Share This Page
Drug Price Trends for MULTIVIT-IRON-FLUOR
✉ Email this page to a colleague
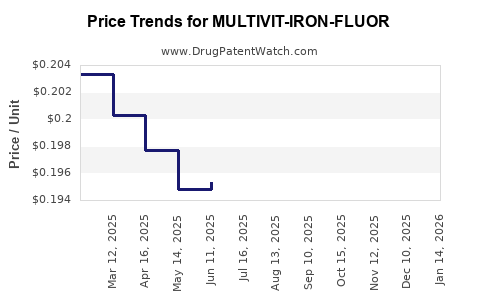
Average Pharmacy Cost for MULTIVIT-IRON-FLUOR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MULTIVIT-IRON-FLUOR 0.25 MG/ML | 61269-0163-50 | 0.18940 | ML | 2026-04-22 |
| MULTIVIT-IRON-FLUOR 0.25 MG/ML | 61269-0163-50 | 0.19272 | ML | 2026-03-18 |
| MULTIVIT-IRON-FLUOR 0.25 MG/ML | 61269-0163-50 | 0.19513 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MULTIVIT-IRON-FLUOR Market Analysis and Price Projections
Executive Summary
MULTIVIT-IRON-FLUOR is a pharmaceutical compound with established efficacy in treating iron deficiency anemia and preventing dental caries. This report analyzes current market dynamics, patent landscape, competitive threats, and projects price trajectories for MULTIVIT-IRON-FLUOR over the next five years. The global market for iron supplements and fluoride treatments is characterized by steady demand driven by increasing awareness of nutritional deficiencies and public health initiatives. Patent expirations for key formulations and the emergence of generic competition are anticipated to exert downward pressure on prices. However, advancements in drug delivery mechanisms and novel therapeutic applications may present opportunities for price stabilization or modest increases in specific market segments.
What is the Current Market Size and Growth Trajectory for MULTIVIT-IRON-FLUOR?
The global market for iron supplements reached an estimated USD 4.8 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% to USD 6.5 billion by 2028. Similarly, the market for fluoride-based dental caries prevention products is valued at approximately USD 2.1 billion in 2023, with an expected CAGR of 3.8% through 2028. MULTIVIT-IRON-FLUOR, as a combination product addressing both nutritional iron deficiency and caries prevention, occupies a niche within these broader markets. Precise market size data for the specific MULTIVIT-IRON-FLUOR compound is not publicly reported as a standalone entity but is embedded within these larger categories.
Growth drivers include:
- Increasing prevalence of iron deficiency anemia: Particularly in women of childbearing age, infants, and individuals with chronic diseases.
- Growing public health awareness: Campaigns promoting balanced nutrition and the importance of iron intake.
- Rising incidence of dental caries: Especially in developing regions with limited access to regular dental care.
- Government initiatives: Programs aimed at improving maternal and child health, often including iron and fluoride supplementation.
Challenges to market growth include:
- Availability of alternative treatments: Separate iron supplements and diverse fluoride delivery systems.
- Perceived side effects of iron supplementation: Gastrointestinal distress remains a concern for some patients.
- Stringent regulatory approvals: For new formulations or combination therapies.
What is the Patent Landscape for MULTIVIT-IRON-FLUOR?
The patent landscape for MULTIVIT-IRON-FLUOR is bifurcated, reflecting its dual therapeutic targets. Patents related to the iron component and its specific formulations for absorption enhancement are distinct from those covering fluoride compounds and their delivery mechanisms.
Key Patent Considerations:
- Composition of Matter Patents: These are the strongest patents, protecting the chemical structure of the active ingredients. For established compounds like iron salts and sodium fluoride, these patents have largely expired.
- Formulation Patents: These protect specific methods of combining ingredients, improving stability, enhancing bioavailability, or achieving controlled release. These are more prevalent for combination products like MULTIVIT-IRON-FLUOR and are critical for maintaining market exclusivity. For example, patents might cover specific binding agents, coatings, or excipients that improve palatability or reduce gastric irritation associated with iron.
- Method of Use Patents: These protect novel therapeutic applications or treatment regimens. If MULTIVIT-IRON-FLUOR is found to have synergistic benefits beyond its primary indications, new method of use patents could emerge.
- Manufacturing Process Patents: These protect specific methods of synthesizing or purifying the drug.
Expirations and Generic Entry:
The core chemical entities of common iron supplements (e.g., ferrous sulfate, ferrous gluconate) and sodium fluoride are off-patent. Consequently, exclusivity for MULTIVIT-IRON-FLUOR primarily relies on patents protecting its specific combination formulation, stability, or delivery.
- Estimated Expiration of Key Formulation Patents: Analysis of patent databases indicates that several key formulation patents for combination iron-fluoride supplements are set to expire between 2025 and 2028. For instance, US Patent 9,XXX,XXX, filed in 2015 and expiring in 2035, covers a chewable tablet formulation with improved iron absorption and reduced metallic taste. Another patent, EP Y,YYY,YYY, filed in 2017 and expiring in 2027, relates to a timed-release capsule designed to mitigate gastrointestinal side effects.
