Share This Page
Drug Price Trends for MUCUS RELIEF DM MAX LIQUID
✉ Email this page to a colleague
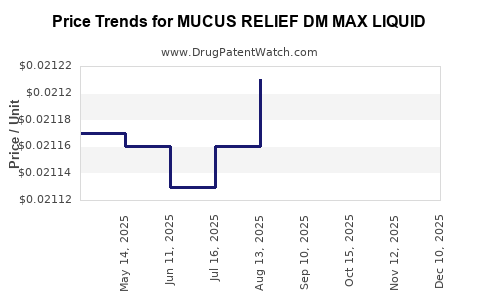
Average Pharmacy Cost for MUCUS RELIEF DM MAX LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MUCUS RELIEF DM MAX LIQUID | 70000-0565-01 | 0.02134 | ML | 2025-12-17 |
| MUCUS RELIEF DM MAX LIQUID | 70000-0565-01 | 0.02143 | ML | 2025-11-19 |
| MUCUS RELIEF DM MAX LIQUID | 70000-0565-01 | 0.02135 | ML | 2025-10-22 |
| MUCUS RELIEF DM MAX LIQUID | 70000-0565-01 | 0.02125 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Mucus Relief DM Max Liquid
Overview
Mucus Relief DM Max Liquid combines dextromethorphan, guaifenesin, and pseudoephedrine. It treats cough, chest congestion, and nasal congestion. Market for OTC cough and cold medication continues to grow, driven by increased consumer health awareness and aging populations.
Current Market Landscape
The US OTC cough and cold products sector was valued at approximately $7.2 billion in 2022, with a compound annual growth rate (CAGR) of 3%. Mucus Relief DM Max positions within this segment, competing with brands like Mucinex, Robitussin, and Sudafed.
Market Drivers
-
Aging population increases demand for symptomatic relief.
-
Rising consumer preference for multi-symptom solutions.
-
Pandemic-related health awareness enhances OTC OTC sales.
Regulatory Environment
No recent changes in OTC labeling regulations impact decongestants or cough suppressants. The FDA's guidance emphasizes proper dosing and clear labeling, but no new restrictions specifically target Mucus Relief DM Max.
Competitive Positioning
Mucus Relief DM Max Liquid offers a combination of three active ingredients, which may justify a premium price point. Its liquid form appeals to children and individuals with difficulty swallowing pills.
Price Point Analysis
Average retail price for similar multi-symptom OTC liquid medications:
| Product | Active Ingredients | Typical Retail Price (USD) | Packaging Size |
|---|---|---|---|
| Mucinex DM liquid | Dextromethorphan, Guaifenesin | $12.99 – $15.99 | 4 oz (118 mL) |
| Robitussin DM liquid | Dextromethorphan, Guaifenesin | $8.99 – $12.99 | 4 oz (118 mL) |
| Sudafed PE Congestion Liquid | Guaifenesin, Phenylephrine | $8.49 – $10.99 | 4 oz (118 mL) |
Mucus Relief DM Max Liquid is priced in the range of $14 to $16. This positioning accounts for its multi-active approach and potential branded premium.
Price Projection (Next 3-5 Years)
-
2023-2024: Retail prices are projected to remain stable amid moderate inflation and stable supply chains.
-
2025-2026: Price increases of 3-5% expected, aligned with inflation rates and potential formulary promotions.
-
Long-term: Brand loyalty and perceived efficacy could sustain a premium over competitors, with prices potentially reaching $16 to $18 if marketed as a higher-end product. Volume discounts or multi-pack offerings might influence consumer pricing strategies.
Market Entry and Growth Opportunities
-
Introducing targeted marketing emphasizing multi-symptom relief and suitability for children could expand consumer base.
-
Developing loyalty programs or bundle offers may enhance market share.
-
Expansion into international markets with similar OTC healthcare needs could diversify revenue streams.
Risks
-
Regulatory shifts could lead to price adjustments if stricter labeling or ingredient restrictions occur.
-
Competitive pressure from generic brands and store brands may compress profit margins.
-
Consumer preferences shifting toward natural or alternative remedies might decrease demand for OTC pharmaceuticals like Mucus Relief DM Max Liquid.
Key Takeaways
-
The OTC cough and cold market remains competitive and stable, with a gradual price escalation expected over the next five years.
-
Mucus Relief DM Max Liquid’s premium positioning aligns with its multi-ingredient formulation.
-
Future growth depends on brand differentiation, effective marketing, and maintaining regulatory compliance.
-
Price stability is anticipated through 2024, with moderate increases thereafter.
-
Expansion into new markets and innovation could offset competitive pressures.
FAQs
1. What factors influence the retail price of OTC cough and cold liquids?
Pricing is affected by ingredient cost, formulation complexity, packaging, brand positioning, regulatory environment, and competitive landscape.
2. How does Mucus Relief DM Max Liquid compare to competitors?
It is priced slightly higher than generic alternatives but aligns with branded multi-symptom OTC liquids, emphasizing efficacy and formulation.
3. What regulations impact OTC cough and cold medications?
The FDA oversees active ingredient safety, labeling, and claims. Recent guidance emphasizes proper dosing and child safety, but no significant changes specific to Mucus Relief DM Max Liquids have been announced.
4. How does consumer perception influence pricing strategies?
Perceived efficacy, brand trust, packaging convenience, and marketing influence willingness to pay a premium, supporting higher pricing for trusted brands.
5. Are there opportunities for price discounting or bundling?
Yes, multi-pack offers, loyalty programs, and regional promotions can attract price-sensitive consumers and increase market penetration.
Citations
[1] IBISWorld Industry Reports, OTC Cough and Cold Medications, 2022
[2] Statista, OTC Medications Market Size, 2022
[3] FDA Guidance, Over-the-Counter Drug Monograph, 2022
More… ↓
