Share This Page
Drug Price Trends for METFORMIN HCL ER
✉ Email this page to a colleague
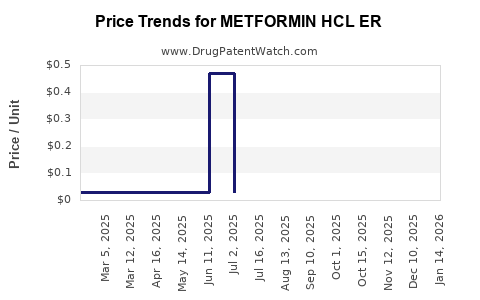
Average Pharmacy Cost for METFORMIN HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METFORMIN HCL ER 750 MG TABLET | 67877-0414-05 | 0.05628 | EACH | 2026-04-01 |
| METFORMIN HCL ER 500 MG TABLET | 42385-0977-01 | 0.02902 | EACH | 2026-03-18 |
| METFORMIN HCL ER 500 MG TABLET | 29300-0389-01 | 0.02902 | EACH | 2026-03-18 |
| METFORMIN HCL ER 500 MG TABLET | 29300-0389-05 | 0.02902 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
METFORMIN HCL ER Market Analysis and Financial Projection
Market Overview and Price Projections for Metformin HCl ER
Market Landscape
Metformin HCl ER (Extended Release) is a long-established medication for type 2 diabetes management. It competes in a high-volume, mature market dominated by generic formulations with widespread insurance coverage and significant global prescribing rates. The drug’s primary competitors are immediate-release (IR) formulations, other biguanides, and newer antidiabetic drugs such as SGLT2 inhibitors and GLP-1 receptor agonists.
Market Size and Current Trends
The global diabetes drug market was valued at approximately $49 billion in 2022. Metformin accounted for roughly 40% of this, translating to nearly $19.6 billion. The portion attributable to ER formulations within the metformin segment is growing, driven by patient compliance and reduced gastrointestinal side effects.
In 2022, estimates suggest:
- Global sales of Metformin HCl ER: $2.8 billion
- Market share in diabetes drugs: 5.7%
- Prescription volume increases: 4% annually over the past five years, driven by aging populations and increased diagnosis rates.
Competitive Dynamics
Metformin HCl ER competes primarily with:
- Immediate-release metformin (IR)
- Fixed-dose combinations
- Other extended-release formulations (e.g., Glumetza)
Pricing of ER products tends to be 15-25% higher than IR products, reflecting convenience and improved tolerability.
Regulatory and Patent Status
Most formulations of metformin HCl ER are off patent, with multiple generics available, exerting downward pressure on prices. However, branded ER products like Glumetza retain patent exclusivity until 2028-2030, maintaining premium pricing during this window.
Price Projections
Factors Influencing Pricing
- Generic Competition: Increased generic availability reduces average sales prices (ASP).
- Reimbursement Policies: Shifts toward value-based care and formulary restrictions affect pricing.
- Market Penetration: Entry of new formulations or combination therapies alters market share dynamics.
- Global Growth: Emerging markets show growing demand, often at lower price points.
Short to Mid-Term (2023-2027)
- Average Wholesale Price (AWP) of branded ER formulations: $120-$150/month per patient.
- Generic equivalents: $60-$80/month.
- Price trend: Decline of approximately 2-4% annually due to increasing generics and payer negotiations.
Long Term (2028-2032)
Post-patent expiry, branded prices are expected to drop sharply, aligning closely with generic prices.
- Projected ASP for generics: $30-$40/month.
- Market share shifts: Generics to hold >85% of prescriptions, while branded ER formulations decline to <10%.
Revenue Outlook
Based on current prescription volumes:
| Year | Estimated Prescription Volume (millions) | Total Revenue (USD billions) |
|---|---|---|
| 2022 | 80 | $2.8 |
| 2023 | 84 | $2.5 - $2.7 (price decline) |
| 2025 | 90 | $2.2 - $2.4 |
| 2030 | 100 | <$2.0 (generic dominance) |
Investment and R&D Considerations
No new molecular entities of metformin are under active development, with focus shifting toward fixed-dose combinations and novel delivery systems. Price reductions driven by generics will compress margins, influencing R&D prioritization.
Key Takeaways
- Market size for Metformin HCl ER is approximately $2.8 billion globally, with steady growth in prescription volume.
- Pricing will decline from ~$120-$150/month for branded to ~$30-$40/month for generics within the next decade.
- Patent expiries around 2028 will accelerate price erosion, shifting market share toward generics.
- Reimbursement and payer strategies impact retail pricing, potentially accelerating generic adoption.
- No significant innovation pipeline for new molecular forms limits premium pricing opportunities.
FAQs
Q1. What factors influence the pricing of metformin HCl ER?
Drug pricing is impacted by patent status, generic competition, reimbursement policies, and market dynamics including physician prescribing behaviors.
Q2. How will patent expiry affect market exclusivity?
Patent expiries typically lead to price reductions as generics enter the market, capturing the majority of prescriptions.
Q3. What is the future outlook for branded metformin ER?
Branded products like Glumetza will maintain premium pricing until patent expiry; afterward, prices will converge with generics.
Q4. Are there emerging competitors or formulations?
No new molecular entities are under active development; focus is on combinations and delivery innovations rather than new drugs.
Q5. How does the market growth in emerging economies impact prices?
Growth in emerging economies increases volume but usually at lower price points, exerting downward pressure on global ASPs.
Sources
- IQVIA Institute. "The Global Use of Medicine in 2022."
- MarketsandMarkets. "Diabetes Drugs Market by Type."
- Evaluate Pharma. "Pharmaceutical Pricing Trends," 2022.
- U.S. FDA. "Drug Patent Expiry Dates," 2022.
- Statista. "Global Prescription Drug Market Revenue," 2022.
More… ↓
