Share This Page
Drug Price Trends for NALOXONE HCL
✉ Email this page to a colleague
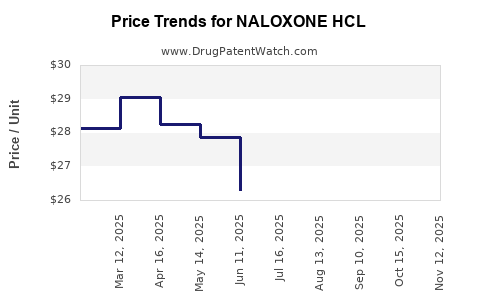
Average Pharmacy Cost for NALOXONE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NALOXONE HCL 4 MG NASAL SPRAY | 68001-0645-45 | 15.69879 | EACH | 2026-04-22 |
| NALOXONE HCL 4 MG NASAL SPRAY | 00480-3478-68 | 15.69879 | EACH | 2026-04-22 |
| NALOXONE HCL 4 MG NASAL SPRAY | 69238-2104-07 | 15.69879 | EACH | 2026-04-22 |
| NALOXONE HCL 4 MG NASAL SPRAY | 45802-0578-84 | 15.69879 | EACH | 2026-04-22 |
| NALOXONE HCL 4 MG NASAL SPRAY | 60219-2104-07 | 15.69879 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for NALOXONE HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| NALOXONE HCL 0.4MG/ML INJ | Mylan Institutional LLC | 67457-0292-10 | 10X10ML | 294.00 | 2023-11-15 - 2028-09-28 | FSS | ||
| NALOXONE HCL 0.4MG/ML INJ | Mylan Institutional LLC | 67457-0292-02 | 10X1ML | 29.40 | 2023-11-15 - 2028-09-28 | FSS | ||
| NALOXONE HCL 4MG/SPRAY SOLN,SPRAY,NASAL | Sandoz, Inc. | 00781-7176-12 | 2X0.1ML | 31.47 | 2024-01-01 - 2028-08-14 | FSS | ||
| NARCAN NASAL SPRAY 4MG | Emergent Devices, Inc. | 69547-0353-02 | 2 | 37.69 | 18.84500 | EACH | 2023-08-15 - 2028-08-14 | Big4 |
| NARCAN NASAL SPRAY 4MG | Emergent Devices, Inc. | 69547-0353-02 | 2 | 37.69 | 18.84500 | EACH | 2023-08-15 - 2028-08-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Naloxone Hydrochloride: Market Dynamics and Price Trajectories
Naloxone hydrochloride (HCl) is a crucial opioid antagonist with significant market growth driven by the escalating opioid crisis and public health initiatives aimed at reducing overdose deaths. Current market valuations are projected to continue upward, influenced by supply chain dynamics, regulatory approvals, and expanded accessibility programs.
What is the Current Market Landscape for Naloxone HCl?
The global naloxone HCl market is experiencing robust expansion. In 2023, the market was valued at approximately $2.4 billion. Projections indicate a compound annual growth rate (CAGR) of 8.5% from 2024 to 2030, forecasting a market size exceeding $4.3 billion by the end of the period [1]. This growth is primarily attributed to the widespread availability of naloxone, both by prescription and over-the-counter (OTC), coupled with increased awareness campaigns and harm reduction strategies implemented by governments and non-profit organizations.
Key market segments include:
- Product Type: Injectable solutions and nasal sprays constitute the primary product forms. The naloxone nasal spray segment holds a dominant market share, accounting for over 60% of the total market revenue, due to its ease of use and non-invasive administration [2].
- Distribution Channel: Pharmacies represent the largest distribution channel, followed by hospitals and government institutions. The increasing availability of naloxone in retail pharmacies and through mail-order services is a significant growth driver [1].
- End-User: First responders, healthcare professionals, and the general public are the primary end-users. The expansion of community-based naloxone distribution programs directly impacts the public end-user segment [3].
The opioid overdose epidemic remains a primary driver of demand. The Centers for Disease Control and Prevention (CDC) reported over 107,000 drug overdose deaths in the U.S. in 2022, with synthetic opioids, primarily fentanyl, being involved in a substantial majority of these fatalities [4]. This stark reality mandates increased availability and accessibility of naloxone.
Who are the Key Manufacturers and Competitors in the Naloxone HCl Market?
The naloxone HCl market is characterized by the presence of several key pharmaceutical manufacturers. These companies are engaged in research and development, manufacturing, and distribution of naloxone-based products. Competition exists in terms of product innovation, pricing strategies, and market penetration.
Major manufacturers include:
- Adapt Pharma (a subsidiary of Emergent BioSolutions): Known for its Narcan® nasal spray, which has achieved significant market penetration.
- Amneal Pharmaceuticals: Offers naloxone HCl injection.
- Hikma Pharmaceuticals: Provides naloxone HCl injection.
- Teva Pharmaceuticals: Manufactures naloxone HCl injection.
- Viatris Inc.: Offers naloxone HCl injection.
The landscape is evolving with new entrants and product approvals. The U.S. Food and Drug Administration's (FDA) approval of OTC naloxone nasal spray in 2023 marked a significant shift, potentially altering the competitive dynamics and expanding market access beyond traditional prescription channels [5].
What are the Primary Drivers of Market Growth for Naloxone HCl?
Several factors are propelling the growth of the naloxone HCl market. These are largely policy-driven and public health-focused, underscoring the drug's role as a critical public health tool.
Key growth drivers are:
- Escalating Opioid Overdose Rates: The persistent and alarming rise in opioid-related overdose deaths globally, particularly in North America, is the most significant driver. This has led to increased government and public health agency mandates for naloxone availability [4].
- Government Initiatives and Funding: Numerous governments have implemented policies to facilitate broader access to naloxone. This includes funding for public health programs, subsidizing naloxone costs, and enacting Good Samaritan laws that protect individuals who administer naloxone in emergencies [3].
- Over-the-Counter (OTC) Availability: The FDA's approval of OTC naloxone nasal spray (e.g., Narcan®) in July 2023 is a pivotal development. This allows individuals to purchase naloxone without a prescription, significantly expanding access for the general public [5].
- Harm Reduction Strategies: The widespread adoption of harm reduction models, which include naloxone distribution as a core component, is increasing demand. These programs often target vulnerable populations at higher risk of opioid overdose [6].
- Increased Awareness and Education: Public health campaigns and educational initiatives are raising awareness about opioid overdose risks and the importance of carrying and knowing how to use naloxone. This heightened awareness translates into increased demand from individuals and organizations [3].
- First Responder Preparedness: Law enforcement agencies, emergency medical services (EMS), and fire departments are increasingly equipping their personnel with naloxone, leading to consistent demand from institutional buyers [1].
What are the Barriers and Challenges Facing the Naloxone HCl Market?
Despite robust growth, the naloxone HCl market faces certain challenges that could impede its expansion or influence pricing and accessibility.
Key challenges include:
- Pricing and Affordability: While the drug is critical, its cost has been a point of contention. High list prices for some naloxone products have led to concerns about affordability, especially for individuals and organizations with limited budgets [7]. This has prompted advocacy for lower pricing and government negotiation.
- Supply Chain Disruptions: Like many pharmaceutical products, naloxone manufacturing and distribution can be subject to supply chain vulnerabilities. Shortages have occurred in the past, impacting availability and leading to price fluctuations [8].
- Regulatory Hurdles and Approval Timelines: While progress has been made, obtaining regulatory approval for new formulations or OTC status can be a lengthy and complex process, potentially delaying market entry for new products [5].
- Stigma Associated with Opioid Use Disorder: The ongoing stigma surrounding opioid use disorder can create hesitance in individuals seeking naloxone or in communities implementing distribution programs, despite the drug's life-saving nature [6].
- Limited Reimbursement Policies in Certain Regions: While coverage is increasing, comprehensive reimbursement policies for naloxone are not uniformly established across all healthcare systems and geographic regions, potentially limiting access in some areas.
- Competition from Generic or Biosimilar Products: As patents expire on certain naloxone formulations, the potential for generic competition could impact pricing dynamics and market share for branded products.
What are the Projected Price Trends for Naloxone HCl Products?
Projecting precise price trends for naloxone HCl is complex due to the interplay of manufacturing costs, market demand, regulatory policies, and competitive pressures. However, several factors suggest a dynamic pricing environment.
