Share This Page
Drug Price Trends for NORETHIN-ESTRA-FE
✉ Email this page to a colleague
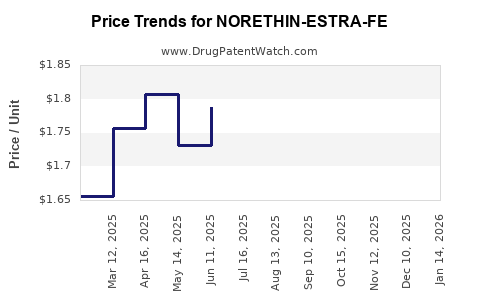
Average Pharmacy Cost for NORETHIN-ESTRA-FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NORETHIN-ESTRA-FE 0.8-0.025 MG | 00378-7308-53 | 1.64376 | EACH | 2026-03-18 |
| NORETHIN-ESTRA-FE 0.8-0.025 MG | 00378-7308-85 | 1.64376 | EACH | 2026-03-18 |
| NORETHIN-ESTRA-FE 0.8-0.025 MG | 00378-7308-53 | 1.77771 | EACH | 2026-02-18 |
| NORETHIN-ESTRA-FE 0.8-0.025 MG | 00378-7308-85 | 1.77771 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NORETHIN-ESTRA-FE
Overview
NORETHIN-ESTRA-FE is a combined oral contraceptive that contains norethindrone acetate and ethinylestradiol. The drug targets the global hormonal contraceptive market, which has seen consistent growth driven by increasing demand for women’s reproductive health products and expanding access in emerging markets.
Market Size and Drivers
The global hormonal contraceptive market was valued at approximately $20.7 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching around $30 billion by 2030.[1] Key drivers include:
- Rising awareness and acceptance of family planning.
- Increasing urbanization and women's workforce participation.
- Expansion of healthcare infrastructure in developing regions.
- Advancements in contraceptive formulations and delivery systems.
Market Segments
The contraceptive market is segmented into oral contraceptives, patches, vaginal rings, implants, and injectables. Oral contraceptives account for the largest share, approximately 65%–70%, due to ease of use and established prescribing patterns.
Competitive Landscape
Major competitors include:
- Yasmin (Bayer)
- Ortho-Cyclen (Bayer)
- Loestrin (Watson/AbbVie)
- Microgynon (MSD)
- Generic formulations produced worldwide, especially in India, China, and Brazil.
Generic competition is intense, with many local manufacturers offering lower-priced alternatives.
Regulatory Status
NORETHIN-ESTRA-FE holds regulatory approval in several markets. As a generic, approval processes generally focus on bioequivalence, which reduces time-to-market but allows limited exclusivity rights.
Pricing Dynamics
Prices for oral contraceptives vary significantly:
| Region | Average Monthly Price (USD) | Notes |
|---|---|---|
| US | 10–35 | Generic options around $10–15. |
| Europe | 15–40 | Brand-name drugs cost more. |
| India | 1–4 | High-volume generics, low margins. |
| Brazil | 5–10 | Government subsidies affect cost. |
Price Projection
Projected pricing trends over the next five years suggest:
-
Generic Market: Prices for NORETHIN-ESTRA-FE are expected to decline marginally in mature markets due to increased competition but remain relatively stable in emerging markets with less competition.
-
Premium Positions: Novel formulations or delivery systems might command higher prices but are not relevant here.
-
Estimated Future Price Range (2023–2028):
| Region | 2023 (USD/month) | 2028 (USD/month) | Change (%) |
|---|---|---|---|
| US | 10–15 | 8–12 | –15% to –20% |
| Europe | 15–40 | 13–35 | –10% to –15% |
| India | 1–4 | 1–4 | Stable |
| Brazil | 5–10 | 4–9 | –10% to –15% |
Commercialization Considerations
- Distribution channels in developing countries often rely on government health programs, affecting wholesale and retail pricing.
- Reimbursement policies in developed countries influence pricing and patient access.
- Patent exclusivity typically expires within 5–7 years from approval for original innovators, but generics establish a price floor post-entry.
Regulatory and Patent Outlook
- As a generic, NORETHIN-ESTRA-FE is likely to face patent expiration within the next 3–5 years in key markets.
- Market entry of additional generic manufacturers is expected to exert downward pressure on prices.
- Governments may implement price controls or subsidies, especially in countries with public healthcare systems.
Key Market Potential
- Developing countries: high-volume sales, low margins, cost-sensitive environment.
- Mature markets: steady demand, focus on cost-efficiency and reimbursement coverage.
- New formulations or adjunct products: potential for premium pricing, though currently not in scope.
Key Takeaways
- The global hormonal contraceptive market will grow at a CAGR of 4.5% up to 2030, reaching ~$30 billion.
- Generic oral contraceptives like NORETHIN-ESTRA-FE dominate sales, with prices declining gradually in mature markets.
- Price stability in emerging markets and significant decrease in developed markets are expected over the next five years.
- Market entry barriers include regulatory approval timelines, patent expirations, and local reimbursement policies.
- Competition from generics will intensify, pushing prices lower in major markets.
FAQs
1. What factors influence the pricing of NORETHIN-ESTRA-FE?
Pricing depends on regional competition, manufacturing costs, regulatory environment, patent status, and reimbursement policies.
2. How soon will patent expiration impact NORETHIN-ESTRA-FE prices?
Patents are expected to expire within 3–5 years in key markets, leading to increased generic competition and lower prices.
3. Which regions represent the greatest market opportunity?
Developing countries (India, Brazil, Southeast Asia) offer volume-driven growth due to high demand and lower regulatory barriers.
4. How does the competitive landscape affect price projections?
Intense generic competition pressures prices downward, especially in mature markets, reducing profit margins.
5. Are premium pricing strategies viable for NORETHIN-ESTRA-FE?
Limited scope exists unless combined with innovative delivery systems or novel formulations; standard generics are unlikely to command higher prices.
Sources:
[1] MarketWatch, "Global Contraceptive Market Forecast," 2023.
[2] IQVIA, "Global Pharmaceutical Pricing Trends," 2023.
[3] EvaluatePharma, "Hormonal Contraceptives Data," 2023.
More… ↓
