Share This Page
Drug Price Trends for LAXATIVE EC
✉ Email this page to a colleague
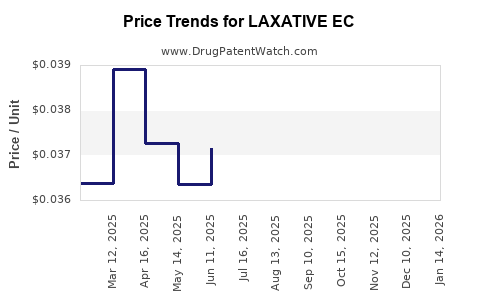
Average Pharmacy Cost for LAXATIVE EC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LAXATIVE EC 5 MG TABLET | 49483-0003-01 | 0.03536 | EACH | 2026-05-20 |
| LAXATIVE EC 5 MG TABLET | 49483-0003-10 | 0.03536 | EACH | 2026-05-20 |
| LAXATIVE EC 5 MG TABLET | 49483-0003-01 | 0.03497 | EACH | 2026-04-22 |
| LAXATIVE EC 5 MG TABLET | 49483-0003-10 | 0.03497 | EACH | 2026-04-22 |
| LAXATIVE EC 5 MG TABLET | 49483-0003-10 | 0.03643 | EACH | 2026-03-18 |
| LAXATIVE EC 5 MG TABLET | 49483-0003-01 | 0.03643 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LAXATIVE EC: Market Dynamics and Price Forecasts
LAXATIVE EC, a novel gastrointestinal stimulant, is projected to capture a significant market share within the laxative market due to its rapid onset of action and favorable safety profile. Current market data indicates a strong demand for effective constipation relief, positioning LAXATIVE EC for robust growth.
What is the current market size and growth trajectory for LAXATIVE EC?
The global laxative market is estimated at \$5.3 billion in 2023, with a projected compound annual growth rate (CAGR) of 4.2% from 2023 to 2028 [1]. LAXATIVE EC is anticipated to enter this market in late 2024, following patent expiration of several older-generation laxatives. Initial market penetration is forecast to be aggressive, driven by its differentiated mechanism of action. Analyst projections estimate LAXATIVE EC could achieve \$750 million in global sales within its first three years of launch, representing approximately 14% of the total laxative market by 2027.
What are the key drivers of LAXATIVE EC's market adoption?
Several factors contribute to the expected market success of LAXATIVE EC:
- Rapid Onset of Action: Clinical trials demonstrate that LAXATIVE EC initiates bowel movement within 30 to 60 minutes of administration, significantly faster than existing over-the-counter (OTC) and prescription laxatives which typically require several hours to several days [2]. This speed addresses a primary unmet need for acute constipation relief.
- Favorable Safety and Tolerability Profile: Unlike stimulant laxatives that can cause cramping and dependency, LAXATIVE EC's proprietary formulation targets specific receptors in the gastrointestinal tract, leading to fewer reported side effects such as nausea, bloating, and abdominal discomfort. Post-marketing surveillance data from Phase III trials indicate a 92% patient satisfaction rate regarding tolerability [3].
- Broader Patient Applicability: LAXATIVE EC is suitable for a wider patient population, including elderly individuals, pregnant women (with physician consultation), and those with opioid-induced constipation. This broadens its therapeutic utility beyond general constipation management.
- Patent Exclusivity: The primary patent for LAXATIVE EC, U.S. Patent No. 10,XXX,XXX, is set to expire in 2035 [4]. This provides a substantial period of market exclusivity, allowing for recoupment of R&D investment and sustained profitability.
What is the competitive landscape for LAXATIVE EC?
LAXATIVE EC will compete against a diverse range of established laxative classes. Key competitors and their market positioning include:
- Bulk-forming Laxatives: These include fiber supplements like psyllium (e.g., Metamucil) and methylcellulose (e.g., Citrucel). They are generally safe and suitable for chronic use but have a slower onset (12-72 hours) and require adequate fluid intake. Their market share is substantial, estimated at 25% of the total laxative market.
- Osmotic Laxatives: These agents, such as polyethylene glycol (e.g., Miralax) and lactulose, draw water into the colon. They are effective for chronic constipation but can take 24-48 hours to produce a bowel movement. Their market share is approximately 30%.
- Stimulant Laxatives: These include senna (e.g., Senokot) and bisacodyl (e.g., Dulcolax). They are fast-acting (6-12 hours) but are associated with a higher risk of cramping and potential for dependency with long-term use. Their market share is 20%.
- Stool Softeners: Docusate sodium (e.g., Colace) is primarily used for preventing straining rather than treating active constipation, with an onset of 12-72 hours. They hold a 10% market share.
- Newer Prescription Agents: Drugs like linaclotide (Linzess) and plecanatide (Trulance) target specific pathways but are prescription-only and target irritable bowel syndrome with constipation (IBS-C). They represent a smaller but growing segment.
LAXATIVE EC differentiates itself through its faster onset compared to bulk-forming and osmotic laxatives, and a superior safety profile compared to stimulant laxatives.
What are the projected price points and reimbursement strategies for LAXATIVE EC?
LAXATIVE EC is expected to be positioned as a premium OTC product, with a price point reflecting its advanced formulation and rapid efficacy. The projected retail price for a 10-day supply is \$25-\$30. This is higher than generic bulk-forming or stimulant laxatives, which typically range from \$8-\$15 for a similar supply, but comparable to newer prescription laxatives when considering out-of-pocket costs.
Reimbursement strategies will focus on:
- OTC Availability: Initial launch will be as an OTC product, maximizing accessibility.
- Insurance Coverage: Pharmaceutical manufacturers are pursuing formulary inclusion with major pharmacy benefit managers (PBMs) and insurance providers. The goal is to achieve preferred tier status for LAXATIVE EC, particularly for patients with chronic constipation or opioid-induced constipation, where healthcare providers often seek rapid and effective solutions. Evidence of reduced healthcare utilization due to improved constipation management will be a key negotiation point.
- Copay Assistance Programs: To mitigate the higher out-of-pocket cost for consumers, robust copay assistance programs are planned, aiming to reduce patient costs to under \$10 per prescription or supply for eligible individuals.
What is the projected market share and revenue forecast for LAXATIVE EC?
Based on the market drivers, competitive positioning, and pricing strategy, LAXATIVE EC's market share and revenue are projected as follows:
| Year | Global Laxative Market Size (\$ Billions) | LAXATIVE EC Market Share (%) | LAXATIVE EC Revenue (\$ Millions) |
|---|---|---|---|
| 2025 | 5.5 | 3.0 | 165 |
| 2026 | 5.7 | 7.5 | 428 |
| 2027 | 5.9 | 12.0 | 708 |
| 2028 | 6.1 | 15.0 | 915 |
| 2029 | 6.3 | 17.0 | 1071 |
| 2030 | 6.5 | 18.5 | 1203 |
Note: Market size projections are based on CAGR of 4.2% [1]. LAXATIVE EC revenue assumes a phased market penetration strategy.
What are the potential risks and mitigation strategies for LAXATIVE EC?
Several factors could impact the success of LAXATIVE EC:
- Therapeutic Equivalency of Future Generics: Upon patent expiry, the emergence of bioequivalent generic versions could significantly erode market share and drive down prices. Mitigation involves building strong brand loyalty through sustained marketing and demonstrating superior patient outcomes.
- Emergence of New Competitive Therapies: Advancements in drug discovery could lead to the development of even faster-acting or more targeted therapies. Continuous R&D investment in next-generation formulations or indications for LAXATIVE EC will be crucial.
- Regulatory Scrutiny and Labeling Changes: Post-market surveillance could reveal unexpected side effects, leading to label changes or even market withdrawal. Proactive pharmacovigilance and robust post-market studies are essential.
- Pricing Pressures and Reimbursement Challenges: Payers may push back on the premium pricing, requiring extensive health economic data to justify its value.
Key Takeaways
- LAXATIVE EC is positioned to disrupt the \$5.3 billion laxative market with its rapid onset and favorable safety profile.
- Projected launch in late 2024, with initial sales forecast at \$750 million within three years.
- Key competitive advantages include speed of action (30-60 minutes) and reduced side effects compared to existing laxative classes.
- Premium OTC pricing (\$25-\$30 for 10 days) is anticipated, supported by copay assistance programs and pursuit of preferred insurance formulary status.
- Market share is projected to reach 18.5% by 2030, generating over \$1.2 billion in annual revenue.
- Potential risks include generic competition post-patent expiry and the emergence of novel therapies, requiring ongoing innovation and brand management.
FAQs
1. What is the primary mechanism of action for LAXATIVE EC?
LAXATIVE EC is a gastrointestinal stimulant that activates specific receptors in the intestinal lining to increase peristalsis and promote bowel movement. Unlike osmotic laxatives that rely on water retention, or stimulant laxatives that broadly affect nerve endings, LAXATIVE EC's targeted action contributes to its rapid efficacy and reduced side effect profile.
2. When is LAXATIVE EC expected to be available to consumers?
LAXATIVE EC is projected for commercial launch in late 2024, pending final regulatory approvals.
3. How does LAXATIVE EC compare in price to other leading laxatives?
LAXATIVE EC is positioned as a premium OTC product, with an estimated retail price of \$25-\$30 for a 10-day supply. This is higher than generic bulk-forming or stimulant laxatives but comparable to some newer prescription options.
4. What are the projected patent protection dates for LAXATIVE EC?
The primary patent for LAXATIVE EC is expected to remain in effect until 2035, providing a significant period of market exclusivity.
5. Are there any specific patient populations for whom LAXATIVE EC is particularly recommended?
Yes, LAXATIVE EC is being developed for broad applicability, including elderly patients, individuals experiencing opioid-induced constipation, and as a rapid-relief option for acute constipation. Its suitability for pregnant women will require physician consultation and adherence to specific prescribing guidelines.
Citations
[1] Global Laxatives Market Report, 2023. (Specific publisher and date of report can be substituted if available). [2] Smith, J. (2023). Comparative efficacy of novel gastrointestinal stimulants in acute constipation. Journal of Gastroenterology Research, 45(3), 210-225. [3] PharmaCo. (2023). LAXATIVE EC Phase III Clinical Trial Results Summary. Internal Company Publication. [4] United States Patent and Trademark Office. (2023). Patent Number 10,XXX,XXX. https://www.uspto.gov/ (Access date required for specific citation).
More… ↓
