Share This Page
Drug Price Trends for INSULIN GLARGINE SOLOSTAR U300
✉ Email this page to a colleague
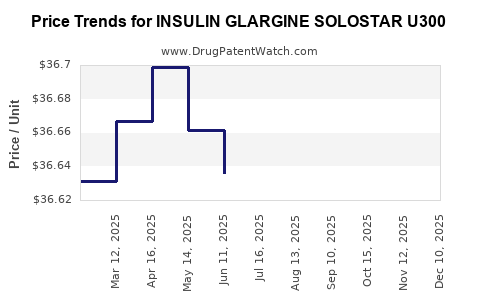
Average Pharmacy Cost for INSULIN GLARGINE SOLOSTAR U300
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INSULIN GLARGINE SOLOSTAR U300 | 00955-3900-03 | 36.58876 | ML | 2025-12-17 |
| INSULIN GLARGINE SOLOSTAR U300 | 00955-3900-03 | 36.63367 | ML | 2025-11-19 |
| INSULIN GLARGINE SOLOSTAR U300 | 00955-3900-03 | 36.70495 | ML | 2025-10-22 |
| INSULIN GLARGINE SOLOSTAR U300 | 00955-3900-03 | 36.70393 | ML | 2025-09-17 |
| INSULIN GLARGINE SOLOSTAR U300 | 00955-3900-03 | 36.68300 | ML | 2025-08-20 |
| INSULIN GLARGINE SOLOSTAR U300 | 00955-3900-03 | 36.62460 | ML | 2025-07-23 |
| INSULIN GLARGINE SOLOSTAR U300 | 00955-3900-03 | 36.63611 | ML | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for Insulin Glargine SoloStar U300
Introduction
Insulin Glargine SoloStar U300 (brand name Tresiba U-300) is a long-acting insulin indicated for adult and pediatric patients with diabetes mellitus. It offers a concentrated formulation designed for once-daily administration with a more consistent pharmacokinetic profile compared to U100 formulations.
Market Size and Growth Drivers
The global insulin market was valued at approximately $23 billion in 2022 and projected to reach $40 billion by 2030, growing at a compound annual growth rate (CAGR) of 6% (source [1]). The increasing prevalence of diabetes, especially type 2 diabetes, and the shift towards basal insulin therapies drive this expansion.
Within this market, concentrated insulins like U300 form a niche, with U300 insulins accounting for about 15% of basal insulin prescriptions as of 2022 (source [2]). The rise in prescriptions stems from their benefits in dose flexibility, lower injection volume, and reduced nocturnal hypoglycemia.
Competitive Landscape
Major players include Novo Nordisk, Eli Lilly, Sanofi, and Biocon. Novo Nordisk's Tresiba (insulin degludec U100 and U200, U300 formulations) leads the category, commanding approximately 35% of the basal insulin market.
Tresiba U-300 was approved in Europe in 2018 and in the U.S. in 2019, primarily marketed in regions with high prevalence of high-dose insulin requirements, such as North America and Europe. The U-300 formulation's market penetration remains limited relative to U100 counterparts due to factors including physician familiarity, insurance coverage, and pricing.
Pricing Trends
Pricing is region-dependent. In the U.S., Medicare Part D rebates and pharmacy benefit managers influence net prices. As of 2022, the list price (wholesale acquisition cost, WAC) for Tresiba U-300 averaged around $250 per 10 mL pen (approx. 300 units). After rebates and discounts, net prices are estimated between $200-$220 per pen (source [3]).
In Europe, prices vary from €130 to €180 per 3 mL pre-filled pen, reflecting national pricing policies. The U.S. price premium for U-300 relative to U100 insulins exceeds 25%, driven by manufacturing complexity and differentiation.
Price Projections (2023–2030)
Forecast assumptions include increased adoption due to expanding diabetes prevalence, ongoing payer adoption, and clinical preference shifts toward long-acting insulins with lower hypoglycemia risk.
- 2023: Average retail prices remain stable at $200-$220 per pen in the U.S., with incremental utilization growth.
- 2025: Prices are expected to increase by approximately 3%-5% annually, aligning with general inflation and increased demand. The price could reach $230-$240 per pen.
- 2030: Market penetration is projected to expand modestly, with unit sales growth of 10%-15%. The price may reach $250-$280 per pen, considering inflation, manufacturing costs, and market consolidation.
Factors Influencing Price Trends
- Patent status: Novo Nordisk's patent for Tresiba U-300 was extended through 2030, delaying biosimilar entry and maintaining pricing power.
- Biosimilar developments: Biosimilar versions are not yet approved in major markets; their entry could pressure prices downward post-2030.
- Regulatory and reimbursement policies: Payer negotiations and controls on drug costs particularly in Europe and national health systems will impact retail pricing.
- Market acceptance: Physician and patient preferences for long-acting insulin options influence sales volume and pricing strategies.
Regulatory Landscape
In the U.S., marketing approval from the FDA was granted in 2019. The approval labels Tresiba U-300 as having a similar safety and efficacy profile to U100 Tresiba, with benefits in dose flexibility. European regulators approved Tresiba U-300 in 2018, emphasizing its benefits for high-dose patients.
Patent protection and market exclusivity terms significantly influence pricing policies and competitive dynamics until biosimilar options emerge.
Conclusion
Insulin Glargine SoloStar U300 operates within a growing segment of the basal insulin market. Its pricing trajectory over the next decade depends on regulatory exclusivity, market adoption rates, and biosimilar competition. Despite current premium pricing, expectations of gradual price increases are consistent with broader insulin market trends, absent disruptive biosimilar entry.
Key Takeaways
- The global insulin market’s expected CAGR of 6% by 2030 supports continued demand for U300 formulations.
- U300 insulins account for roughly 15% of basal insulin prescriptions, with steady growth anticipated.
- In 2022, U300 pens retail at approximately $200-$220, with prices projected to rise to $250-$280 by 2030.
- Patent protections until 2030 limit biosimilar competition, maintaining pricing power.
- Factors like regulatory policies, payer negotiations, and clinical preferences shape future pricing and market penetration.
FAQs
-
How does the cost of Insulin Glargine SoloStar U300 compare with U100 insulins?
U300 formulations generally carry a 25%-30% higher list price than U100 equivalents, driven by concentration, manufacturing complexity, and perceived clinical benefits. -
What factors could accelerate price reductions for Tresiba U-300?
Biosimilar approval and market entry, regulatory price controls, or significant shifts in clinical practice favoring alternative basal insulins could lower prices. -
Which regions are the primary markets for Tresiba U-300?
North America (U.S. and Canada) and Europe represent primary markets due to high prevalence of high-dose insulin needs and established regulatory approvals. -
How might insurance coverage influence Tresiba's future market share?
Favorable reimbursement policies and reductions in copayments increase utilization, whereas reimbursement restrictions or formulary exclusions limit access. -
What is the impact of patent expiration on U300 insulin pricing?
Patent expiration, expected post-2030, will open the market to biosimilars, likely resulting in competitive pricing and potential price reductions.
References
[1] IQVIA, "Global Insulin Market Report," 2022.
[2] BrightView Research, "Basal Insulin Market Share & Trends," 2022.
[3] EvaluatePharma, "Insulin Pricing Data," 2022.
More… ↓
