Share This Page
Drug Price Trends for LEUCOVORIN CALCIUM
✉ Email this page to a colleague
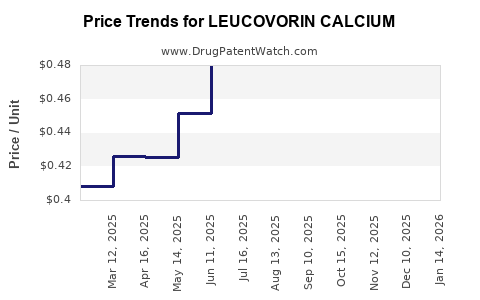
Average Pharmacy Cost for LEUCOVORIN CALCIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEUCOVORIN CALCIUM 10 MG TAB | 00054-4497-10 | 2.01608 | EACH | 2026-05-20 |
| LEUCOVORIN CALCIUM 10 MG TAB | 42806-0133-24 | 2.01608 | EACH | 2026-05-20 |
| LEUCOVORIN CALCIUM 10 MG TAB | 50742-0182-12 | 2.01608 | EACH | 2026-05-20 |
| LEUCOVORIN CALCIUM 10 MG TAB | 42806-0133-21 | 2.01608 | EACH | 2026-05-20 |
| LEUCOVORIN CALCIUM 5 MG TAB | 69315-0184-03 | 0.39212 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LEUCOVORIN CALCIUM
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LEUCOVORIN CALCIUM 50MG/VIL (PF) INJ | Sagent Pharmaceuticals | 25021-0813-10 | 10 | 15.08 | 1.50800 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| LEUCOVORIN CALCIUM 100MG/VIL (PF) INJ | Sagent Pharmaceuticals | 25021-0814-30 | 1 | 3.32 | 3.32000 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| LEUCOVORIN CALCIUM 200MG/VIL (PF) INJ | Sagent Pharmaceuticals | 25021-0815-30 | 1 | 5.98 | 5.98000 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| LEUCOVORIN CALCIUM 350MG/VIL (PF) INJ | Sagent Pharmaceuticals | 25021-0816-30 | 1 | 11.06 | 11.06000 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| LEUCOVORIN CALCIUM 500MG/VIL (PF) INJ | Sagent Pharmaceuticals | 25021-0828-50 | 1 | 18.34 | 18.34000 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Leucovorin Calcium Market Analysis and Price Projections
Leucovorin calcium, a folic acid derivative, is essential in managing methotrexate toxicity and as a potentiator in chemotherapy regimens, particularly with fluorouracil. The global market is driven by increasing cancer incidence and the demand for supportive care in oncology. Current market pricing is subject to generic competition, manufacturing costs, and regulatory landscapes in key regions.
What is the current market size and growth trajectory for leucovorin calcium?
The global leucovorin calcium market was valued at approximately $250 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.2% through 2030, reaching an estimated $332 million. Growth is primarily fueled by the rising prevalence of various cancers, including colorectal, breast, and lung cancers, which frequently utilize chemotherapy protocols involving leucovorin calcium [1].
The market's expansion is also influenced by the increasing use of leucovorin calcium as a rescue therapy following high-dose methotrexate administration in osteosarcoma and lymphoma treatment. Furthermore, ongoing research into novel therapeutic applications and combination therapies involving leucovorin calcium contributes to sustained market demand.
Key market drivers include:
- Increasing Cancer Incidence: Global cancer diagnoses are projected to rise, leading to higher demand for chemotherapy and associated supportive agents [2].
- Advancements in Oncology Treatment: The integration of leucovorin calcium into more treatment protocols enhances its market penetration.
- Generic Drug Availability: The presence of multiple generic manufacturers intensifies competition, potentially impacting pricing while increasing accessibility.
- Healthcare Infrastructure Development: Improved healthcare access in emerging economies supports market growth.
What are the key therapeutic applications and their market impact?
Leucovorin calcium's primary therapeutic applications are its use as a rescue agent for high-dose methotrexate therapy and its synergistic role with fluorouracil in treating colorectal cancer [3].
Methotrexate Rescue Therapy: This application is critical in treating osteosarcoma and certain lymphomas where high doses of methotrexate are administered to achieve therapeutic efficacy. Leucovorin calcium mitigates the hematologic and mucositis toxicity associated with these high doses. The market segment dedicated to this application is substantial, driven by the established protocols for these cancers.
