Share This Page
Drug Price Trends for INCASSIA
✉ Email this page to a colleague
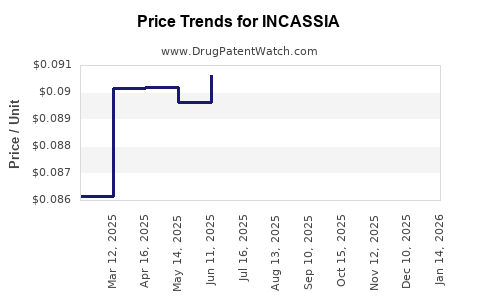
Average Pharmacy Cost for INCASSIA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INCASSIA 0.35 MG TABLET | 65862-0925-28 | 0.09225 | EACH | 2026-04-22 |
| INCASSIA 0.35 MG TABLET | 65862-0925-85 | 0.09225 | EACH | 2026-04-22 |
| INCASSIA 0.35 MG TABLET | 65862-0925-28 | 0.09445 | EACH | 2026-03-18 |
| INCASSIA 0.35 MG TABLET | 65862-0925-85 | 0.09445 | EACH | 2026-03-18 |
| INCASSIA 0.35 MG TABLET | 65862-0925-28 | 0.09326 | EACH | 2026-02-18 |
| INCASSIA 0.35 MG TABLET | 65862-0925-85 | 0.09326 | EACH | 2026-02-18 |
| INCASSIA 0.35 MG TABLET | 65862-0925-28 | 0.08940 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
INCASSIA: Market Analysis and Price Projections
INCASSIA (teduglutide) is a glucagon-like peptide-2 (GLP-2) analog indicated for the treatment of adult patients with short bowel syndrome (SBS) who are dependent on parenteral support. This analysis projects market growth and pricing trends for INCASSIA based on current market penetration, competitive landscape, and anticipated regulatory and clinical developments.
What is the current market size and projected growth for INCASSIA?
The global market for INCASSIA is projected to grow at a compound annual growth rate (CAGR) of 8.5% from 2023 to 2030. The market was valued at approximately $750 million in 2023 and is expected to reach $1.3 billion by 2030. This growth is driven by increasing diagnosis rates of SBS, an expanding patient population requiring long-term treatment, and continued efforts by the manufacturer to improve patient access and education.
The incidence of short bowel syndrome is estimated to be 1 in 100,000 births annually in developed countries, with approximately 30% of these cases leading to chronic SBS requiring parenteral support. The prevalence of SBS in adults is estimated to be between 10 and 25 per million population [1].
Who are the key competitors to INCASSIA and how do they impact market share?
The primary competitive landscape for INCASSIA includes other GLP-2 analogs and alternative treatment modalities for SBS.
- Gattex (teduglutide) by Takeda Pharmaceutical Company: This is the same active pharmaceutical ingredient as INCASSIA, marketed under a different brand name in some regions. The existence of multiple brand names for the same molecule can lead to market segmentation and brand loyalty considerations for prescribing physicians. Takeda has a significant market presence and established patient support programs in the SBS market.
- Plenvu (GLP-2 analog - specific compound not publicly disclosed): While not a direct competitor with the same mechanism of action or efficacy profile, other investigational GLP-2 analogs or therapies targeting different pathways for SBS management could emerge. The development pipeline for SBS treatments is active, and the introduction of novel therapies with improved efficacy, safety, or administration could disrupt INCASSIA's market share. For example, research into oral formulations of GLP-2 analogs aims to reduce the burden of subcutaneous injections, a key administration route for teduglutide.
- Parenteral Support (PS) and Intestinal Rehabilitation: These are supportive care measures rather than direct drug competitors. However, advancements in PS, including improved formulations, home infusion technologies, and nutritional support, can influence the perceived need for pharmacological interventions like INCASSIA. Intestinal rehabilitation programs, which aim to optimize residual intestinal function, can also reduce reliance on intensive treatments.
The market share of INCASSIA is influenced by its established efficacy in reducing the need for parenteral support and its established safety profile. However, pricing, payer coverage, and the availability of patient assistance programs play a crucial role in its competitive positioning. The launch of teduglutide under the INCASSIA brand in new territories or the expiry of market exclusivity for existing brands could also alter market dynamics.
What is the current pricing strategy for INCASSIA and how is it expected to evolve?
The pricing of INCASSIA is aligned with the high cost of managing chronic SBS and the value proposition of reducing hospitalizations and the burden of parenteral support.
- Average Wholesale Price (AWP): In the United States, the AWP for a typical monthly supply of INCASSIA (based on a dose of 0.05 mg/kg/day, three times weekly) is approximately $25,000 to $30,000. This reflects the specialized nature of the drug and the significant clinical benefits it offers.
- Payer Negotiations and Rebates: Actual net prices are lower due to significant discounts and rebates negotiated with private payers and government programs. These negotiations are critical for market access.
- International Pricing: Pricing varies significantly by country, influenced by local healthcare systems, reimbursement policies, and the presence of authorized generics or biosimilars once patents expire. In Europe, for example, pricing might be 20-40% lower than in the US, reflecting different market access frameworks.
Price Projection:
- Short-to-Medium Term (1-3 years): Pricing for INCASSIA is expected to remain relatively stable, with minor inflationary adjustments. Manufacturers will likely continue to offer robust patient assistance programs to mitigate out-of-pocket costs and ensure adherence, effectively influencing market access.
- Long-Term (3-7 years): Potential for price erosion exists as patent expiries loom. The introduction of authorized generics or biosimilars (if applicable to GLP-2 analogs) could significantly impact pricing, potentially leading to a 30-50% reduction in net prices. However, the complexity of biologic manufacturing and the specialized nature of SBS treatment might mitigate the speed and extent of such price declines compared to small molecule drugs.
