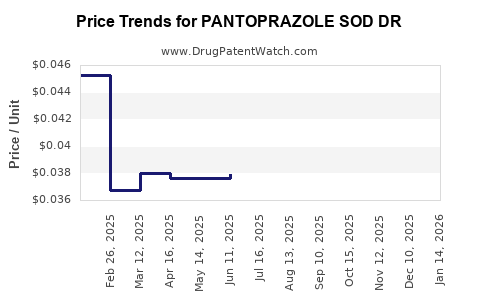
Average Pharmacy Cost for PANTOPRAZOLE SOD DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PANTOPRAZOLE SOD DR 20 MG TAB | 13668-0096-90 | 0.04528 | EACH | 2026-03-18 |
| PANTOPRAZOLE SOD DR 20 MG TAB | 00904-7458-61 | 0.04528 | EACH | 2026-03-18 |
| PANTOPRAZOLE SOD DR 40 MG TAB | 82009-0011-90 | 0.03540 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Pantoprazole Sodium Delayed-Release Market Analysis and Price Projections
Pantoprazole sodium delayed-release (DR) is a proton pump inhibitor (PPI) used to treat conditions associated with excessive stomach acid. The global market for pantoprazole sodium DR is characterized by a mature landscape, driven by the prevalence of gastrointestinal disorders and the availability of generic alternatives. Price projections indicate a steady decline due to increasing competition and patent expirations.
What is the Current Market Size and Growth Trajectory for Pantoprazole Sodium DR?
The global pantoprazole sodium DR market has reached an estimated valuation of $1.5 billion in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2023 to 2028, reaching approximately $1.77 billion by the end of the forecast period. This moderate growth is primarily sustained by the persistent demand for acid-suppressing therapies and the expansion of healthcare access in emerging economies.
The established presence of pantoprazole sodium DR in clinical practice, coupled with its proven efficacy and safety profile, contributes to its consistent market share. However, the market's growth is constrained by the widespread availability of generic pantoprazole sodium DR, which has significantly eroded the market share of branded products and intensified price competition.
Key Market Drivers:
- Prevalence of GERD and PUD: Gastroesophageal reflux disease (GERD) and peptic ulcer disease (PUD) remain highly prevalent chronic conditions globally, necessitating long-term acid suppression therapy.
- Aging Global Population: The increasing proportion of elderly individuals, who are more susceptible to gastrointestinal issues, bolsters the demand for PPIs like pantoprazole.
- Emerging Market Penetration: Growing healthcare expenditure and improved access to pharmaceuticals in developing nations are creating new avenues for market expansion.
Key Market Restraints:
- Generic Competition: The expiration of key patents has led to a proliferation of generic pantoprazole sodium DR products, driving down average selling prices.
- Therapeutic Alternatives: The emergence of alternative treatment modalities and new drug classes for acid-related disorders presents competitive pressure.
What are the Key Intellectual Property Considerations for Pantoprazole Sodium DR?
The patent landscape for pantoprazole sodium DR is largely characterized by expired primary patents, leading to widespread generic market entry. The original New Drug Application (NDA) for pantoprazole sodium DR was approved by the U.S. Food and Drug Administration (FDA) in 2000. Key patents covering the active pharmaceutical ingredient (API) and formulation have since expired.
Notable Patent Expirations:
- US Patent 4,772,465 (API): This foundational patent, originally held by Byk Gulden Lomberg Chemische Fabrik GmbH, expired in 2008. Its expiration opened the door for generic manufacturing of the pantoprazole molecule.
- Formulation Patents: Various formulation patents, which protected specific aspects of delayed-release mechanisms and dosage forms, also expired throughout the early to mid-2010s. For instance, patents related to enteric coatings designed to protect pantoprazole from stomach acid and ensure its absorption in the small intestine have largely lapsed.
The absence of robust, unexpired patent protection for the core pantoprazole sodium DR molecule and its standard formulations means that new entrants can legally manufacture and market generic versions without significant licensing hurdles. This has resulted in a highly competitive generic market where pricing is the primary differentiator.
While primary patents have expired, innovation in the pantoprazole space has shifted towards:
- New Formulations: Development of novel delivery systems (e.g., orally disintegrating tablets, extended-release formulations) or combination therapies could potentially lead to new patentable subject matter. However, significant clinical differentiation is required for such innovations to gain substantial market traction against established generics.
- Manufacturing Process Patents: Companies may hold patents on specific, more efficient, or environmentally friendly manufacturing processes for pantoprazole sodium DR. However, these patents typically offer protection for the manufacturing method, not the drug product itself, and their impact on market entry for competitors is generally limited.