Combination Therapy with Fluorouracil (5-FU): This is the most significant market driver for leucovorin calcium, particularly in the treatment of advanced colorectal cancer. The combination enhances the binding of fluorouracil to thymidylate synthase, thereby increasing its cytotoxic effect. The widespread adoption of this regimen in clinical practice underpins a consistent and robust demand for leucovorin calcium [4].
Other emerging and niche applications include:
- Treatment of Folate Deficiencies: Though less common as a primary indication, it can be used in specific cases of folate deficiency.
- Adjunctive Therapy in Other Cancers: Investigational use in combination with other chemotherapeutic agents for various solid tumors.
The market impact of these applications is directly correlated with the incidence rates of the specific cancers and the established treatment guidelines for them. Colorectal cancer remains the dominant factor influencing market volume.
Who are the major manufacturers and what is their market share?
The leucovorin calcium market is characterized by a mix of originator and generic manufacturers. The landscape is fragmented due to the drug's long-standing presence and patent expirations for the original formulations.
Key players include:
- Fresenius Kabi: A significant supplier of injectable generic drugs, including leucovorin calcium.
- Pfizer (and its subsidiary Hospira): Historically a major player, offering both branded and generic versions.
- Accord Healthcare: A prominent generic pharmaceutical company with a global presence.
- Teva Pharmaceutical Industries: A leading global generic drug manufacturer.
- BMS Pharmaceuticals: Involved in the supply chain through various intermediates or specialized formulations.
Market share is difficult to precisely quantify due to the nature of generic drug sales and the proprietary information held by manufacturers. However, it is estimated that the top five generic manufacturers account for over 60% of the global market volume.
The market share distribution is influenced by:
- Manufacturing Capacity and Cost-Effectiveness: Companies with efficient production processes and economies of scale command larger market shares.
- Regulatory Approvals and Distribution Networks: Access to key markets and established distribution channels are critical.
- Product Portfolio Diversification: Companies offering a broader range of oncology support drugs can leverage their existing customer base.
What are the current pricing trends and factors influencing price?
Current pricing for leucovorin calcium varies significantly based on the form (injectable vs. oral), dosage strength, packaging, and geographical region.
Injectable Formulations: A typical vial of leucovorin calcium (e.g., 50 mg or 100 mg) for injection can range from $15 to $50 in the United States, depending on the supplier and bulk purchasing agreements. This pricing reflects manufacturing costs, sterile processing requirements, and regulatory compliance.
Oral Formulations: Oral tablets are generally less expensive per dose but are used less frequently in high-dose rescue protocols. Pricing for oral formulations is often driven by the cost of excipients and tableting processes.
Factors influencing price include:
- Active Pharmaceutical Ingredient (API) Costs: The cost of synthesizing leucovorin calcium itself is a foundational price determinant. Fluctuations in raw material availability and cost directly impact API prices.
- Manufacturing and Overhead Costs: Energy, labor, facility maintenance, and quality control expenses contribute to the final product cost.
- Regulatory Compliance: Adherence to Good Manufacturing Practices (GMP), pharmacopeial standards (USP, EP), and the costs associated with maintaining these standards are factored in.
- Generic Competition: The presence of multiple generic suppliers leads to price erosion. Manufacturers compete on cost to gain market share.
- Supply Chain Dynamics: Disruptions in the supply chain, including transportation and raw material sourcing, can lead to price volatility.
- Reimbursement Policies: Payer policies and reimbursement rates from insurance providers and government health programs in different countries influence the effective price realized by manufacturers and distributors.
- Demand Fluctuations: Unforeseen increases or decreases in demand, often tied to shifts in cancer treatment protocols or the introduction of new therapies, can affect pricing.
What are the projected price changes for leucovorin calcium over the next five years?
The price of leucovorin calcium is expected to remain relatively stable with minor fluctuations over the next five years, largely influenced by the continued prevalence of generic competition.
Projected Price Trends (2024-2029):
- Injectable Leucovorin Calcium (50mg/100mg vial): Prices are anticipated to range between $15 and $45 per vial. A slight downward trend of 1-2% per year may be observed in highly competitive markets, offset by potential modest increases driven by rising manufacturing and regulatory compliance costs.
- Oral Leucovorin Calcium (Tablets): Prices are expected to remain stable or see a marginal decrease of less than 1% annually.
Factors supporting price stability or modest decline:
- Mature Generic Market: The drug has been off-patent for many years, with a well-established generic supply chain.
- Sufficient Manufacturing Capacity: Existing manufacturing infrastructure is generally capable of meeting current demand.