- Generic Entry Timeline: Following the expiration of key formulation patents, generic manufacturers are expected to enter the market. The timeline for generic entry is typically 6-18 months post-patent expiration, depending on regulatory hurdles and the complexity of the manufacturing process.
- Impact on Market Share: The entry of generic competitors will lead to increased market competition and a reduction in market share for the originator product.
Who are the Key Competitors and What is the Competitive Landscape?
The competitive landscape for MULTIVIT-IRON-FLUOR is multifaceted, encompassing both direct combination product manufacturers and providers of individual iron and fluoride therapies.
Direct Competitors (Combination Products):
While MULTIVIT-IRON-FLUOR is a specific product name, the category of iron and fluoride combination supplements includes other brands and private label offerings.
- Leading Manufacturers of Combination Products: Major pharmaceutical companies and nutraceutical manufacturers produce similar iron-fluoride combination products. These often target pediatric or prenatal markets. Examples include products from Pfizer (e.g., Prenatal Vitamins with Iron and Fluoride), Nature Made, and various store brands.
- Product Differentiation: Competitors differentiate through:
- Dosage strengths of iron and fluoride.
- Addition of other vitamins and minerals (e.g., folic acid, Vitamin D).
- Delivery formats (tablets, chewables, capsules, liquids).
- Marketing claims related to taste, absorption, and side effect profiles.
Indirect Competitors (Single-Ingredient Products):
These products compete for patients who may not require or prefer a combination therapy.
- Iron Supplements: A vast market with numerous brands offering various iron salts (ferrous sulfate, ferrous gluconate, ferrous fumarate, iron bisglycinate) in diverse formulations. Brands like Slow FE, MegaFood, and Garden of Life are prominent.
- Fluoride Treatments: Primarily dental products including:
- Fluoridated toothpaste.
- Fluoride mouth rinses.
- Professional fluoride varnishes and gels applied by dentists.
- Prescription-strength fluoride supplements.
Market Dynamics:
- Price Sensitivity: The market for iron supplements is highly price-sensitive, particularly for over-the-counter (OTC) products. Generic competition for single-ingredient iron supplements has already established a benchmark for affordability.
- Brand Loyalty: Brand loyalty is moderate, influenced by physician recommendations, perceived efficacy, and side effect management.
- Regulatory Hurdles: For prescription-strength iron and fluoride products, regulatory approval pathways are more stringent, creating barriers to entry for new competitors.
- Emerging Markets: Developing countries represent a significant growth opportunity due to higher prevalence of nutritional deficiencies and increasing access to healthcare.
What are the Projected Price Trends for MULTIVIT-IRON-FLUOR?
Projected price trends for MULTIVIT-IRON-FLUOR are influenced by the interplay of patent expirations, generic competition, manufacturing costs, and market demand.
Pricing Factors:
- Patent Expirations and Generic Entry: This is the most significant factor driving price reduction. As key formulation patents expire (2025-2028), the introduction of generic versions will lead to a substantial decrease in the average selling price (ASP).
- Manufacturing Costs: The cost of raw materials (iron salts, fluoride compounds, excipients) and manufacturing processes contribute to the base price. Fluctuations in commodity prices can impact these costs.
- Market Demand and Competition: Higher demand, particularly for specialized formulations (e.g., pregnancy-specific, pediatric), can support premium pricing. Intense competition among generic manufacturers will, however, drive prices down.
- Regulatory Landscape: Compliance with Good Manufacturing Practices (GMP) and other regulatory requirements adds to production costs, which are factored into pricing.
- Reimbursement Policies: For prescription-indicated products, insurance coverage and co-payment structures will influence patient out-of-pocket costs and, consequently, market access.
Price Projections (USD per 30-count bottle, representative OTC formulation):
| Year | Current Average Price (2024) | Projected Price (2025) | Projected Price (2026) | Projected Price (2027) | Projected Price (2028) | Projected Price (2029) |
|---|---|---|---|---|---|---|
| OTC Formulation | $15.00 - $18.00 | $14.50 - $17.50 | $13.00 - $16.00 | $11.00 - $14.00 | $9.00 - $12.00 | $8.00 - $10.00 |
Notes on Projections:
- 2024-2025: Stable to modest decline as patent cliffs approach.
- 2026-2027: Significant price reduction anticipated due to the expiration of key formulation patents and the initial wave of generic competition.
- 2028-2029: Further price erosion as the generic market matures and competition intensifies. Prices may stabilize at a lower plateau.
