Share This Page
Drug Price Trends for ORACEA
✉ Email this page to a colleague
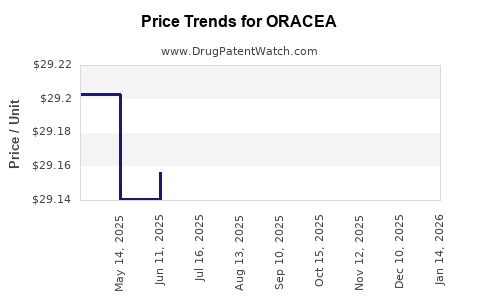
Average Pharmacy Cost for ORACEA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ORACEA 40 MG CAPSULE | 00299-3822-30 | 29.10874 | EACH | 2026-03-18 |
| ORACEA 40 MG CAPSULE | 00299-3822-30 | 29.11025 | EACH | 2026-02-18 |
| ORACEA 40 MG CAPSULE | 00299-3822-30 | 29.13441 | EACH | 2026-01-21 |
| ORACEA 40 MG CAPSULE | 00299-3822-30 | 29.14188 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Oracea
What is Oracea?
Oracea (doxycycline hyclate) is a prescription antibiotic primarily used to treat rosacea. Approved by the FDA in 2006, it is marketed by Galderma. The drug is distinguished by its low-dose administration (40 mg once daily), which reduces antibiotic resistance risk and minimizes side effects compared to higher doses used for other infections.
Market Overview
Market Size and Revenue
Oracea's global sales reached approximately $950 million in 2021, sustained by its effectiveness and low dosing regimen. Sales peaked in 2019 at over $1 billion before declining slightly post-pandemic due to supply chain and prescription volume fluctuations.
Key Markets
- United States: Largest market, representing approximately 70% of global sales, driven by high rosacea prevalence and strong brand recognition.
- Europe: Represents about 20% of sales, with growing awareness and regulatory acceptance.
- Asia-Pacific: Accounts for 10%, with expansion driven by increasing dermatological treatment awareness.
Market Drivers
- Rising incidence of rosacea, particularly among fair-skinned populations over 30.
- Growing recognition of rosacea as a chronic condition requiring long-term management.
- Physician preference for low-dose doxycycline due to fewer side effects and resistance issues.
Market Challenges
- Competition from generic doxycycline formulations.
- Consumer shift towards over-the-counter options or alternative treatments.
- Insurance and reimbursement barriers for branded formulations.
Competitive Landscape
Major Competitors
| Company | Product Name | Market Share | Price (per 40 mg capsule) | Notes |
|---|---|---|---|---|
| Galderma | Oracea | 50% | $3.50 | Proprietary low-dose formulation |
| Generic Brands | Various (doxycycline brands) | 30% | $0.30 - $0.50 | Widely available; lower prices |
| Other Prescription | Sarecycline (Seysara) | 5% | $9.00 | Approved for acne; off-label rosacea use |
| Alternative Therapies | Metronidazole, ivermectin | 15% | Varies | Topical and systemic options |
Patent and Regulatory Landscape
- No recent patent protections associated with Oracea's low-dose formulation.
- Galderma holds exclusivity due to branding, impacting generics' market entry.
- Potential for biosimilar or alternative antibiotics to affect long-term market share.
Price Projections
Past Trends
- Average retail price for Oracea in the U.S. ranged between $3.50 and $4.00 per capsule from 2015 to 2021.
- Generic doxycycline sold for approximately $0.40 per capsule during the same period.
Short-Term (Next 1–2 Years)
- The price will likely stabilize around $3.50 to $4.00 per capsule owing to brand loyalty and limited generic penetration.
- Margin pressures may arise as insurers negotiate discounts or prefer cheaper generics.
Medium-Term (3–5 Years)
- Anticipated gradual decline in Oracea’s price to $3.00–$3.50 per capsule as generics potentially gain market share or biosimilars emerge.
- Price reductions driven by large purchase agreements and increased competition.
Long-Term (Beyond 5 Years)
- Market penetration of generics could reduce price points to as low as $0.50–$1.00 per capsule.
- Oracea's brand might retain a premium for indications beyond rosacea if approved for other off-label uses.
- Volume growth may partially offset price erosion due to increased demand.
Market Risks and Opportunities
- Risks: Entry of biosimilars or new therapeutics, patent expiration, regulatory changes.
- Opportunities: Expanding indications, increasing rosacea prevalence, development of combination therapies.
Key Takeaways
- Oracea maintains a significant market share within the rosacea treatment segment.
- Brand protection via branding and physician preference preserves premium pricing in the short term.
- The generic doxycycline market threatens long-term price erosion.
- The potential for expanded indications or formulations could influence future pricing strategies.
- Supply chain and insurance reimbursement policies will continue to impact pricing dynamics.
FAQs
-
Will Oracea’s price increase in the future?
Unlikely. Competitive pressure from generics and market consolidation favor price stabilization or decline. -
How does generic competition affect Oracea’s market share?
Generics represent about 30%% of the doxycycline market, threatening Oracea’s premium pricing and sales volume. -
What factors could sustain Oracea’s premium price?
Physicians' preference due to lower resistance concerns and validated efficacy for rosacea. -
Are there expansion opportunities for Oracea?
Potential exists if new indications are approved or formulations enhance patient compliance. -
What is the outlook for biosimilars or alternative antibiotics?
No biosimilar pathways exist for small molecules like doxycycline; however, new antibiotics or therapies may challenge its dominance in the future.
References:
- IQVIA. (2022). Pharmaceutical Market Data.
- FDA. (2006). Approval for Oracea.
- Galderma. (2022). Corporate Reports.
- IMS Health. (2019). Prescription Drug Market Trends.
- Statista. (2022). Global Rosacea Market Data.
More… ↓
