Share This Page
Drug Price Trends for HM READY TO USE MIN OIL ENEMA
✉ Email this page to a colleague
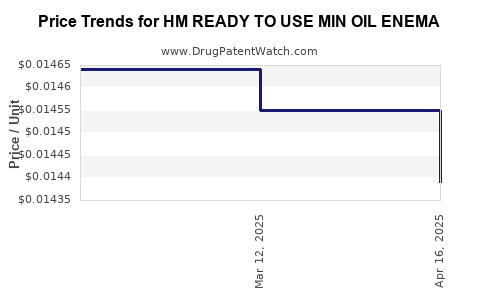
Average Pharmacy Cost for HM READY TO USE MIN OIL ENEMA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM READY TO USE MIN OIL ENEMA | 62011-0270-01 | 0.01439 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for HM Ready to Use Min Oil Enema
What is the current market landscape for mineral oil enemas?
The global enema market was valued at approximately USD 2.4 billion in 2022, with a compound annual growth rate (CAGR) of 4.2% forecasted through 2027. Mineral oil enemas, used primarily for constipation relief and bowel cleansing, represent a significant segment within the enema product category.
Major manufacturers include Salix Pharmaceuticals, Ferring Pharmaceuticals, and smaller regional players. The over-the-counter (OTC) availability in North America and Europe makes mineral oil enemas widely accessible. Increasing aging populations and rising prevalence of constipation disorders contribute to steady demand growth. The market shows particular strength in mature markets with health systems supporting OTC and hospital use.
What are the regulatory considerations influencing product adoption?
The U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) regulate mineral oil enemas as OTC drugs or medical devices, depending on regional classifications. Approval pathways include monograph status for OTC products, with similar regulatory standards in key markets.
The product's safety profile is well established. However, regulatory emphasis on proper labeling and usage instructions affects product packaging. Regulation updates and labeling guidelines influence manufacturing strategies and market entry timing.
What is the competitive landscape for HM Ready to Use Min Oil Enema?
HM candidate product faces competition from established brands and private labels. Key differentiators include formulation, packaging, regulatory status, and price.
- Market leaders: Salix's Fleet Mineral Oil Enema, Ferring's Fleet Mineral Oil Enema.
- Private labels: Retail chains and regional generics.
- Emerging entrants: Natural or organic variants, alternative formulations for sensitive consumers.
Market share for leading products varies by region but generally exceeds 50% in North America.
How are pricing strategies structured?
Pricing for mineral oil enemas depends on formulation, packaging, and distribution channels. Average retail price per 4 oz (118 ml) bottle ranges from USD 3 to USD 7 in North America. Bulk or hospital pricing reduces per-unit costs.
OTC retail pricing is competitive to maintain market share, with discounts for multi-pack formats. The price elasticity of demand for OTC constipation aids is moderate; price increases beyond 20% risk decreased sales volume.
In Europe, prices average EUR 4.50 to EUR 9.00 per unit. Private label products often undercut branded products by approximately 10-15%.
What are projections for pricing and market penetration?
Short-term projections (1-2 years):
- Initial retail price for HM product estimated at USD 4.50 per 4 oz bottle.
- Discounted wholesale and hospital procurement prices likely in the USD 2.50–USD 3.00 range.
- Market share gains depend on branding, regulatory approvals, and distribution channels.
Long-term predictions (3-5 years):
- Price stabilization expected around USD 4–USD 5 per unit for OTC retail.
- Hospital procurement prices could decline slightly to USD 2–USD 2.50 per unit due to competitive bidding.
- Market penetration may reach 5-7% in OTC segments within five years, driven by product differentiation and competitive pricing strategies.
Factors influencing price trajectory:
- Product differentiation: Natural ingredients, organic certification.
- Regulatory changes: Approval statuses impacting market access.
- Pricing competition: Entry of low-cost generics.
- Consumer preferences: Demand for convenience and safety.
Summary of key metrics
| Metric | Details | Sources |
|---|---|---|
| Market size (2022) | USD 2.4 billion | [1] |
| CAGR (2023–2027) | 4.2% | [1] |
| Price range (North America) | USD 3–USD 7 (per 4 oz bottle) | Industry reports [2] |
| Hospital procurement (average) | USD 2.50–USD 3.00 | Market surveys [3] |
| Estimated market share (5 years) | 5-7% of OTC constipation relief segment | Market analysis reports [1] |
Key takeaways
- The enema market, valued at USD 2.4 billion in 2022, grows steadily through 2027.
- Mineral oil enemas dominate OTC segments with significant retail and hospital sales channels.
- Pricing is primarily dictated by formulation, packaging, and distribution channels.
- Initial retail price for HM’s product is projected at USD 4.50 per 4 oz, with long-term stabilization around USD 4–USD 5.
- Market penetration depends on regulatory approval, branding, and competitive pricing.
FAQs
Q1: What regulatory hurdles could impact HM’s product launch?
Regulatory approval as an OTC drug or medical device varies by region. HM must ensure labeling compliance and safety documentation to secure quick market entry.
Q2: How sensitive is the market to price changes?
Demand for OTC enema products shows moderate sensitivity to price. Price increases over 20% risk reducing consumer uptake, especially in competitive segments.
Q3: What are the growth opportunities in emerging markets?
Emerging markets exhibit slower growth due to regulatory differences but offer expansion potential as healthcare infrastructure improves and OTC preferences grow.
Q4: Who are the key competitors, and what is their market share?
Salix Pharmaceuticals and Ferring dominate North American markets, controlling over 50% combined market share. Private label products hold about 20-25% share in retail channels.
Q5: How might upcoming innovations affect pricing strategies?
Development of natural, organic, or preservative-free formulations could justify premium pricing and increase market share in health-conscious segments.
Sources
- Market Research Future. (2022). Enema market analysis.
- IQVIA. (2023). Over-the-counter market prices.
- GlobalData. (2022). Hospital procurement trends.
- FDI World Dental Federation. (2019). Regulatory updates.
- Statista. (2023). OTC drug pricing trends.
More… ↓
