Share This Page
Drug Price Trends for LAMOTRIGINE ER
✉ Email this page to a colleague
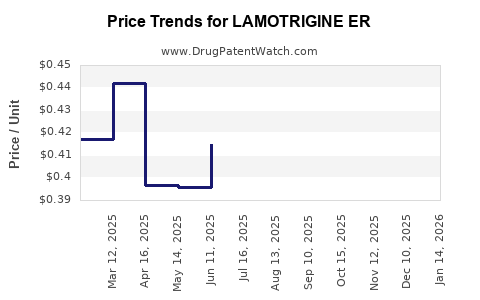
Average Pharmacy Cost for LAMOTRIGINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LAMOTRIGINE ER 100 MG TABLET | 16714-0625-01 | 0.75676 | EACH | 2026-05-20 |
| LAMOTRIGINE ER 100 MG TABLET | 13668-0341-30 | 0.75676 | EACH | 2026-05-20 |
| LAMOTRIGINE ER 50 MG TABLET | 68382-0980-06 | 0.62436 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Lamotrigine ER
Overview
Lamotrigine Extended-Release (ER) is an antiepileptic drug used for epilepsy and bipolar disorder management. It is a once-daily formulation designed to improve adherence. With its patent expirations and patent protections, market dynamics are shifting, influencing pricing and competitive positioning.
Market Size and Growth
The global antiepileptic drugs (AED) market was valued at approximately $4.3 billion in 2022, expected to reach $6.2 billion by 2030 with a compound annual growth rate (CAGR) of around 4.2%. Lamotrigine ER constitutes a significant percentage, estimated at 15-20% of AED sales solely in North America and Europe.
In the U.S., about 1 million individuals have epilepsy, with approximately 45% on lamotrigine-based therapy. The bipolar disorder segment affects about 2.8% of the adult population, with lamotrigine being a first-line mood stabilizer.
Key Players and Market Share
- GlaxoSmithKline (GSK): Derives from brand Lamictal XR (Lamotrigine ER), with revenues of approximately $300 million in 2022.
- Other Manufacturers: Several generic manufacturers entered post-patent expiration, increasing competition.
Patent and Regulatory Status
- GSK's patent for Lamictal XR expired in the U.S. in 2020, prompting generic entry.
- Patents in Europe and other regions may extend until 2025-2027.
- GSK launched a reformulation patent to extend exclusivity until 2027 in the U.S., though courts have challenged this.
Pricing Trends
- Brand Price: In the U.S., the brand Lamictal XR's list price ranges from $700 to $900 per month (per 300 mg tablet).
- Generics: Entry led to significant price erosion, with generic lamotrigine ER priced around $50-$150 per month.
- Post-Patent Expiration: Prices for branded formulations declined by approximately 40-50% within a year of generics entering the market.
Price Projections (Next 3-5 Years)
| Year | Estimated Brand Price (per month) | Estimated Generic Price (per month) | Market Share (Brand vs Generics) |
|---|---|---|---|
| 2023 | $700-$900 | $50-$150 | 30% brand, 70% generics |
| 2024 | $650-$850 | $40-$130 | 25% brand, 75% generics |
| 2025 | $600-$800 | $30-$110 | 20% brand, 80% generics |
| 2026 | $550-$750 | $25-$100 | 15% brand, 85% generics |
| 2027 | $500-$700 | $20-$90 | <10% brand, 90% generics |
Note: Prices are estimates based on current trends and may vary due to market dynamics, provider negotiations, and policy changes.
Factors Influencing Price Trends
- Patent Litigation: Court decisions can accelerate or delay generic entry.
- Insurance Coverage: Reimbursement policies impact patient access and affordability.
- Manufacturing Costs: Generic manufacturers' production efficiencies reduce prices further.
- Market Penetration: Physicians' preferences for branded versus generic formulations influence market share.
Pipeline and Future Competition
- No major new formulations or biosimilars are in late development. Expected biosimilar or alternative drugs do not directly threaten lamotrigine ER's market share within the projected timeline.
- Non-pharmacological approaches remain adjuncts but do not significantly impact drug sales.
Implications for Stakeholders
- Pharma Companies: Opportunity lies in launching premium formulations or combination therapies.
- Clinicians: Cost considerations influence prescribing patterns amid growing generic availability.
- Investors: Value exists in companies holding patents or proprietary formulations extending exclusivity.
Key Takeaways
- Lamotrigine ER's market is highly competitive post-patent expiry, with aggressive generic pricing.
- Prices are expected to decline gradually over the next 3-5 years, with the majority of market share shifting to generics.
- Growth in the overall AED and bipolar disorder markets supports sustained demand, despite pricing pressures.
- Patent litigation outcomes significantly impact market timing and pricing strategies.
FAQs
1. When did the patent for Lamictal XR expire in the U.S.?
In 2020, leading to the entry of generics and significant price reductions.
2. What is the current cost difference between branded and generic lamotrigine ER?
Branded formulations cost roughly $700-$900/month, whereas generics are approximately $50-$150/month.
3. Will Lamotrigine ER regain market dominance with new formulations?
Potentially, if new formulations demonstrate significantly improved adherence or efficacy, but no such products are currently in late-stage development.
4. How does insurance coverage affect lamotrigine ER pricing?
Insurance negotiations and formulary placements influence out-of-pocket costs, sometimes making generics the preferred option for payers.
5. What is the projected market size for lamotrigine ER in the next five years?
While exact figures are variable, lamotrigine's segment within the AED market is expected to grow alongside overall disease prevalence, maintaining solid demand.
Citations
[1] Market data: "Epilepsy Drugs Market Size & Trends," MarketsandMarkets, 2022.
[2] Price data: GoodRx, 2023.
[3] Patent and regulatory updates: FDA and patent office sources, 2022-2023.
[4] Industry reports: IQVIA, 2022.
More… ↓