- Stable Therapeutic Utility: While new cancer treatments emerge, leucovorin calcium's established role in specific protocols ensures consistent demand without dramatic increases that would drive prices up.
Factors that could lead to minor price increases:
- Increased Raw Material Costs: Upward pressure on chemical precursor costs.
- Stricter Regulatory Requirements: New or enhanced GMP requirements could increase compliance costs for manufacturers.
- Supply Chain Bottlenecks: Global events or logistical challenges could temporarily disrupt supply and cause short-term price spikes.
- Consolidation Among Manufacturers: If market consolidation occurs, it could reduce competitive pressure and allow for minor price adjustments.
Conversely, aggressive pricing strategies by key generic players or the entry of new manufacturers with lower cost structures could exert further downward pressure on prices. The overall market volume growth, driven by cancer incidence, is expected to absorb some of the price decline effects, maintaining overall market revenue.
What is the regulatory landscape impacting leucovorin calcium production and sales?
The regulatory landscape for leucovorin calcium is governed by national and regional health authorities, primarily focusing on product quality, safety, and efficacy.
Key Regulatory Bodies and Requirements:
- U.S. Food and Drug Administration (FDA): Manufacturers must comply with FDA regulations for drug approval, manufacturing (21 CFR Part 210/211), labeling, and post-market surveillance. Generic drug applications (ANDA) are required for market entry.
- European Medicines Agency (EMA): In Europe, Marketing Authorisation Applications (MAA) are necessary. Compliance with EU GMP guidelines is mandatory for manufacturing facilities.
- Other National Regulatory Authorities: Bodies like Health Canada, the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and similar agencies in other countries have their own approval and manufacturing standards.
Impact on Production and Sales:
- Good Manufacturing Practices (GMP): Strict adherence to GMP is paramount. Facilities undergo regular inspections. Any non-compliance can lead to product recalls, import alerts, or manufacturing halts, directly impacting supply and costs.
- Pharmacopeial Standards: Leucovorin calcium must meet the purity, potency, and quality standards set by major pharmacopeias (e.g., United States Pharmacopeia (USP), European Pharmacopoeia (EP)).
- Drug Master Files (DMFs): Manufacturers often maintain DMFs with regulatory agencies, detailing the chemistry, manufacturing, and controls of the API.
- Abbreviated New Drug Applications (ANDAs): For generic versions in the U.S., ANDAs demonstrating bioequivalence to the reference listed drug are required, a process that involves significant R&D and regulatory submission costs.
- Import/Export Regulations: International trade of leucovorin calcium is subject to customs regulations, tariffs, and specific import permits in various countries.
- Labeling and Packaging Requirements: Regulations dictate accurate labeling, including dosage, warnings, and expiration dates, as well as secure and compliant packaging.
The regulatory environment fosters product quality but also increases the barrier to entry and operational costs for manufacturers. Maintaining compliance is an ongoing expense that is indirectly factored into product pricing. Changes in regulatory stringency can influence manufacturing processes and associated costs.
What are the supply chain vulnerabilities for leucovorin calcium?
The supply chain for leucovorin calcium, like many pharmaceuticals, is susceptible to several vulnerabilities.
Key Vulnerabilities:
- API Sourcing: The synthesis of leucovorin calcium relies on specific chemical precursors. A significant portion of these intermediates is manufactured in a limited number of countries, primarily in Asia. Disruptions in production in these regions due to geopolitical events, environmental regulations, or health crises can create API shortages [5].
- Manufacturing Concentration: While many finished dosage form manufacturers exist, the production of the API or critical intermediates may be concentrated among a few key suppliers. This lack of diversification poses a risk if a major API producer faces operational issues.
- Logistics and Transportation: Global shipping disruptions, port congestion, rising freight costs, and geopolitical instability can impact the timely delivery of both raw materials and finished products.
- Quality Control and Batch Failures: Stringent quality control measures are necessary at every stage. A single batch failure due to manufacturing or testing issues can lead to delays and supply shortages.
- Regulatory Compliance Shifts: Changes in manufacturing site inspections or new regulatory requirements in exporting countries can halt the flow of goods.
- Inventory Management: Over-reliance on just-in-time inventory models can leave the supply chain exposed to sudden demand surges or unexpected disruptions.
Mitigation strategies employed by manufacturers and distributors include dual sourcing for critical raw materials, maintaining strategic inventory levels, robust quality assurance protocols, and diversification of manufacturing sites where feasible. However, the global nature of pharmaceutical manufacturing means that vulnerabilities remain inherent.