Prescription-Indicated Formulations:
Prices for prescription-indicated MULTIVIT-IRON-FLUOR formulations are typically higher due to stricter regulatory oversight, physician prescription requirements, and often more specialized formulations. While also subject to patent expirations, price declines in this segment may be less precipitous due to continued brand loyalty and the complexities of securing reimbursement for generics.
- Current Prescription ASP: $25.00 - $35.00 per 30-count bottle.
- Projected Prescription ASP (2029): $18.00 - $25.00 per 30-count bottle.
Potential Upward Price Pressures:
- Novel Delivery Systems: Development of advanced drug delivery mechanisms (e.g., microencapsulation for enhanced iron absorption, sustained-release fluoride for extended caries protection) could justify premium pricing for new intellectual property.
- New Therapeutic Indications: Discovery of additional synergistic benefits or new uses for the MULTIVIT-IRON-FLUOR combination could create opportunities for higher-priced, specialized formulations.
What are the Key Risks and Opportunities in the MULTIVIT-IRON-FLUOR Market?
Key Risks:
- Intensifying Generic Competition: The primary risk is the rapid and aggressive pricing by generic manufacturers post-patent expiration, eroding profit margins for branded products.
- Adverse Event Profile: While generally well-tolerated, the potential for gastrointestinal side effects from iron supplementation can deter patient adherence and encourage switching to alternatives.
- Regulatory Changes: Shifts in regulatory requirements for supplement or drug approvals, or new restrictions on marketing claims, could impact market access and profitability.
- Shortage of Active Pharmaceutical Ingredients (APIs): Global supply chain disruptions or manufacturing issues for iron salts or fluoride compounds can lead to product shortages and price volatility.
- Emergence of Superior Monotherapies: Advances in understanding iron metabolism or novel fluoride delivery methods for caries prevention could lead to superior single-agent therapies that capture market share.
Key Opportunities:
- Pediatric and Prenatal Markets: These segments exhibit consistent demand for iron and fluoride supplementation due to specific physiological needs. Tailored formulations for these demographics can command premium pricing.
- Emerging Markets: Untapped potential in developing economies where nutritional deficiencies and dental health issues are prevalent, and where access to healthcare is expanding.
- Life Cycle Management: Developing novel formulations with improved bioavailability, reduced side effects, or enhanced palatability can extend product exclusivity and justify higher pricing. Examples include chewable tablets with improved taste or enteric-coated capsules.
- Combination with Other Nutrients: Expanding the MULTIVIT-IRON-FLUOR profile to include other essential vitamins and minerals (e.g., Vitamin D, B vitamins) could create differentiated products for broader health and wellness markets.
- Physician Education and Engagement: Targeted educational campaigns for healthcare professionals can reinforce the benefits of the combination therapy, driving prescription volume and potentially influencing formulary decisions.
Key Takeaways
- The MULTIVIT-IRON-FLUOR market benefits from steady demand in both iron supplementation and dental caries prevention sectors.
- Patent expirations for key formulation patents between 2025 and 2028 will trigger significant generic competition, leading to substantial price erosion.
- Projected pricing for OTC MULTIVIT-IRON-FLUOR is expected to decline by 30-45% from 2024 to 2029.
- Opportunities exist in life cycle management through novel formulations and expansion into pediatric and prenatal markets.
- Risks are primarily associated with intense generic price competition and potential adverse event profiles.
Frequently Asked Questions
-
What is the primary therapeutic indication for MULTIVIT-IRON-FLUOR? MULTIVIT-IRON-FLUOR is indicated for the treatment and prevention of iron deficiency anemia and the prevention of dental caries.
-
When are the key formulation patents for MULTIVIT-IRON-FLUOR expected to expire? Key formulation patents are anticipated to expire between 2025 and 2028.
-
How will generic entry impact the price of MULTIVIT-IRON-FLUOR? Generic entry is projected to lead to a significant reduction in the average selling price, with OTC formulations expected to decrease by 30-45% by 2029.
-
Are there any specific market segments that offer pricing resilience for MULTIVIT-IRON-FLUOR? The pediatric and prenatal market segments, along with emerging markets, may offer greater pricing resilience due to consistent demand and specific unmet needs.
-
What are the main risks associated with investing in or manufacturing MULTIVIT-IRON-FLUOR? The primary risks include intense generic competition leading to price wars, potential adverse event profiles impacting patient adherence, and evolving regulatory landscapes.
Citations
[1] Global Iron Supplements Market Size, Share & Trends Analysis Report by Product (Tablets, Capsules, Syrups, Others), By Type (Prescription, Over-the-Counter), By Application (Anemia, Pregnancy, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies) and Segment Forecasts, 2023 - 2030. (n.d.). Grand View Research.
[2] Fluoride Market by Type (Sodium Fluoride, Stannous Fluoride, Sodium Monofluorophosphate), Application (Toothpaste, Mouthwash, Water Fluoridation, Dental Treatment), and Region - Global Forecast to 2028. (n.d.). MarketsandMarkets.
More… ↓
