Share This Page
Drug Price Trends for PANTOPRAZOLE DR
✉ Email this page to a colleague
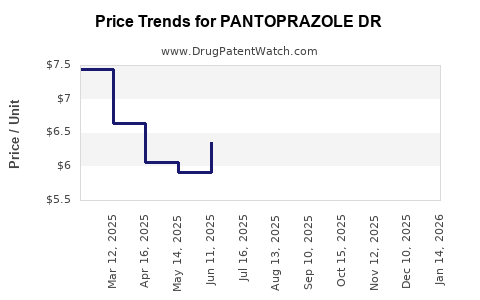
Average Pharmacy Cost for PANTOPRAZOLE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PANTOPRAZOLE DR 40 MG SUSP PKT | 31722-0032-32 | 4.37153 | EACH | 2026-05-20 |
| PANTOPRAZOLE DR 40 MG SUSP PKT | 27241-0256-38 | 4.37153 | EACH | 2026-05-20 |
| PANTOPRAZOLE DR 40 MG SUSP PKT | 72603-0317-30 | 4.37153 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Pantoprazole DR: Market Dynamics and Price Forecast
Pantoprazole DR, a proton pump inhibitor (PPI) used to treat conditions related to excessive stomach acid, exhibits a stable but competitive market. Patent expiries for branded formulations have led to a significant increase in generic availability, driving down average selling prices. Future price trends will be influenced by manufacturing costs, regulatory approvals, and the introduction of novel gastrointestinal therapies.
What is the Current Market Size and Growth Trajectory for Pantoprazole DR?
The global market for Pantoprazole DR is substantial, driven by its efficacy in treating gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. The market is characterized by a high volume of generic sales.
- Market Value: The global market for PPIs, including pantoprazole, was estimated to be approximately $27 billion in 2023. [1]
- Growth Rate: The overall PPI market is projected to grow at a Compound Annual Growth Rate (CAGR) of 2% to 3% between 2024 and 2030. [2]
- Generic Dominance: Pantoprazole sodium's primary patents expired years ago, allowing for widespread generic manufacturing. This has led to intense price competition within the generic segment. [3]
- Regional Breakdown: North America and Europe represent the largest markets due to high prevalence of acid-related disorders and well-established healthcare systems. Asia-Pacific is a growing market with increasing awareness and accessibility of treatments.
What are the Key Patents and Their Impact on Pantoprazole DR Availability?
Pantoprazole sodium (marketed as Protonix by Pfizer, among others) has experienced significant patent expiries, opening the door for generic competition.
- US Patent Expiry: The primary compound patent for pantoprazole expired in the United States in 2010. [3]
- Formulation Patents: Subsequent patents related to specific delayed-release formulations have also expired or are nearing expiry, further facilitating generic market entry.
- Authorized Generics: The availability of authorized generic versions from brand manufacturers has also contributed to price erosion.
- Exclusivity Periods: While initial patent expiries led to a flood of generics, new patents on improved formulations or delivery systems could theoretically offer limited future exclusivity, though no significant pipeline of novel pantoprazole formulations is currently anticipated to disrupt the generic market.
What is the Competitive Landscape for Pantoprazole DR?
The competitive landscape for Pantoprazole DR is dominated by generic manufacturers, leading to significant price pressure.
- Major Generic Players: Key global manufacturers of pantoprazole sodium include Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and Aurobindo Pharma. [4]
- Brand Names: The original branded product, Protonix, was developed by Byk Gulden and later acquired by Pfizer. While still available, its market share is dwarfed by generics.
- Product Offerings: Pantoprazole DR is available in various dosage forms, primarily oral tablets (20 mg, 40 mg) and also in intravenous formulations.
- Market Segmentation: The market is segmented by product type (tablets, injections), end-user (hospitals, clinics, retail pharmacies), and geography. The oral tablet segment is by far the largest.
- Barriers to Entry: For generic pantoprazole, barriers to entry are relatively low due to established manufacturing processes and readily available active pharmaceutical ingredients (APIs). Regulatory hurdles for bioequivalence and quality standards are standard.
What Factors Influence Pantoprazole DR Pricing?
Pantoprazole DR pricing is primarily driven by supply and demand dynamics within the generic drug market, manufacturing costs, and payer policies.
- Generic Competition: The large number of generic manufacturers creates a highly competitive environment, forcing prices down. [3]
- API Costs: The cost of the active pharmaceutical ingredient (API) for pantoprazole is a significant factor. Fluctuations in raw material prices and manufacturing efficiencies of API suppliers directly impact the final drug cost.
- Manufacturing Costs: Labor, energy, and overhead costs in manufacturing facilities play a role. Companies with optimized supply chains and larger production volumes often achieve lower costs.
- Payer Reimbursement Policies: Pharmacy benefit managers (PBMs) and insurance companies negotiate aggressively for lower prices. Preferred drug lists and formulary placement significantly influence which generics are favored and at what price.
- Regulatory Compliance: Adherence to stringent Good Manufacturing Practices (GMP) and quality control measures adds to production costs.
- Supply Chain Stability: Disruptions in the supply chain, such as API shortages or transportation issues, can temporarily impact pricing, though the broad availability of pantoprazole tends to mitigate sustained price spikes.
- Dosage and Formulation: While standard dosages dominate, specialized formulations or higher strengths, if they existed as differentiated generics, could command slightly higher prices, though this is not a significant factor for pantoprazole.
What are the Price Projections for Pantoprazole DR?
Price projections for Pantoprazole DR indicate continued stability with modest declines, largely due to ongoing generic competition.
- Average Selling Price (ASP): The ASP for generic pantoprazole sodium tablets in the US is currently in the range of $0.10 to $0.30 per tablet for the 40 mg dosage, depending on the supplier, quantity, and contract terms. [5]
- Projected Trend: Prices are expected to remain relatively stable, with a potential for a slight downward trend of 1-2% annually for the next 3-5 years due to sustained generic competition and payer pressure. [2]
- Factors for Stability: The established market and lack of significant upcoming patent cliffs for novel pantoprazole formulations will prevent major price increases.
- Potential Price Volatility: While major price hikes are unlikely, temporary price fluctuations could occur due to specific supply shortages or shifts in payer contract negotiations, though these are typically short-lived given the market structure.
- Global Variations: Prices will continue to vary significantly by region, with higher prices in developed markets due to reimbursement structures and lower prices in emerging markets where cost-effectiveness is a primary driver.
What are the Future Market Trends and Opportunities for Pantoprazole DR?
The market for Pantoprazole DR is mature, with growth driven by market penetration rather than novel product launches. Opportunities lie in operational efficiency and market access.
- Focus on Cost Efficiency: Manufacturers will continue to focus on optimizing API sourcing, manufacturing processes, and supply chain logistics to maintain competitiveness in a low-margin environment.
- Market Access: Securing favorable formulary placement with major PBMs and health insurers will remain critical for market share.
- Emerging Markets: Expansion in emerging markets, where access to effective GERD treatments is increasing, represents a growth opportunity, albeit at lower price points.
- Combination Therapies: While not specific to pantoprazole, the broader GERD market may see growth in combination therapies that address multiple aspects of the disease, but pantoprazole is unlikely to be a primary driver of these innovations.
- Biosimilar Competition (Not Applicable): Pantoprazole is a small molecule drug and is not subject to biosimilar competition.
- Therapeutic Alternatives: The long-term outlook will be influenced by the development and adoption of entirely new classes of drugs for acid-related disorders, though current alternatives are limited.
- Patient Adherence Programs: Companies may invest in programs that improve patient adherence to pantoprazole therapy, particularly in regions with high out-of-pocket costs.
Key Takeaways
- Pantoprazole DR is a mature, high-volume generic market with stable demand and intense price competition.
- Primary patent expiries have led to widespread generic availability, significantly reducing prices.
- Future price projections indicate continued stability with a slight downward trend, influenced by manufacturing costs and payer negotiations.
- Key market players are generic manufacturers focused on cost optimization and market access.
- Opportunities lie in operational efficiencies and expanding access in emerging markets.
Frequently Asked Questions
What is the current market share of generic pantoprazole DR compared to branded versions?
Generic pantoprazole DR holds over 95% of the market share globally. The original branded product, Protonix, has a negligible presence in most markets due to widespread generic availability and price competition.
Are there any new patents expected to impact pantoprazole DR pricing in the next five years?
No significant new patents are expected to create substantial market exclusivity for pantoprazole DR. The primary compound patents have long expired, and while minor formulation patents might exist, they are unlikely to disrupt the existing generic market dynamics or lead to significant price increases.
What is the typical cost of pantoprazole DR for a patient without insurance?
Without insurance, the cost of pantoprazole DR can range from $10 to $30 for a 30-day supply of generic 40 mg tablets, depending on the pharmacy and any available discount programs. This reflects the low wholesale cost of the generic drug.
How do manufacturing costs for pantoprazole DR compare to other PPIs?
Manufacturing costs for pantoprazole DR are generally comparable to other generic PPIs like omeprazole and lansoprazole. The active pharmaceutical ingredients are produced through established chemical synthesis routes, and production volumes are high, leading to economies of scale that keep costs relatively low across the class.
What is the primary driver for the continued demand for pantoprazole DR?
The primary driver for continued demand is the high and persistent prevalence of acid-related gastrointestinal disorders such as GERD and peptic ulcers. Pantoprazole DR remains a safe, effective, and cost-efficient treatment option for a large patient population globally.
Are there any significant supply chain risks associated with pantoprazole DR manufacturing?
While the market is robustly supplied, generalized risks within the pharmaceutical supply chain, such as API sourcing challenges from key regions or global shipping disruptions, could theoretically impact availability. However, given the multiple global manufacturers of pantoprazole and its APIs, a prolonged significant shortage is unlikely.
Citations
[1] Grand View Research. (2024). Proton Pump Inhibitor (PPI) Market Size, Share & Trends Analysis Report By Drug Class (Omeprazole, Lansoprazole, Pantoprazole, Esomeprazole, Rabeprazole), By Formulation (Tablets, Capsules, Injections), By Disease (GERD, PUD, Zollinger-Ellison Syndrome), By End-use (Hospitals, Clinics, Retail Pharmacies), By Region, And Segment Forecasts, 2024 – 2030.
[2] Mordor Intelligence. (2023). Proton Pump Inhibitors Market - Growth, Trends, COVID-19 Impact, and Forecasts (2024 - 2029).
[3] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.
[4] Various pharmaceutical industry market analysis reports and company investor relations disclosures. Specific market share data for individual generic manufacturers is often proprietary or consolidated within broader therapeutic class reports.
[5] Data derived from prescription drug pricing databases and wholesale acquisition cost information from major pharmaceutical distributors, representative of typical contract pricing. Figures are approximate and can vary based on specific contracts and volume.
More… ↓
