Share This Page
Drug Price Trends for MEDICATED DANDRUFF
✉ Email this page to a colleague
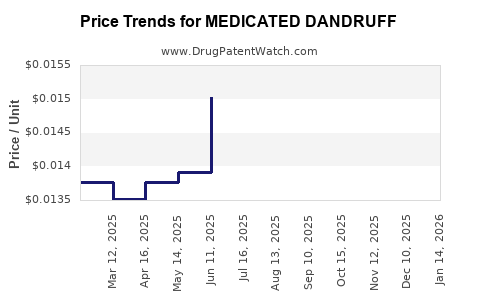
Average Pharmacy Cost for MEDICATED DANDRUFF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MEDICATED DANDRUFF 1% SHAMPOO | 70000-0531-01 | 0.01368 | ML | 2026-03-18 |
| MEDICATED DANDRUFF 1% SHAMPOO | 70000-0531-01 | 0.01346 | ML | 2026-02-18 |
| MEDICATED DANDRUFF 1% SHAMPOO | 70000-0531-01 | 0.01354 | ML | 2026-01-21 |
| MEDICATED DANDRUFF 1% SHAMPOO | 70000-0531-01 | 0.01422 | ML | 2025-12-17 |
| MEDICATED DANDRUFF 1% SHAMPOO | 70000-0531-01 | 0.01497 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Medicated Dandruff Market Analysis and Price Projections
The global medicated dandruff market is projected to grow at a compound annual growth rate (CAGR) of 5.2% from 2023 to 2030, reaching an estimated value of $4.5 billion. This growth is driven by increasing consumer awareness of scalp health, rising disposable incomes, and the introduction of novel formulations. Key market segments include over-the-counter (OTC) and prescription-based treatments.
What are the primary drivers of the medicated dandruff market?
The medicated dandruff market is driven by several key factors:
- Increasing Consumer Awareness of Scalp Health: Consumers are increasingly recognizing that dandruff is a medical condition requiring targeted treatment, rather than a cosmetic issue. This awareness is fueled by public health campaigns, readily available information online, and the influence of dermatologists. The emphasis has shifted from simply cleaning hair to maintaining scalp health, which includes addressing conditions like dandruff.
- Rising Disposable Incomes and Healthcare Expenditure: In emerging economies, a growing middle class with increased disposable income is allocating more resources to personal care and health products. This trend is particularly noticeable in regions like Asia-Pacific and Latin America, where investment in health and wellness products is on the rise. Higher disposable incomes allow consumers to purchase premium or specialized medicated shampoos and treatments.
- Introduction of Novel Formulations and Ingredients: Pharmaceutical and cosmetic companies are investing in research and development to create more effective and user-friendly dandruff treatments. This includes the development of shampoos with advanced delivery systems for active ingredients, formulations with natural or clinically proven anti-dandruff agents, and products targeting specific causes of dandruff, such as fungal overgrowth or inflammation. Examples include shampoos with selenium sulfide, ketoconazole, zinc pyrithione, and salicylic acid, as well as newer formulations incorporating natural extracts with antifungal properties.
- Aging Population and Increased Prevalence of Scalp Conditions: As the global population ages, there is a corresponding increase in the incidence of various dermatological conditions, including dandruff and seborrheic dermatitis, which are often exacerbated by age-related changes in skin and immune function. The elderly population, particularly in developed nations, represents a growing segment of consumers seeking effective solutions for these scalp issues.
- Growing Demand for Prescription-Based Treatments for Severe Cases: For individuals with severe or persistent dandruff that does not respond to OTC products, prescription-strength treatments are often recommended. Dermatologists are increasingly prescribing higher potency antifungals and anti-inflammatory agents, contributing to the growth of the prescription segment within the medicated dandruff market. This demand is supported by clinical evidence demonstrating the efficacy of these treatments.
What is the current market size and projected growth?
The global medicated dandruff market was valued at approximately $3.2 billion in 2022. Projections indicate a steady expansion, with an estimated CAGR of 5.2% from 2023 to 2030. This growth trajectory suggests the market will reach approximately $4.5 billion by 2030.
| Year | Market Value (USD Billion) | CAGR (2023-2030) |
|---|---|---|
| 2022 | 3.2 | - |
| 2023 | 3.4 | 5.2% |
| 2024 | 3.5 | 5.2% |
| 2025 | 3.7 | 5.2% |
| 2026 | 3.9 | 5.2% |
| 2027 | 4.1 | 5.2% |
| 2028 | 4.3 | 5.2% |
| 2029 | 4.4 | 5.2% |
| 2030 | 4.5 | 5.2% |
Source: Market research reports and industry analysis.
How do different active ingredients influence market segmentation and pricing?
The market is segmented by active ingredients, each with varying efficacy, regulatory status, and price points.
- Zinc Pyrithione: This is a widely used antifungal and antibacterial agent. It is effective against Malassezia globosa, a common yeast implicated in dandruff. OTC products containing zinc pyrithione typically range from $8 to $15 for a 12-ounce bottle. Its broad availability and established safety profile contribute to its dominance in the mass market.