What are the future market opportunities and potential threats?
The market for leucovorin calcium presents both opportunities for expansion and potential threats to its current standing.
Market Opportunities:
- Emerging Markets: Increasing access to healthcare and rising cancer rates in developing economies in Asia, Africa, and Latin America represent significant growth opportunities for generic leucovorin calcium.
- Expansion of Oncology Treatment Protocols: As research uncovers new ways to utilize leucovorin calcium in combination therapies or for different cancer types, demand could increase.
- Development of Novel Delivery Systems: While less likely given the drug's established nature, innovations in oral or sustained-release formulations could create niche market segments.
- Increased Use in Preventative or Supportive Care: Further research into non-treatment-specific applications of folate support could broaden its utility.
Potential Threats:
- Development of Alternative Therapies: The emergence of novel targeted therapies or immunotherapies that reduce reliance on traditional cytotoxic chemotherapy could diminish the demand for leucovorin calcium.
- Shifts in Treatment Guidelines: Changes in oncology treatment guidelines that de-emphasize or replace leucovorin calcium-containing regimens would directly impact market volume.
- Intensifying Price Competition: Continuous pressure from an increasing number of generic manufacturers could lead to unsustainable profit margins.
- Supply Chain Disruptions: As noted, persistent or severe supply chain issues could force healthcare providers to seek alternatives or reduce usage.
- Regulatory Stringency: Unforeseen increases in regulatory compliance costs or stricter approval processes for generics could impact market accessibility and profitability.
The market will likely continue to be dominated by generic competition, with growth tied to the overall trajectory of cancer treatment and the specific role leucovorin calcium plays within it.
Key Takeaways
Leucovorin calcium's global market, valued at approximately $250 million in 2023, is projected to grow at a CAGR of 4.2% through 2030. This growth is driven by rising cancer incidence and its established role in methotrexate rescue therapy and combination treatments with fluorouracil for colorectal cancer. The market is fragmented with numerous generic manufacturers, leading to competitive pricing. Current injectable formulations range from $15 to $50 per vial, with oral forms being less expensive. Prices are expected to remain relatively stable, with minor downward pressure from generic competition and potential modest increases due to rising manufacturing and regulatory costs. Key vulnerabilities lie in API sourcing concentration and global logistics. Future opportunities exist in emerging markets and potential expansion of therapeutic uses, while threats include the development of alternative therapies and shifts in treatment guidelines.
Frequently Asked Questions
-
What is the primary driver of demand for leucovorin calcium? The primary driver is its use in oncology, specifically as a rescue agent following high-dose methotrexate therapy and as a potentiator in combination chemotherapy regimens with fluorouracil for treating cancers like colorectal cancer.
-
How does generic competition affect the price of leucovorin calcium? The presence of multiple generic manufacturers intensifies price competition, leading to a downward pressure on prices and increased accessibility for healthcare providers and patients.
-
Are there any significant supply chain risks unique to leucovorin calcium production? A notable risk is the potential concentration of active pharmaceutical ingredient (API) or critical intermediate manufacturing in specific geographic regions, making the supply chain vulnerable to disruptions in those areas.
-
What is the typical lifespan of a patent for a drug like leucovorin calcium, and how does this impact the market? Leucovorin calcium is a well-established drug with patents that expired many years ago. This has led to a mature generic market where competition is primarily based on cost and manufacturing efficiency rather than intellectual property.
-
Could the development of new cancer treatments make leucovorin calcium obsolete? While novel therapies are emerging, leucovorin calcium holds established roles in specific, widely used treatment protocols. Its obsolescence would require a fundamental shift in the standard of care for major cancers it currently serves, which is unlikely in the near to medium term.
Citations
[1] Global Cancer Observatory. (2020). Global Cancer Statistics 2020. International Agency for Research on Cancer. [2] World Health Organization. (2022). Cancer Fact Sheet. [3] Pizzo, P. A., & Poplack, D. G. (Eds.). (2016). Principles and Practice of Pediatric Oncology (7th ed.). Lippincott Williams & Wilkins. [4] De Gramont, A., Bosset, J. F., Villard, J., etc. (1997). Randomized trial comparing two dosage regimens of fluorouracil plus leucovorin in advanced colorectal cancer: a study of the Fédération Francophone de Cancérologie Digestive. Journal of Clinical Oncology, 15(3), 817-822. [5] U.S. Food and Drug Administration. (2022). Report on the Availability of Critical Medicines.
More… ↓