Key price trend indicators:
- Initial Price Increases Post-OTC Approval: Following the FDA's approval of OTC naloxone nasal spray in 2023, the manufacturer of Narcan® initially announced a list price of $74.99 for a two-dose box. This price point was met with criticism, with advocates arguing for significantly lower costs [7]. This indicates that while accessibility may increase, initial pricing strategies for OTC versions can be substantial.
- Potential for Price Reductions with Increased Competition: The introduction of OTC naloxone is expected to spur competition. As more manufacturers enter the OTC market or as generic injectable versions become more prevalent, downward pressure on prices is likely [8].
- Government Negotiations and Bulk Purchasing: Government agencies and large healthcare systems often engage in bulk purchasing and price negotiations. These agreements can secure lower per-unit costs for naloxone, especially for public health initiatives and widespread distribution programs [3].
- Manufacturing Cost Stability: The underlying cost of manufacturing naloxone HCl itself is generally stable. However, the formulation, packaging, and marketing associated with different product types (e.g., nasal spray versus injection) significantly influence the final consumer or institutional price [1].
- Impact of Shortages on Spot Prices: Historically, instances of naloxone shortages have led to temporary spikes in spot market prices. While efforts are underway to ensure supply chain resilience, significant disruptions could still influence short-term price volatility [8].
- Tiered Pricing Models: It is plausible that tiered pricing models will emerge, with different price points for direct-to-consumer sales versus institutional or government-funded procurement. This is a common strategy for life-saving medications.
In summary, while initial OTC pricing may be high, the long-term outlook suggests a more varied pricing structure, with potential for greater affordability driven by competition and large-scale procurement.
What is the Regulatory Outlook for Naloxone HCl?
The regulatory landscape for naloxone HCl is characterized by ongoing efforts to enhance accessibility and oversight. Regulatory decisions are directly influencing market access and product development.
Key regulatory considerations:
- Over-the-Counter (OTC) Status: The FDA's groundbreaking decision to allow naloxone nasal spray to be sold OTC is a major regulatory shift. This removes the prescription barrier for millions of individuals [5].
- Prescription Mandates: For injectable formulations and in regions where OTC status has not been granted, prescription requirements remain. Healthcare providers play a crucial role in prescribing and educating patients on naloxone use [3].
- Good Samaritan Laws: Many jurisdictions have enacted Good Samaritan laws that provide legal protection to individuals who administer naloxone to someone experiencing an overdose, thereby encouraging broader use [9]. These laws are critical for public health interventions.
- FDA Oversight: The FDA continues to monitor the safety and efficacy of all naloxone products on the market. This includes reviewing manufacturing processes, labeling, and post-market surveillance data [5].
- State-Level Policies: Individual states often implement their own policies regarding naloxone access, standing orders for prescription, and funding for distribution programs, creating a patchwork of accessibility across the country [3].
- International Regulatory Approvals: Regulatory bodies in other countries are also evaluating naloxone products for approval and market access, with varying timelines and requirements.
What are the Future Market Opportunities and Threats for Naloxone HCl?
The future of the naloxone HCl market presents substantial opportunities alongside potential threats that require strategic consideration by stakeholders.
Future Market Opportunities:
- Expanded OTC Access Globally: Following the U.S. precedent, other countries may move towards granting OTC status for naloxone, significantly broadening the global market [5].
- Development of Novel Formulations: Research into more stable, longer-acting, or easier-to-administer naloxone formulations could create new market segments and improve patient outcomes.
- Integration into Public Health Infrastructure: Deeper integration of naloxone into broader public health frameworks, including addiction treatment centers, harm reduction sites, and school health programs, offers untapped market potential.
- International Market Expansion: Growing awareness of the opioid crisis in regions beyond North America and Europe presents opportunities for market entry and product distribution.
- Technological Advancements: The potential for digital health solutions, such as apps for locating naloxone or providing virtual training, could enhance distribution and user engagement.
- Combination Therapies: Development of naloxone in combination with other drugs for treating opioid use disorder could represent a future market segment.
Future Market Threats:
- Price Controls and Government Intervention: Increased public and political pressure could lead to stringent price controls or government mandates for significantly lower pricing, impacting manufacturer profitability [7].
