Share This Page
Drug Price Trends for HEMORRHOIDAL SUPPOSITORY
✉ Email this page to a colleague
Average Pharmacy Cost for HEMORRHOIDAL SUPPOSITORY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
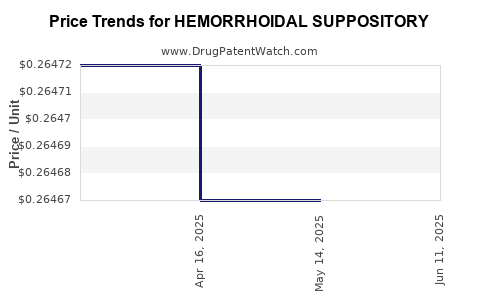
| HEMORRHOIDAL SUPPOSITORY | 00904-6977-12 | 0.26467 | EACH | 2025-06-18 |
| HEMORRHOIDAL SUPPOSITORY | 00904-6977-12 | 0.26467 | EACH | 2025-05-21 |
| HEMORRHOIDAL SUPPOSITORY | 00904-6977-12 | 0.26467 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Hemorrhoidal Suppositories
Market Overview
Hemorrhoidal suppositories are a common treatment for hemorrhoids, used to reduce pain, inflammation, and bleeding. The global market for hemorrhoidal treatments, including suppositories, was valued at approximately $2.3 billion in 2021 and is projected to grow at a compound annual growth rate (CAGR) of 3.8% through 2027[1].
The key drivers include increasing prevalence of hemorrhoidal disease, over-the-counter availability, and a preference for minimally invasive therapies. Regionally, North America leads with around 40% of the market, driven by high healthcare awareness and extensive OTC sales networks. Europe accounts for about 30%, with growing demand in Asia-Pacific, which is expected to see significant growth due to expanding healthcare infrastructure.
Key Players and Product Landscape
Leading companies include Johnson & Johnson, Bayer, GlaxoSmithKline, and local generic manufacturers. Most products feature active ingredients like hydrocortisone, phenylephrine, or witch hazel, either alone or in combination, to address symptoms.
New formulations focus on improved delivery, longer-lasting effects, and reduced side effects — emerging as competitive differentiators. Over-the-counter status in many regions facilitates rapid market penetration.
Regulatory Status
In the U.S., OTC hemorrhoidal suppositories are FDA-approved as Class I devices or drugs, depending on ingredients. Many jurisdictions classify these products as OTC drugs, simplifying distribution but requiring adherence to local regulatory standards before market entry.
Market Segmentation
Segmentation by formulation type:
- Hydrocortisone-based suppositories: Approx. 55% market share, due to anti-inflammatory efficacy.
- Combination products: Comprise around 35%, combining analgesics or vasoconstrictors.
- Others (witch hazel, phenylephrine): 10%.
By distribution channel:
- OTC pharmacy sales account for 80%.
- Online sales are rising, representing about 10% currently but expected to reach 20% by 2025.
Price Projections
Current average retail price in developed markets (e.g., U.S., Europe):
- Single unit: $5–$8.
- Pack of 12: $40–$70.
For generic versions, prices can be approximately 20–30% lower, depending on manufacturing costs and branding.
Forecasted Pricing Trends (2023-2027):
| Year | Average Single Unit Price | Pack Price Range | Factors Influencing Prices |
|---|---|---|---|
| 2023 | $6.50 | $45–$65 | Marginal price stabilization; generic competition intensifies |
| 2024 | $6.20 | $42–$62 | Cost reductions from manufacturing efficiencies |
| 2025 | $6.00 | $40–$60 | Entry of low-cost generics; patent expirations in certain markets |
| 2026 | $5.80 | $38–$58 | Increased online sales and direct-to-consumer channels |
| 2027 | $5.50 | $36–$55 | Market saturation; pricing pressure persists |
Pricing Strategies
Manufacturers tend to leverage value-based pricing for branded products in high-income regions, while generic and private-label suppositories compete mainly on price. New entrants focus on cost-efficient manufacturing, aiming to undercut existing prices, especially in emerging markets.
Market Entry Considerations
- Achieving regulatory approval requires regional compliance (e.g., FDA, EMA, PMDA).
- Establishing distribution channels through pharmacies and online platforms accelerates market penetration.
- Formulation differentiation, such as sustained-release components, can justify premium pricing.
Consolidation and Future Trends
Market consolidation is anticipated, with larger pharmaceutical companies acquiring smaller firms to expand portfolios. Growing demand for combination products and herbal or natural formulations may influence pricing structures and product offerings.
Key Takeaways
- The global hemorrhoidal suppository market is valued at $2.3 billion and expected to grow steadily.
- Pricing remains competitive, with average retail prices declining slightly due to generic competition.
- Online sales channels will exert downward pressure on prices but also expand market access.
- Innovation focuses on delivery mechanisms and combination therapies, which can sustain premium pricing.
- Regulatory landscapes vary across regions, influencing market entry and pricing strategies.
FAQs
-
What are the primary active ingredients in hemorrhoidal suppositories?
Hydrocortisone, phenylephrine, witch hazel, lidocaine. -
How does the OTC status affect pricing and market entry?
OTC products enjoy easier market entry and wider distribution but face price competition from generics. -
What is the typical price range for a pack of hemorrhoidal suppositories?
$40–$70 in developed markets. -
How do patents and exclusivity impact pricing?
Patent protections sustain higher prices; expiration typically leads to generic entry and price reductions. -
What regional differences affect market growth?
High prevalence and healthcare infrastructure in North America and Europe favor growth; Asia-Pacific is emerging as a significant market segment.
References
[1] MarketWatch. “Hemorrhoidal Treatment Market – Growth, Trends, Forecasts (2022-2027).”
More… ↓