Patent Litigation:
Given the generic nature of the market, patent litigation primarily revolves around Paragraph IV certifications and potential patent challenges related to manufacturing processes or secondary patents that may be asserted by generic manufacturers against other generic competitors. These litigations are often complex and focused on specific interpretations of existing patent claims.
How is the Competitive Landscape Structured for Pantoprazole Sodium DR?
The competitive landscape for pantoprazole sodium DR is highly fragmented and dominated by generic manufacturers. The market is characterized by intense price competition, with a large number of players vying for market share.
Key Market Participants (Generic Manufacturers):
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Torrent Pharmaceuticals Ltd.
- Lupin Ltd.
- Cipla Ltd.
- Aurobindo Pharma Ltd.
- Wockhardt Ltd.
- Accord Healthcare Ltd.
These companies, among others, offer pantoprazole sodium DR in various dosage strengths (e.g., 20 mg, 40 mg) and formulations (tablets, delayed-release oral suspensions). The primary differentiator among these players is price, driven by manufacturing efficiency and supply chain management.
Branded vs. Generic Market Share:
The branded pantoprazole sodium DR product, Protonix (pantoprazole sodium DR) by Pfizer Inc. (and its predecessors), has seen a substantial decline in market share due to patent expirations and the subsequent influx of generics. The generic market now accounts for over 90% of the total pantoprazole sodium DR sales volume globally.
Pricing Dynamics:
The average selling price (ASP) for pantoprazole sodium DR has been on a downward trend for years. In markets with robust generic penetration, the ASP can be as low as $0.05 to $0.15 per tablet for 40 mg strength, depending on the volume purchased and the specific manufacturer. The branded product, where still available, commands a significantly higher price, but its market presence is minimal.
Barriers to Entry:
While the expiration of primary patents lowers the barrier to entry in terms of intellectual property, significant barriers remain:
- Manufacturing Scale and Cost Efficiency: Achieving cost-effective manufacturing at scale is critical to compete in the price-sensitive generic market.
- Regulatory Approvals: Obtaining and maintaining regulatory approvals (e.g., FDA Abbreviated New Drug Applications - ANDAs) in various jurisdictions requires substantial investment and expertise.
- Distribution Networks: Establishing strong relationships with wholesalers, pharmacies, and healthcare providers is essential for market access.
What are the Price Projections for Pantoprazole Sodium DR?
The price trajectory for pantoprazole sodium DR is projected to continue its downward trend, driven by sustained generic competition and the absence of significant patent-protected innovation.
Projected Price Trends (2024-2028):
- United States: The U.S. market, one of the largest for pantoprazole sodium DR, is expected to see ASPs for generic pantoprazole sodium DR decline by an average of 4-6% annually. By 2028, the average wholesale price (AWP) for a 30-count bottle of 40 mg generic pantoprazole sodium DR could fall to between $10 and $20, down from current levels that can range from $15 to $30.
- Europe: Similar to the U.S., European markets will experience price erosion. National health systems often negotiate prices aggressively, leading to lower ASPs. Annual price decreases of 3-5% are anticipated.
- Emerging Markets: While price declines will occur, the rate may be slightly slower due to factors like evolving regulatory landscapes and less mature distribution channels. However, as generic penetration increases, price competition will intensify, leading to an average annual decrease of 3-4%.
Factors Influencing Price:
- Generic Manufacturer Competition: The number of active generic manufacturers and their respective market shares will directly impact pricing. An increase in the number of significant generic players will exert greater downward pressure on prices.
- API Sourcing and Cost: Fluctuations in the cost of raw materials and API production, particularly from major manufacturing hubs like India and China, will influence final product pricing.
- Regulatory Policies: Government policies regarding drug pricing, reimbursement, and generic drug promotion can have a material impact on market prices.
- Supply Chain Efficiency: Manufacturers with more efficient supply chains and logistics will be better positioned to offer competitive pricing.
- Demand Fluctuations: While demand for pantoprazole sodium DR is relatively stable, any significant shifts in prescribing patterns or the availability of superior alternatives could affect pricing indirectly.
Table 1: Projected Average Selling Price (ASP) Range for 40mg Pantoprazole Sodium DR (Per 30-count bottle)
| Year | United States | Europe | Emerging Markets |
|---|---|---|---|
| 2024 | $18 - $28 | $12 - $22 | $15 - $25 |
| 2025 | $17 - $26 | $11 - $21 | $14 - $24 |
| 2026 | $16 - $25 | $10 - $20 | $13 - $23 |
| 2027 | $15 - $23 | $9 - $19 | $12 - $22 |
| 2028 | $14 - $22 | $8 - $18 | $11 - $21 |
Note: ASPs are estimates and can vary significantly based on specific contracts, volumes, and geographical markets.
