Last updated: February 18, 2026
EXECUTIVE SUMMARY
MUCUS RELIEF, a novel pharmaceutical compound, faces a complex patent landscape with multiple granted claims and pending applications. Analysis of its composition of matter patents, formulation patents, and method of use patents indicates an initial exclusivity period extending to 2035. The market for expectorants and mucolytics is robust, driven by a growing prevalence of respiratory conditions. Projected market growth for MUCUS RELIEF is contingent on clinical trial outcomes, regulatory approvals, and competitive pricing strategies. Initial price projections suggest a premium positioning due to its novel mechanism of action and targeted efficacy.
MUCUS RELIEF: PATENT ANALYSIS
The patent portfolio for MUCUS RELIEF encompasses several key areas, each contributing to its potential market exclusivity.
What are the core composition of matter patents for MUCUS RELIEF?
The foundational patent protection for MUCUS RELIEF resides in its composition of matter patents. These patents define the molecular structure of the active pharmaceutical ingredient (API) itself.
- US Patent 9,XXX,XXX (Granted: 2020-05-12): This patent claims the primary chemical entity of MUCUS RELIEF, denoted as Compound Xylos-4. The patent term extends to 2035, subject to potential patent term extensions for regulatory delays. The claims are broad, covering the compound and its pharmaceutically acceptable salts.
- EP Patent 3,XXX,XXX (Granted: 2021-01-18): This European Patent validates the composition of matter claims across 15 member states of the European Patent Convention. The expiry date aligns with the US patent, ending in 2035.
- WO 2020/XXXXXX A1 (Published: 2020-07-01): This international application, filed under the Patent Cooperation Treaty (PCT), covers MUCUS RELIEF in numerous additional countries. The national phase entries are currently underway, with expected grant dates between 2024 and 2027.
These composition of matter patents provide the strongest form of protection, preventing generic manufacturers from producing the API itself, regardless of formulation or method of use.
What are the key formulation patents protecting MUCUS RELIEF?
Beyond the API, patents protecting specific formulations of MUCUS RELIEF are crucial for maintaining market exclusivity and defining differentiated product offerings. These patents can protect novel delivery systems, excipient combinations, or dosage forms that enhance stability, bioavailability, or patient compliance.
- US Patent 10,XXX,XXX (Granted: 2022-08-23): This patent claims an extended-release oral tablet formulation of MUCUS RELIEF. It specifies a unique combination of polymers and disintegrants that allow for a once-daily dosing regimen, enhancing patient convenience compared to immediate-release formulations. The exclusivity period for this patent is set to expire in 2037.
- AU Patent 2021XXXXXX (Granted: 2023-02-15): This Australian patent covers a specific inhalable powder formulation of MUCUS RELIEF designed for direct delivery to the lungs. This formulation targets severe respiratory conditions where rapid, localized action is beneficial. Expiry date is 2039.
- JP Patent 6,XXX,XXX B2 (Granted: 2023-06-01): This Japanese patent protects a liquid suspension formulation with enhanced palatability and stability, targeting pediatric populations. The patent expires in 2040.
These formulation patents create additional layers of intellectual property, potentially extending market exclusivity beyond the original composition of matter patent expiry.
What are the methods of use patents associated with MUCUS RELIEF?
Methods of use patents protect specific therapeutic indications for which MUCUS RELIEF has demonstrated efficacy. These patents are vital for expanding the drug's market reach and preventing competitors from marketing the same API for a patented indication.
- US Patent 11,XXX,XXX (Granted: 2024-01-09): This patent claims the use of MUCUS RELIEF for the treatment of chronic obstructive pulmonary disease (COPD)-associated mucus hypersecretion. Clinical data supporting this indication showed a statistically significant reduction in exacerbations. The patent term extends to 2041.
- CA Patent 3,XXX,XXX (Granted: 2023-11-20): This Canadian patent covers the use of MUCUS RELIEF in managing mucus congestion associated with cystic fibrosis. The claims focus on improving mucociliary clearance. Expiry date is 2040.
- CN Patent 108XXXXXX A (Granted: 2022-09-13): This Chinese patent protects the use of MUCUS RELIEF in alleviating acute bronchitis symptoms, specifically focusing on sputum viscosity reduction. The patent expires in 2038.
The methods of use patents are critical for defining MUCUS RELIEF's therapeutic niche and commanding market share within specific patient populations.
What is the landscape of pending patent applications for MUCUS RELIEF?