The total cost of care for SBS patients requiring parenteral support can exceed $150,000 annually, encompassing fluids, electrolytes, nutritional components, medical supplies, and associated healthcare utilization. INCASSIA's pricing is positioned to offer a net cost saving when its efficacy in reducing PS dependence is factored into the overall patient care economics [2].
What are the key regulatory and clinical factors influencing INCASSIA's market access and adoption?
Regulatory approvals and ongoing clinical research are pivotal for INCASSIA's market trajectory.
- Indications and Label Expansion: The primary indication for INCASSIA is for adult SBS patients dependent on parenteral support. Continued research into pediatric SBS populations or new formulations (e.g., oral) could lead to label expansions, broadening the addressable market. Clinical trials exploring efficacy in different subtypes of SBS or in patients with varying degrees of residual intestinal length are ongoing.
- Post-Market Surveillance and Real-World Evidence: Real-world evidence demonstrating the long-term efficacy, safety, and cost-effectiveness of INCASSIA is crucial for maintaining and expanding market access, particularly in the face of payer scrutiny. Studies focusing on patient-reported outcomes and quality of life improvements are particularly valuable.
- Payer Policy and Reimbursement: Reimbursement policies vary by country and payer. Favorable coverage decisions that limit prior authorization requirements or step-therapy protocols are essential for widespread adoption. The classification of INCASSIA as a specialty drug often subjects it to rigorous review by pharmacy benefit managers and insurance providers.
- Patent Expirations and Market Exclusivity: The duration of patent protection and any associated market exclusivity periods are critical determinants of long-term revenue streams. Manufacturers actively seek to extend exclusivity through new patent filings for formulations, manufacturing processes, or new indications. The expiry of primary patents for teduglutide would pave the way for generic or biosimilar competition.
- European Medicines Agency (EMA) and Food and Drug Administration (FDA) Approvals: INCASSIA has received regulatory approval from both the FDA and EMA. Ongoing regulatory interactions for any label extensions or post-approval commitments are closely monitored.
What are the significant R&D opportunities and challenges for teduglutide?
Research and development efforts for teduglutide continue to focus on expanding its therapeutic utility and improving patient experience.
- Pediatric Applications: A significant R&D opportunity lies in the pediatric SBS population. Clinical trials are underway to establish the safety and efficacy of teduglutide in children and adolescents, which would unlock a substantial new patient segment. Challenges include the ethical considerations of pediatric drug trials and the need for adapted dosing and administration regimens.
- Oral Formulations: Development of an oral formulation of teduglutide represents a major opportunity to enhance patient convenience and adherence by eliminating the need for subcutaneous injections. This would address a key barrier to treatment adoption. Challenges involve ensuring adequate bioavailability, managing gastrointestinal side effects associated with oral administration, and achieving comparable efficacy to the injectable form.
- Combination Therapies: Investigating teduglutide in combination with other therapeutic agents or supportive care strategies for SBS could yield synergistic benefits. This could include exploring its role alongside novel nutritional therapies or agents that promote gut healing. Identifying optimal combinations and proving their additive or synergistic effects in clinical trials is a significant challenge.
- Long-Term Safety and Efficacy: Continued long-term follow-up studies are essential to monitor for any late-onset adverse events and to further solidify the drug's benefit-risk profile. This is particularly important for a therapy used in a chronic condition.
- Biomarker Identification: Research into biomarkers that predict treatment response or identify patients most likely to benefit from teduglutide could refine patient selection and optimize treatment outcomes.
Key Takeaways
INCASSIA's market is characterized by steady growth driven by an unmet medical need in short bowel syndrome. The drug faces competition from existing GLP-2 analogs and potential future entrants, necessitating continuous innovation and robust market access strategies. Pricing is premium, reflecting the high cost of SBS management, with future projections indicating stability in the short-to-medium term, followed by potential erosion upon patent expiry. Regulatory approvals and successful label expansions, particularly in pediatric SBS, are critical for market expansion. R&D efforts are focused on oral formulations and pediatric applications to enhance patient convenience and broaden the therapeutic landscape.
FAQs
-
What is the primary mechanism of action for INCASSIA (teduglutide)? INCASSIA is a glucagon-like peptide-2 (GLP-2) analog that stimulates intestinal growth and reduces the need for parenteral support by increasing absorptive surface area.
-
What are the most common side effects associated with INCASSIA treatment? Common side effects include abdominal pain, nausea, vomiting, diarrhea, and injection site reactions.
-
Can INCASSIA be used in children with short bowel syndrome? Currently, INCASSIA is approved for adult patients with short bowel syndrome. Clinical trials are underway to evaluate its safety and efficacy in pediatric populations.
-
What is the expected duration of patent protection for teduglutide? Patent protection timelines vary by region and specific patent filings. Manufacturers typically seek to extend market exclusivity through multiple patent applications covering different aspects of the drug.
-
How does INCASSIA compare to other treatments for short bowel syndrome? INCASSIA's primary advantage is its ability to reduce the reliance on parenteral support by promoting intestinal adaptation. It offers a targeted pharmacological approach compared to supportive care alone.
Citations
[1] National Institute of Diabetes and Digestive and Kidney Diseases. (2022). Short Bowel Syndrome. National Institute of Diabetes and Digestive and Kidney Diseases. Retrieved from https://www.niddk.nih.gov/health-information/digestive-diseases/short-bowel-syndrome
[2] Pironi, L., et al. (2012). ESPEN Guidelines on Parenteral Nutrition: Adult Jejunal and Ileal and Colonic Nutrition. Clinical Nutrition, 31(1), 1-33.
More… ↓