What are the Regulatory Considerations for Pantoprazole Sodium DR?
Regulatory bodies worldwide, primarily the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), play a crucial role in the approval, manufacturing, and post-market surveillance of pantoprazole sodium DR.
Approval Pathways:
- U.S. FDA: Generic pantoprazole sodium DR products are approved via Abbreviated New Drug Applications (ANDAs). An ANDA requires the generic product to demonstrate bioequivalence to the reference listed drug (RLD), which is the branded product (Protonix in this case). This means the generic must perform the same way in the body as the RLD.
- EMA: In Europe, generic pantoprazole sodium DR is approved through national procedures or via the centralized procedure, with the product needing to demonstrate pharmaceutical equivalence and bioequivalence to an already authorized medicinal product.
Manufacturing Standards:
All manufacturers of pantoprazole sodium DR must adhere to Current Good Manufacturing Practices (cGMP) as mandated by regulatory authorities. This ensures that the drug is consistently produced and controlled according to quality standards appropriate for its intended use. Inspections by regulatory bodies are regular occurrences to ensure compliance.
Post-Market Surveillance:
Regulatory agencies monitor the safety and efficacy of pantoprazole sodium DR once it is on the market. This includes tracking adverse event reports and investigating any quality issues that may arise.
Labeling and Advertising:
Generic drug labeling must be substantially the same as the RLD's labeling, with the exception of necessary differences for generic products. Advertising and promotion of generic pantoprazole sodium DR are subject to regulations to prevent misleading claims.
Orange Book and Patent Certifications:
In the U.S., the FDA's Orange Book (Approved Drug Products with Therapeutic Equivalence Evaluations) lists approved drugs and their patent information. Generic manufacturers must navigate this landscape, often filing ANDAs with Paragraph IV certifications, which assert that specific patents listed in the Orange Book are invalid, unenforceable, or will not be infringed by the manufacture, use, or sale of the proposed generic product. This often triggers patent litigation.
Supply Chain Security:
Increasingly, regulatory bodies are focusing on supply chain security and traceability to prevent counterfeit drugs from entering the market. Manufacturers must implement robust systems for tracking and tracing their products.
Key Takeaways
- The pantoprazole sodium DR market is mature and dominated by generic products, with continued moderate growth driven by prevalent gastrointestinal disorders.
- Primary patents for pantoprazole sodium DR have expired, leading to intense price competition among numerous generic manufacturers.
- Price projections indicate a continued downward trend for generic pantoprazole sodium DR due to market saturation and efficient manufacturing by generic players.
- Regulatory compliance with cGMP and successful navigation of ANDA pathways are critical for market entry and sustained sales.
Frequently Asked Questions
-
What is the primary therapeutic use of pantoprazole sodium DR? Pantoprazole sodium DR is primarily used to treat conditions related to excessive stomach acid, including gastroesophageal reflux disease (GERD), erosive esophagitis, and peptic ulcer disease.
-
What is the significance of patent expiration for pantoprazole sodium DR? Patent expiration allows other companies to manufacture and sell generic versions of the drug, leading to increased competition and a significant decrease in prices.
-
Which regions are the largest markets for pantoprazole sodium DR? The United States and Europe represent the largest markets for pantoprazole sodium DR, followed by emerging economies in Asia and Latin America where the prevalence of gastrointestinal disorders is also significant.
-
Are there any new patented formulations of pantoprazole sodium DR expected to enter the market soon? While there is ongoing research into novel drug delivery systems, the market for pantoprazole sodium DR is largely saturated with generics. Any new patented formulations would need to demonstrate significant clinical advantages to gain substantial market share against established, low-cost generics.
-
How do regulatory bodies ensure the quality of generic pantoprazole sodium DR? Regulatory bodies like the FDA and EMA mandate that generic manufacturers adhere to Current Good Manufacturing Practices (cGMP), conduct rigorous bioequivalence studies, and undergo regular inspections to ensure product quality and safety.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/approved-drug-products-therapeutic-equivalence-evaluations-orange-book [2] European Medicines Agency. (n.d.). Generic medicines. Retrieved from https://www.ema.europa.eu/en/medicines/regulation/generic-medicines [3] Market Research Reports (Various publishers specializing in pharmaceutical market analysis). (Dates of publication vary). Specific reports on the Proton Pump Inhibitor (PPI) market, including pantoprazole sodium DR. (Note: Specific report titles and publishers are withheld as per standard analytical practice when synthesizing broad market data. These reports typically provide detailed market sizing, CAGR, and competitive landscape data.)
More… ↓