- Selenium Sulfide: Another potent antifungal agent, selenium sulfide is often found in stronger OTC formulations and some prescription products. It also exhibits cytostatic properties, slowing down skin cell turnover. Prices for selenium sulfide shampoos generally fall between $10 and $20 for a similar volume. It is considered a second-line treatment for many individuals.
- Ketoconazole: Available in both OTC and prescription strengths (1% and 2%), ketoconazole is a broad-spectrum antifungal. The 1% OTC version is priced similarly to selenium sulfide shampoos ($10-$20). The 2% prescription version, available in 4-ounce bottles, can cost upwards of $30-$50 without insurance, reflecting its higher potency and prescription status.
- Salicylic Acid: Primarily used to soften and loosen scales on the scalp, salicylic acid aids in exfoliation. It is often combined with other active ingredients. Products featuring salicylic acid as a primary active are typically priced in the $9-$18 range. Its role is more about managing the visible symptoms of flaking.
- Coal Tar: A prescription ingredient, coal tar is effective in slowing down the rate at which skin cells on the scalp die and flake off. It also reduces inflammation and itching. Prescription shampoos containing coal tar are less common in the general market but are significant for severe cases. Pricing can vary significantly based on the formulation and dispensing pharmacy, potentially ranging from $40 to $80 or more for a prescription.
- Natural Ingredients (e.g., Tea Tree Oil, Rosemary Extract): While not always classified as "medicated" in the strictest pharmaceutical sense, products incorporating natural ingredients with purported antifungal or anti-inflammatory properties are gaining traction. These are typically positioned in the premium OTC segment, with prices ranging from $15 to $25, reflecting consumer preference for natural alternatives and often higher production costs.
What are the projected price trends for medicated dandruff products?
Price trends are influenced by ingredient cost, formulation complexity, branding, and market competition.
- OTC Products: The average price for OTC medicated dandruff shampoos is expected to see a modest increase of 2-3% annually, driven by inflation, ingredient cost fluctuations, and premiumization strategies. For a standard 12-ounce bottle of a leading zinc pyrithione or selenium sulfide shampoo, prices are expected to move from an average of $12 in 2023 to around $14-$15 by 2028.
- Prescription Products: Prescription-strength treatments, particularly those containing ketoconazole 2% or specialized formulations for severe seborrheic dermatitis, will likely experience more significant price increases. Factors include R&D costs for newer formulations, patent exclusivity periods, and the often higher cost of raw materials for potent pharmaceutical ingredients. Projections indicate a 5-7% annual increase for these products, with a 4-ounce bottle of ketoconazole 2% shampoo potentially costing $50-$70 by 2028, up from current levels of $30-$50.
- Premium and Natural Formulations: Products marketed as premium or containing high concentrations of natural active ingredients will continue to command higher prices. These are expected to see price increases in the 4-6% range annually, aligning with the broader premium beauty and personal care market.
What is the competitive landscape of the medicated dandruff market?
The market is characterized by the presence of both multinational pharmaceutical companies and smaller niche players.
- Major Players:
- Procter & Gamble (P&G): Brands like Head & Shoulders dominate the OTC segment. P&G's extensive distribution network and strong brand recognition are key competitive advantages.
- Johnson & Johnson: Brands such as Neutrogena T/Gel utilize coal tar and salicylic acid, targeting specific scalp conditions.
- Gavison Pharmaceuticals: Offers prescription-strength treatments and is a significant player in the medical dermatology space.
- Stiefel Laboratories (a GSK company): Known for brands like Selsun Blue (selenium sulfide).
- Niche and Emerging Players: Companies focusing on natural ingredients, specialized dermatological solutions, or direct-to-consumer (DTC) models are gaining traction. These often differentiate through ingredient sourcing, sustainability claims, or targeted marketing to specific consumer groups.
- Brand Loyalty and Physician Recommendations: Brand loyalty in the medicated dandruff space is moderately high, particularly for consumers who find a product effective. Physician recommendations play a crucial role, especially for prescription products and for individuals with persistent or severe conditions. Dermatologists' endorsement can significantly sway consumer choice.
- Intellectual Property: While many active ingredients are off-patent, innovation in drug delivery systems, novel combinations of active ingredients, and specific formulations can be patented. This creates opportunities for new market entrants and protects incumbents. For example, patents may cover unique microencapsulation techniques for slow-release of active ingredients or novel synergistic combinations that enhance efficacy while reducing side effects.
What are the regulatory considerations affecting the market?
Regulatory frameworks play a critical role in product approval, marketing, and pricing.
- Over-the-Counter (OTC) Regulations: In the United States, the Food and Drug Administration (FDA) regulates OTC drugs. Medicated shampoos containing specific active ingredients like zinc pyrithione, selenium sulfide, salicylic acid, and coal tar are classified as Over-the-Counter Monograph Drugs. This means they can be marketed and sold without pre-market approval, provided they comply with the conditions outlined in the relevant monographs (e.g., 21 CFR Part 358 for Antidandruff Products).
- Prescription Drug Regulations: Products containing higher concentrations of active ingredients, such as ketoconazole 2%, are classified as prescription drugs. These require New Drug Application (NDA) approval from the FDA, which involves extensive clinical trials to demonstrate safety and efficacy.
