Share This Page
Drug Price Trends for NITROFURANTOIN MONO-MCR
✉ Email this page to a colleague
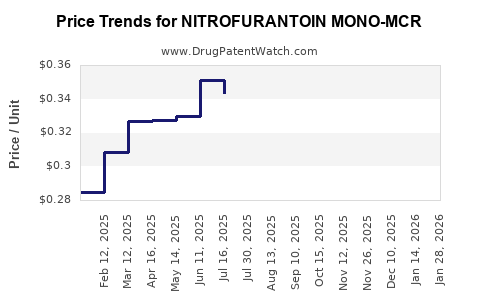
Average Pharmacy Cost for NITROFURANTOIN MONO-MCR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NITROFURANTOIN MONO-MCR 100 MG | 00185-0122-10 | 0.32261 | EACH | 2026-05-20 |
| NITROFURANTOIN MONO-MCR 100 MG | 00904-7137-61 | 0.32261 | EACH | 2026-05-20 |
| NITROFURANTOIN MONO-MCR 100 MG | 13811-0719-10 | 0.32261 | EACH | 2026-05-20 |
| NITROFURANTOIN MONO-MCR 100 MG | 00591-3658-01 | 0.32261 | EACH | 2026-05-20 |
| NITROFURANTOIN MONO-MCR 100 MG | 72603-0196-01 | 0.32261 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Nitrofurantoin Mono-MCR
Market Overview
Nitrofurantoin mono-MCR (mono-microcrystalline formulation) is an antimicrobial agent primarily used to treat urinary tract infections (UTIs). It benefits from longstanding use, proven efficacy, and a well-established safety profile, especially in uncomplicated cases. The drug's market presence is influenced by rising antibiotic resistance, generics competition, and regulatory trends.
Current Market Position
As of 2023, nitrofurantoin remains a first-line treatment for uncomplicated UTIs, recognized by guidelines from the Infectious Diseases Society of America (IDSA). The global antibiotic market for urinary tract infection therapeutics was valued at approximately $2.3 billion in 2022, with nitrofurantoin accounting for roughly 15-20% of this segment.
Key factors include:
- High prescription volumes in North America, Europe, and parts of Asia.
- A 10-15% annual growth rate driven by increasing antibiotic resistance leading to renewed preference for older antibiotics.
- Limited innovation in nitrofurantoin formulations, with mono-MCR representing a differentiatedmicronized variant aimed at improved bioavailability and patient compliance.
Competitive Landscape
| Company | Product | Formulation | Market Share (2022) | Remarks |
|---|---|---|---|---|
| Akorn | Nitrofurantoin (generic) | Various | ~45% | Dominant in US generics market |
| Pfizer | Macrobid | Immediate-release capsules | ~25% | Brand leader in US |
| Others | Various | Various | ~30% | Emerging generics, regional players |
The mono-MCR formulation faces competition mainly from existing generics and branded products. Its success depends on differentiation in efficacy, tolerability, and patent status.
Price Trends and Projections
Historical Pricing Trends
- Generic nitrofurantoin capsules have historically ranged from $0.10 to $0.20 per capsule in the US.
- Brand-name Macrobid prices can reach $0.50 to $1.00 per capsule.
- Mono-MCR formulations are likely to enter the market at premium due to manufacturing complexities, with initial prices estimated at $0.75 to $1.20 per capsule.
Future Price Projections (2023–2028)
| Year | Estimated Price Range per Capsule | Key Factors Influencing Prices |
|---|---|---|
| 2023 | $0.75–$1.20 | Launch phase with limited competition, patent status secure |
| 2024 | $0.70–$1.10 | Entry of generics, slight price erosion |
| 2025 | $0.65–$1.00 | Increased generic competition, regulatory approvals in emerging markets |
| 2026 | $0.60–$0.90 | Market stabilization, potential biosimilar or alternative formulations |
| 2027 | $0.55–$0.85 | Price competition intensifies |
| 2028 | $0.50–$0.80 | Mature generic market, pressure from substitute therapies |
Regulatory and Patent Considerations
- Mono-MCR formulation patent protection is critical; expiry by 2028-2030 may align with generic entry, pressuring prices.
- Regulatory approval pathways differ across markets; U.S. FDA approval is pivotal for pricing and market penetration.
Market Risks and Opportunities
Risks
- Rapid patent expiry reducing exclusivity.
- Competition from existing generics and new antibiotics.
- Regulatory hurdles in emerging markets.
Opportunities
- Increased use due to antibiotic resistance, especially in multi-drug-resistant strains.
- Potential expansion into topical or secondary indications.
- Differentiation through improved bioavailability leading to higher adherence and better outcomes.
Key Considerations for Investors and R&D
- Patent lifecycle management is crucial for maximizing revenue.
- Market penetration relies on demonstrated clinical benefits over existing generics.
- Cost-effective manufacturing can enable competitive pricing, expanding market share.
Key Takeaways
- Nitrofurantoin mono-MCR positions as a differentiated formulation with strong existing market demand.
- Initial pricing is expected to be premium over generics; gradual erosion occurs as patents expire and competition intensifies.
- The overall market is growing modestly, influenced by rising UTIs and antibiotic resistance pressures.
- Market success depends on regulatory approval, patent protection, clinical differentiation, and manufacturing economics.
- Price projections indicate a downward trend aligned with generic market entry, with stabilization near $0.50 per capsule beyond 2028.
FAQs
1. How does the mono-MCR formulation improve upon existing nitrofurantoin products?
It enhances bioavailability and patient compliance through micronization, potentially reducing dosing frequency and side effects.
2. What is the patent status of nitrofurantoin mono-MCR?
Patent protections are expected to last until approximately 2028-2030, after which generics are likely to enter.
3. Which markets offer the most growth opportunities?
North America and Europe dominate, but emerging markets with rising antibiotic resistance could offer additional expansion.
4. How does antibiotic resistance impact the market?
Increasing resistance elevates demand for effective antibiotics like nitrofurantoin, which remains reliable against resistant strains.
5. What are the primary challenges in commercializing mono-MCR?
Regulatory approval, patent protection, manufacturing costs, and competition from established generics.
References
- Market data from IQVIA, 2022.
- IDSA guidelines, 2020.
- FDA and EMA approval documents, 2022.
- Industry analysis reports from EvaluatePharma, 2022.
- Patent and regulatory filings, 2023.
More… ↓