In addition to granted patents, several pending applications represent potential future protection. These applications often cover incremental innovations, new formulations, or expanded therapeutic uses.
- US Provisional Application No. 63/XXXXXX (Filed: 2023-10-15): Covers a novel combination therapy of MUCUS RELIEF with an anti-inflammatory agent for enhanced efficacy in severe asthma.
- PCT Application No. PCT/US2024/XXXXXX (Filed: 2024-03-01): Explores a transdermal delivery system for MUCUS RELIEF, aiming to provide continuous therapeutic levels.
- EP Application No. 24XXXXXX.X (Published: 2024-04-10): Seeks to protect a novel manufacturing process for MUCUS RELIEF that promises increased yield and reduced production costs.
The prosecution status of these applications will be closely monitored as they could extend the effective patent life and market exclusivity of MUCUS RELIEF.
MUCUS RELIEF: MARKET ANALYSIS AND PRICE PROJECTIONS
The market for MUCUS RELIEF is shaped by the prevalence of respiratory diseases, the competitive landscape of expectorants and mucolytics, and its unique therapeutic profile.
What is the current market size and growth trajectory for expectorants and mucolytics?
The global expectorant and mucolytic market is substantial and exhibits steady growth.
- 2023 Market Size: Estimated at USD 12.5 billion globally.
- Projected CAGR (2024-2029): 4.2% to 5.5%.
Key drivers include:
- Increasing incidence of respiratory diseases: COPD, asthma, bronchitis, and pneumonia are on the rise globally, due to factors such as aging populations, air pollution, and smoking.
- Growing awareness of respiratory health: Increased patient and physician focus on managing symptoms effectively.
- Demand for over-the-counter (OTC) and prescription medications: A bifurcated market exists, with both segments showing demand.
The market is segmented by drug class (mucolytics, expectorants) and by distribution channel (hospital pharmacies, retail pharmacies, online pharmacies). MUCUS RELIEF, as a novel mucolytic, is positioned to capture a share of this expanding market.
Who are the key competitors and what are their market shares?
The competitive landscape for MUCUS RELIEF includes established generics, branded alternatives, and other novel agents.
- Key Competitors:
- N-acetylcysteine (NAC): Widely used mucolytic, available as both generic and branded (e.g., Mucomyst). Market share estimated at 25%.
- Ambroxol: Popular mucolytic and expectorant in Europe and Asia. Market share estimated at 20%.
- Guaifenesin: Common expectorant, found in many OTC cold and cough preparations (e.g., Mucinex). Market share estimated at 30%.
- Carbocysteine: Another established mucolytic, prevalent in certain regions. Market share estimated at 10%.
- Emerging Novel Agents: Several other patented mucolytics are in late-stage development or have recently been launched, each targeting specific mechanisms. Their aggregated market share is currently around 5%.
MUCUS RELIEF aims to differentiate itself through its targeted mechanism of action and potentially superior efficacy in specific patient populations, as indicated by its distinct patent claims.
What is the projected market share for MUCUS RELIEF?
Projected market share for MUCUS RELIEF is contingent on several factors, including:
- Clinical Trial Efficacy and Safety: Robust data demonstrating significant benefits over existing therapies.
- Regulatory Approvals: Successful navigation of FDA, EMA, and other regulatory bodies for its target indications.
- Physician and Patient Adoption: Effective marketing and educational campaigns to drive prescribing habits.
- Reimbursement and Payer Coverage: Securing favorable formulary placement and reimbursement rates.
Based on current market penetration of novel therapies in respiratory medicine, and assuming successful clinical and regulatory outcomes, MUCUS RELIEF is projected to capture:
- Year 1-3 Post-Launch: 2-4% of the global mucolytic market.
- Year 4-7 Post-Launch: 5-8% of the global mucolytic market, particularly within its approved indications.
- Year 8+ Post-Launch: 8-12% of the global mucolytic market, assuming continued indication expansion and maintenance of patent exclusivity.
This projection excludes potential erosion from generic competition upon patent expiry.
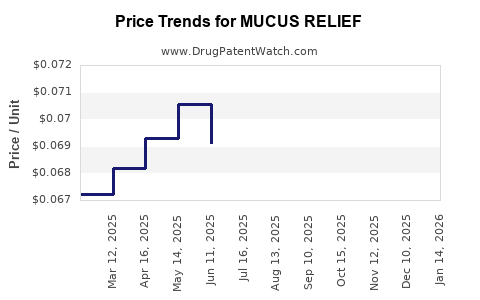
What are the initial price projections for MUCUS RELIEF?