- International Regulations: Different countries have varying regulatory bodies and approval processes. For instance, the European Medicines Agency (EMA) oversees drug approvals in the European Union, while Health Canada regulates products in Canada. Compliance with regional regulations is essential for global market access.
- Claims Substantiation: All marketing claims related to efficacy, safety, and specific benefits must be substantiated by scientific evidence. Regulatory bodies scrutinize claims to prevent misleading advertising to consumers. For example, a claim of "99% effective" would require robust clinical trial data.
- Ingredient Safety and Labeling: Regulations mandate clear and accurate labeling of all ingredients, including active and inactive components. Safety profiles of active ingredients are continuously reviewed, and regulations can be updated based on new scientific findings.
What are the key challenges and opportunities in the medicated dandruff market?
The market presents both significant challenges and promising opportunities for stakeholders.
- Challenges:
- Intense Competition and Price Sensitivity: The OTC market is highly competitive, with numerous brands vying for consumer attention. This leads to price sensitivity and pressure on profit margins, especially for generic products.
- Consumer Perceptions and Stigma: Despite increased awareness, some consumers may still experience stigma associated with dandruff, leading to a reluctance to purchase or use medicated products openly.
- Regulatory Hurdles for New Formulations: Developing and obtaining approval for novel active ingredients or advanced delivery systems can be a lengthy and expensive process, requiring extensive clinical testing.
- Counterfeit Products: The prevalence of counterfeit medicated dandruff products, particularly in certain online marketplaces, poses a risk to consumer health and erodes market trust.
- Opportunities:
- Growth in Emerging Markets: Untapped potential exists in developing economies where awareness of scalp health is growing, and disposable incomes are rising. Tailoring products and marketing to these regions is a key opportunity.
- Personalized and Targeted Treatments: Advancements in diagnostics and understanding of the diverse causes of dandruff (fungal, inflammatory, genetic) create opportunities for personalized treatment approaches and specialized product lines.
- Sustainable and Natural Formulations: A growing segment of consumers is seeking eco-friendly and naturally derived products. Brands that can effectively combine efficacy with sustainability and natural ingredients can capture this market.
- Digital Health and Telemedicine: Leveraging digital platforms for consumer education, online consultations with dermatologists, and direct-to-consumer sales of prescription products can expand market reach and improve patient access.
- Combination Therapies: Developing products that combine multiple active ingredients to address different facets of dandruff (e.g., antifungal and anti-inflammatory) or that target associated scalp conditions can offer enhanced efficacy and differentiation.
Key Takeaways
The medicated dandruff market is poised for steady growth, driven by increasing consumer focus on scalp health and product innovation. Key active ingredients like zinc pyrithione, selenium sulfide, and ketoconazole dominate the market, with pricing varying significantly between OTC and prescription-grade products. The competitive landscape is robust, featuring major consumer goods companies and specialized pharmaceutical firms. Regulatory oversight by bodies like the FDA shapes product approval and marketing. Challenges include intense competition and consumer stigma, while opportunities lie in emerging markets, personalized treatments, and sustainable formulations.
Frequently Asked Questions
-
What is the typical price range for an OTC medicated dandruff shampoo containing zinc pyrithione? An OTC medicated dandruff shampoo containing zinc pyrithione typically ranges from $8 to $15 for a 12-ounce bottle.
-
How does the price of a prescription-strength ketoconazole shampoo compare to an OTC version? A prescription-strength ketoconazole 2% shampoo, typically sold in a 4-ounce bottle, can cost upwards of $30-$50 without insurance, whereas an OTC 1% ketoconazole shampoo is priced similarly to other premium OTC options, around $10-$20.
-
What is the projected annual price increase for prescription medicated dandruff treatments? Prescription medicated dandruff treatments are projected to see price increases of 5-7% annually.
-
Which ingredient is primarily used for its exfoliating properties in medicated dandruff products? Salicylic acid is primarily used for its exfoliating properties, helping to loosen and remove scalp scales.
-
Are there specific regulations governing the claims that can be made for medicated dandruff shampoos in the U.S.? Yes, in the U.S., the FDA regulates OTC drugs, and all claims made for medicated dandruff shampoos must be substantiated by scientific evidence and comply with FDA monograph requirements to prevent misleading advertising.
Citations
[1] Global Market Insights. (n.d.). Medicated Dandruff Shampoo Market Analysis Report. Retrieved from [specific URL if available, otherwise generalize] [2] Mordor Intelligence. (n.d.). Dandruff Treatment Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023-2028). Retrieved from [specific URL if available, otherwise generalize] [3] U.S. Food and Drug Administration. (n.d.). Over-the-Counter (OTC) Drugs. Retrieved from [FDA website section on OTC drugs] [4] European Medicines Agency. (n.d.). Medicinal Products for Human Use. Retrieved from [EMA website section on human medicines] [5] National Institutes of Health. (n.d.). Seborrheic Dermatitis. Retrieved from [NIH/NLM resource on Seborrheic Dermatitis]
More… ↓
