Drug Price Trends for INFANT IBUPROFEN
✉ Email this page to a colleague
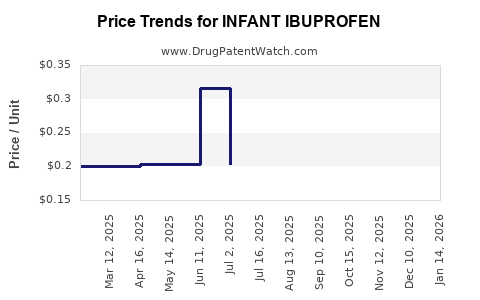
Average Pharmacy Cost for INFANT IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INFANT IBUPROFEN 50 MG/1.25 ML | 70000-0298-01 | 0.19794 | ML | 2024-11-20 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 00904-5463-35 | 0.28150 | ML | 2024-11-20 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 24385-0550-10 | 0.19794 | ML | 2024-11-20 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 45802-0057-05 | 0.28150 | ML | 2024-11-20 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 70000-0298-01 | 0.19842 | ML | 2024-10-23 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 00904-5463-35 | 0.27827 | ML | 2024-10-23 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 45802-0057-05 | 0.27827 | ML | 2024-10-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |


