Share This Page
Drug Price Trends for HM ESOMEPRAZOLE MAG DR
✉ Email this page to a colleague
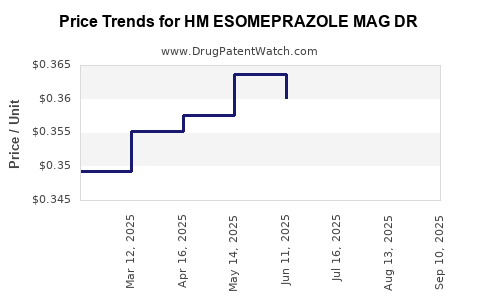
Average Pharmacy Cost for HM ESOMEPRAZOLE MAG DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM ESOMEPRAZOLE MAG DR 20 MG | 62011-0332-02 | 0.35740 | EACH | 2025-09-17 |
| HM ESOMEPRAZOLE MAG DR 20 MG | 62011-0332-02 | 0.35358 | EACH | 2025-08-20 |
| HM ESOMEPRAZOLE MAG DR 20 MG | 62011-0332-02 | 0.35611 | EACH | 2025-07-23 |
| HM ESOMEPRAZOLE MAG DR 20 MG | 62011-0332-02 | 0.36013 | EACH | 2025-06-18 |
| HM ESOMEPRAZOLE MAG DR 20 MG | 62011-0332-02 | 0.36369 | EACH | 2025-05-21 |
| HM ESOMEPRAZOLE MAG DR 20 MG | 62011-0332-02 | 0.35753 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HM ESOMEPRAZOLE MAG DR Market Analysis and Financial Projection
Market Analysis and Price Projections for HM Esomeprazole Mag DR
Overview of the Drug
HM Esomeprazole Magnesium DR (Delayed Release) is a proton pump inhibitor (PPI) used primarily to treat gastroesophageal reflux disease (GERD), Zollinger-Ellison syndrome, and prevent gastric ulcers caused by NSAIDs. Its formulation as a delayed-release capsule aims to enhance bioavailability and prolong its efficacy.
Current Market Landscape
Existing Competition
The PPI market is dominated by several key brands, including:
- NEXIUM (Esomeprazole) — AstraZeneca
- VASTAREX (Esomeprazole) — Teva Pharmaceuticals
- NEXIUM IV — AstraZeneca
- Noltec — Cipla
These products, particularly NEXIUM, hold substantial market share due to Brand loyalty and extensive marketing. Generic versions of esomeprazole have gained prominence since patent expirations, increasing price competition.
Patent Status and Market Entry
As of 2023, AstraZeneca's patent for esomeprazole has expired in many markets. However, some proprietary formulations, like delayed-release mechanisms, retain patent protection, delaying generic entry.
In markets where patents have expired, generic formulations proliferate at significantly lower prices, compressing the pricing landscape for new entrants like HM Esomeprazole Mag DR.
Market Size and Growth Trends
Global Market Value
The global PPI market was valued at approximately USD 12 billion in 2022.[1] It is projected to grow at a compound annual growth rate (CAGR) of around 4-6% from 2023 to 2028, reaching an estimated USD 16 billion by 2028.[2]
Regional Dynamics
- North America: Largest market, driven by high prevalence of GERD and robust healthcare expenditure.
- Europe: Mature market with steady growth.
- Asia-Pacific: Fastest growth due to rising urbanization, increasing GERD diagnoses, and expanding healthcare infrastructure.
Key Market Drivers
- Aging population increasing the demand.
- Rising incidences of GERD, peptic ulcers.
- Growing awareness and diagnosis.
Price Dynamics and Factors Influencing Pricing
Current Pricing Benchmarks
In the United States, brand-name esomeprazole (NEXIUM) costs about USD 150-200 for a 30-day supply, depending on dosage and pharmacy networks. Generics are priced substantially lower, around USD 30-50 for a comparable supply.
In Europe, prices range from EUR 20-40 for generics, with branded drugs priced higher.
Price Differentiation
Newer formulations with delayed-release mechanisms tend to command premium pricing due to perceived benefits or proprietary delivery mechanisms, though competitive pressure from generics constrains pricing.
Price Projections for HM Esomeprazole Mag DR
Market Entry Strategies
-
Pricing at Launch: Expected to position at a slight discount to branded NEXIUM but higher than generics, approximately USD 80-120 per 30-day supply, to reflect formulation innovation and quality.
-
Post-Launch Evolution: As patent protections, exclusivity, and market acceptance develop, prices may stabilize between USD 70-100 per 30-day supply.
Sales Volume and Penetration
Assuming a conservative penetration rate within the first three years — approximately 1-3% of the total PPI market in developed regions — sales could reach USD 50-150 million annually. In emerging markets, lower price points could facilitate higher volume, increasing revenue potential.
Long-term Outlook
In markets with patent or regulatory exclusivity, prices could eventually decline to USD 50-70 as generics become more prevalent. Alternatively, specialty indications or formulation advantages might sustain higher price points.
Risks and Opportunities
Risks:
- Patent overturns or legal challenges.
- Rapid entry of generics post-patent expiry.
- Cost-effectiveness compared with existing treatments.
Opportunities:
- Emphasizing delayed-release formulation advantages.
- Targeting niche indications like Zollinger-Ellison syndrome.
- Leveraging regional regulatory exclusivity.
Conclusions
In a mature market, HM Esomeprazole Mag DR's initial pricing will likely be positioned slightly below established brands but above generics, anticipated around USD 80-120 per month. Market expansion depends on formulation advantages, regulatory exclusivity, and competitive responses.
Key Takeaways
- The global PPI market is projected to grow at 4-6% CAGR, reaching USD 16 billion by 2028.
- Pricing for new esomeprazole formulations remains constrained by generic competition post-patent expiration.
- HM Esomeprazole Mag DR's initial market price is expected between USD 80-120 per month, with long-term consolidation likely leading to lower prices.
- Market share will depend on regional patent protections, efficacy perceptions, and formulary positioning.
- Entry strategies should focus on differentiation and regional exclusivity.
FAQs
1. How does delayed-release esomeprazole differ from existing formulations?
It prolongs drug release, potentially improving efficacy, reducing dosing frequency, and minimizing side effects associated with peak plasma levels.
2. What is the typical market share of branded versus generic PPIs?
Branded PPIs like NEXIUM dominate in early patent-protected periods, comprising about 50-70% of sales pre-generic entry. Generics then capture 80% or more of the market.
3. Which regions offer the highest sales potential for new PPI formulations?
North America and Europe are mature markets with high per capita consumption. Asia-Pacific presents rapid growth due to increasing GERD prevalence.
4. How will patent status influence HM Esomeprazole Mag DR's pricing?
Patent or regulatory exclusivity could sustain higher prices for 3-5 years, after which price pressures from generics will likely reduce prices significantly.
5. What regulatory hurdles could impact market entry?
Approvals depend on demonstrating bioequivalence and formulation safety. Patent challenges or restrictions on delayed-release mechanisms could delay or limit launch.
References
[1] Market Research Future, "Proton Pump Inhibitors Market," 2022.
[2] Grand View Research, "Proton Pump Inhibitors Market Size, Share & Trends," 2023.
More… ↓