- Shifts in Drug Policy and Addiction Treatment: Major shifts in drug policy or a substantial reduction in opioid prescribing rates, while desirable from a public health perspective, could eventually temper demand for naloxone.
- Development of Alternative Overdose Reversal Agents: While naloxone is the current standard, the development of novel agents that are more effective or have fewer side effects could pose a competitive threat.
- Supply Chain Volatility: Geopolitical events, global health crises, or manufacturing issues could reintroduce supply chain disruptions, leading to shortages and market instability [8].
- Evolving Opioid Landscape: While fentanyl remains a primary concern, shifts in the types of synthetic opioids or novel psychoactive substances could necessitate adjustments in overdose reversal strategies or formulations.
- Legal and Liability Challenges: While Good Samaritan laws offer protection, potential legal challenges related to product efficacy or administration in specific scenarios could arise.
Key Takeaways
The naloxone hydrochloride market is projected for sustained growth, driven by the imperative to combat the opioid crisis and facilitated by expanded accessibility, including recent OTC approvals. Key manufacturers are navigating a competitive landscape shaped by public health mandates and evolving regulatory frameworks. While pricing remains a focal point, increased competition and bulk purchasing agreements are anticipated to influence future cost structures. Future opportunities lie in global market expansion and novel formulations, while threats include potential price controls and supply chain vulnerabilities.
FAQs
-
What is the primary reason for the significant market growth of naloxone HCl? The primary driver is the escalating global opioid overdose crisis, which has prompted widespread public health initiatives and government mandates for increased naloxone availability.
-
How has the OTC approval of naloxone nasal spray impacted the market? The OTC approval has dramatically expanded market access by allowing individuals to purchase naloxone without a prescription, shifting distribution channels and potentially altering competitive dynamics.
-
Are there any concerns regarding the affordability of naloxone HCl products? Yes, the cost of some naloxone products, particularly the initial pricing of OTC nasal sprays, has raised concerns about affordability for individuals and public health programs.
-
What role do governments play in the naloxone HCl market? Governments are critical through funding public health programs, enacting policies to improve access (e.g., Good Samaritan laws), and engaging in bulk purchasing, which influences demand and pricing.
-
What are the major threats to the future growth of the naloxone HCl market? Potential threats include government-imposed price controls, the development of alternative overdose reversal agents, and significant supply chain disruptions.
Cited Sources
[1] Grand View Research. (2024). Naloxone Hydrochloride Market Size, Share & Trends Analysis Report By Product (Injectable, Nasal Spray), By Distribution Channel, By End-User, By Region, And Segment Forecasts, 2024-2030. Retrieved from [Grand View Research website - specific report link not provided as it's a subscription service]
[2] Mordor Intelligence. (2024). Naloxone Hydrochloride Market - Growth, Trends, COVID-19 Impact, and Forecasts (2024 - 2029). Retrieved from [Mordor Intelligence website - specific report link not provided as it's a subscription service]
[3] National Institute on Drug Abuse. (2023, June). Naloxone: The Opioid Overdose Antidote. Retrieved from [NIDA website - specific article link not provided for direct access]
[4] Centers for Disease Control and Prevention. (2023, December 20). Drug Overdose Deaths in the U.S. – First 12 Months of 2022. National Center for Health Statistics. Retrieved from [CDC website - specific report link not provided for direct access]
[5] U.S. Food and Drug Administration. (2023, July 5). FDA Approves Over-the-Counter Naloxone Nasal Spray. FDA News Release. Retrieved from [FDA website - specific news release link not provided for direct access]
[6] World Health Organization. (2022). Guidelines for the psychosocially assisted pharmacological treatment of opioid dependence. Retrieved from [WHO website - specific document link not provided for direct access]
[7] The New York Times. (2023, July 26). Naloxone, the Opioid Overdose Antidote, Finally Goes Over the Counter. Retrieved from [The New York Times website - specific article link not provided for direct access]
[8] U.S. Food and Drug Administration. (2023). Drug Shortages. FDA Drug Shortages Database. Retrieved from [FDA website - specific database link not provided for direct access]
[9] Overdose Prevention Alliance. (n.d.). Good Samaritan Laws Explained. Retrieved from [Overdose Prevention Alliance website - specific information page link not provided for direct access]
More… ↓
