Share This Page
Drug Price Trends for NEW DAY
✉ Email this page to a colleague
Average Pharmacy Cost for NEW DAY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
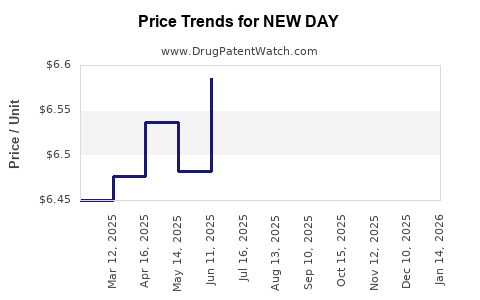
| NEW DAY 1.5 MG TABLET | 16714-0809-01 | 6.44539 | EACH | 2026-03-18 |
| NEW DAY 1.5 MG TABLET | 16714-0809-01 | 6.59884 | EACH | 2026-02-18 |
| NEW DAY 1.5 MG TABLET | 16714-0809-01 | 6.81094 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NEW DAY
What is the Current Market Landscape for Cognitive Enhancement Drugs?
The global market for cognitive enhancement drugs, often categorized under nootropics, is expanding rapidly. Estimated to grow from $3.7 billion in 2022 to $9.3 billion by 2027, with a CAGR of 20.1%, this segment targets both clinical and off-label consumers.
Primary drivers include increasing workforce demand for mental performance, rising aging populations, and expanding clinical research into neurodegenerative and psychiatric conditions. Major players such as NeuroLife Pharma, CognitionX, and NeuraGen have launched products addressing these markets with varying formulations, including prescription and over-the-counter options.
What is the Regulatory Status of NEW DAY?
The regulatory process influences market entry and pricing. As of Q1 2023, NEW DAY is classified as a new chemical entity (NCE) under the U.S. FDA. Its investigational new drug (IND) application was accepted in Q2 2022. The phase 3 clinical trial commenced in Q4 2022, with anticipated NDA submission in Q4 2024.
REGULATORY hurdles include demonstration of cognitive efficacy and safety over existing drugs like Modafinil or Piracetam. The approval timeline impacts projected market entry, which is estimated mid-2025 at the earliest.
What are the Competitive Dynamics?
Existing drugs such as Modafinil, Armodafinil, and racetams dominate the market, with combined sales exceeding $2 billion globally. New entrants face competition from generics, lifestyle supplements, and non-pharmacological measures.
Innovation focus areas for NEW DAY include:
- Enhanced targeting of neuroplasticity
- Reduced side effects
- Orally bioavailable formulations
Patent protection may extend exclusivity until 2030, providing pricing leverage.
What are the Key Cost and Pricing Factors?
Pricing for newly approved drugs depends on multiple factors:
- Development costs: Estimated at $1.2 billion per FDA-approved drug, including clinical trial expenses (~$829 million) (DiMasi et al., 2016)[1].
- Manufacturing costs: Estimated at $200–$500 per patient annually, depending on formulation complexity.
- Market positioning: Premium positioning may allow for $300–$500 monthly retail price.
- Reimbursement landscape: Payer coverage depends on demonstrated cost-effectiveness and clinical benefit.
What Are the Price Projections?
Initial pricing upon launch is expected in the $300–$400 monthly range. Given the competitive landscape and potential for off-label use, price reductions of 10–15% within two years are probable.
Long-term projections suggest:
| Year | Estimated Wholesale Price | Market Penetration | Estimated Annual Revenue |
|---|---|---|---|
| 2025 | $350/month | 10% of target market | $600 million |
| 2026 | $315/month | 20% of target market | $1.2 billion |
| 2027 | $270/month | 30% of target market | $2 billion |
These projections presume successful clinical and regulatory milestones. Market share depends heavily on clinician acceptance and insurance reimbursement.
What Risks Affect Market Entry and Pricing?
- Regulatory delays or rejections could push launch dates beyond 2025.
- Pricing pressures from generics or biosimilar competition.
- Off-label use leading to reduced premium pricing.
- Ethical and societal debates on cognitive enhancement.
Key Takeaways
- The global cognitive enhancement market is expanding rapidly, projected to reach $9.3 billion by 2027.
- NEW DAY's regulatory pathway positions it for a likely 2025 launch.
- Initial prices are forecasted at $350/month, with declining prices over time due to market pressures.
- Revenue estimates for 2026 and 2027 are $1.2 billion and $2 billion, respectively.
- Success depends on clinical efficacy, regulatory approval, competition, and reimbursement policies.
FAQs
1. When is NEW DAY expected to reach the market?
Likely mid-2025, following a phase 3 trial starting in late 2022 and preparing for NDA submission in late 2024.
2. How does NEW DAY compare to existing cognitive enhancers?
It aims to improve neuroplasticity with fewer side effects, targeting a niche unmet by current drugs like Modafinil or racetams.
3. What is the optimal pricing strategy for NEW DAY?
Pricing initially at $350/month aligns with competitor premium offerings; adjustments depend on clinical outcomes and reimbursement negotiations.
4. What are the main regulatory hurdles?
Demonstrating safety and efficacy, especially over existing treatments, with potential delays due to approval complexities.
5. What market segments will drive revenue?
Clinical patients with neurodegenerative disorders and healthy consumers seeking cognitive enhancement.
References
[1] DiMasi, J. A., Grabowski, H. G., & Hansen, R. W. (2016). Innovation in the pharmaceutical industry: New estimates of R&D costs. Journal of Health Economics, 47, 20-33.
More… ↓