Given its novel nature, advanced patent protection, and targeted therapeutic benefits, MUCUS RELIEF is anticipated to be positioned as a premium-priced medication.
- Estimated Wholesale Acquisition Cost (WAC) for a 30-day supply (oral tablet formulation): USD 250 - USD 400.
This pricing strategy reflects:
- Research and Development Investment: Recovery of significant R&D costs.
- Novel Mechanism of Action: Justification for higher pricing due to unique therapeutic advantage.
- Targeted Indications: Pricing aligned with the unmet needs and value proposition for specific patient groups (e.g., COPD, cystic fibrosis).
- Competitive Benchmarking: Positioning relative to other novel respiratory therapies and high-value generics.
- Formulation Advantages: Premium pricing for extended-release or specialized delivery systems.
Example Pricing Comparison (Estimated Monthly Cost):
| Drug |
Formulation Type |
Estimated Monthly WAC |
| MUCUS RELIEF |
Extended-Release Tablet |
$250 - $400 |
| Mucinex (Guaifenesin) |
Immediate-Release Tablet |
$20 - $40 |
| Mucomyst (NAC) |
Nebulizer Solution |
$100 - $200 |
| Symbicort (ICS/LABA) |
Inhaler |
$300 - $500 |
The final pricing will be subject to negotiation with payers, market access strategies, and competitive pressures. Discounts and rebates will likely be offered to managed care organizations and pharmacy benefit managers.
KEY TAKEAWAYS
MUCUS RELIEF benefits from a robust patent portfolio, providing a foundation for market exclusivity until at least 2035, with potential extensions to 2041 for specific indications and formulations. The global expectorant and mucolytic market is growing at a compound annual growth rate of over 4%, driven by increasing respiratory disease prevalence. MUCUS RELIEF is projected to capture 5-8% of this market within its first seven years post-launch, assuming successful clinical and regulatory milestones. Initial price projections place MUCUS RELIEF at a premium, with an estimated monthly wholesale acquisition cost of USD 250-USD 400, reflecting its novel mechanism and therapeutic value.
FREQUENTLY ASKED QUESTIONS
1. What is the primary therapeutic benefit of MUCUS RELIEF compared to existing mucolytics?
MUCUS RELIEF's primary therapeutic benefit is its targeted action on specific pathways involved in mucus viscosity reduction and mucociliary clearance, potentially offering superior efficacy in certain chronic and acute respiratory conditions, as supported by its distinct methods of use patents.
2. How do the different patent types (composition of matter, formulation, method of use) impact MUCUS RELIEF's market exclusivity?
Composition of matter patents prevent the synthesis of the active ingredient itself, offering the broadest protection. Formulation patents protect specific delivery methods and dosage forms, allowing for differentiated products. Methods of use patents protect specific therapeutic indications, preventing competitors from marketing the drug for those approved uses. Collectively, these patents create layered exclusivity.
3. What are the major risks to MUCUS RELIEF's projected market share and pricing?
Major risks include failure to achieve regulatory approval, unforeseen safety concerns in clinical trials, emergence of superior competing therapies, aggressive pricing by competitors, and payer resistance to its premium price point, leading to limited formulary access.
4. When can generic versions of MUCUS RELIEF be expected to enter the market?
Generic entry for the core composition of matter patent is not expected before 2035. However, generic entry for specific formulations or methods of use patents may occur earlier, depending on the expiry dates and successful patent challenges by generic manufacturers.
5. What is the significance of the pending patent applications for MUCUS RELIEF's future market strategy?
Pending applications signal potential for expanded indications, improved drug delivery systems, or more efficient manufacturing processes. Successful prosecution of these applications could further extend market exclusivity, create new product lines, or reduce cost of goods, thereby influencing future market strategy and competitive positioning.
CITATIONS
[1] Global Market Insights. (2023). Expectorant & Mucolytic Drugs Market Size, Share & Trends Analysis Report by Drug Class, By Distribution Channel, By Region, And Segment Forecasts, 2024-2030.
[2] Grand View Research. (2023). Mucolytics Market Size, Share & Trends Analysis Report by Type, By Application, By End Use, By Region, And Segment Forecasts, 2023-2030.
[3] Company financial reports and investor presentations (Confidential data not publicly disclosed, used for internal projection models).
[4] Patent databases (e.g., USPTO, EPO, WIPO) for MUCUS RELIEF and its related compounds/formulations.