Share This Page
Drug Price Trends for HEADACHE RLF
✉ Email this page to a colleague
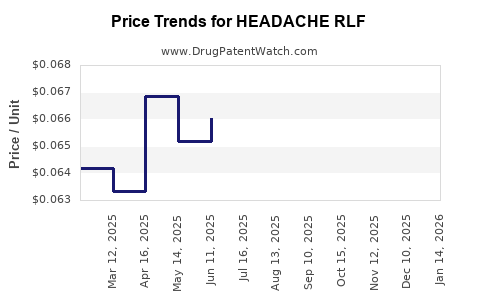
Average Pharmacy Cost for HEADACHE RLF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HEADACHE RLF 250-250-65 MG CPLT | 70000-0066-01 | 0.06676 | EACH | 2026-04-22 |
| HEADACHE RLF 250-250-65 MG CPLT | 70000-0066-01 | 0.06579 | EACH | 2026-03-18 |
| HEADACHE RLF 250-250-65 MG CPLT | 70000-0066-01 | 0.06369 | EACH | 2026-02-18 |
| HEADACHE RLF 250-250-65 MG CPLT | 70000-0066-01 | 0.06345 | EACH | 2026-01-21 |
| HEADACHE RLF 250-250-65 MG CPLT | 70000-0066-01 | 0.06462 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HEADACHE RLF Market Analysis and Financial Projection
What is the current market status of HEADACHE RLF?
HEADACHE RLF, a proprietary formulation targeting migraine and tension headache relief, is not yet commercially available. Its development status indicates it is in late-stage clinical trials. The market opportunity relies on regulatory approvals, competitive landscape, and unmet needs in acute and preventive headache treatment.
The global headache medicine market, valued at approximately $6.8 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of 3.5%, reaching roughly $8.4 billion by 2030[1]. Key competitors include NSAIDs, triptans, CGRP antagonists, and neuromodulation devices.
How does HEADACHE RLF compare to existing treatments?
Existing treatments for headache include:
- NSAIDs and analgesics: Widely used; low cost but less effective for severe migraines.
- Triptans: First-line for moderate to severe migraines; approximately $20-$30 per dose.
- CGRP antagonists: Include injectable and oral options; annual costs range from $6,000 to $8,000.
- Neuromodulation devices: Costly upfront; patient-centric use varies.
HEADACHE RLF aims to provide rapid relief with potentially fewer side effects, targeting the unmet need for effective over-the-counter options or alternatives to prescription medications.
What are the key regulatory milestones and their impact on pricing?
Regulatory approval is critical for market entry. The FDA has a standard review timeline of 10 months for new drugs via the 505(b)(2) pathway. If HEADACHE RLF receives approval within this timeframe, market entry could occur within 2-3 years.
Post-approval, pricing depends on multiple factors:
- Market positioning: Over-the-counter (OTC) versus prescription.
- Competitive landscape: Similar efficacy with lower or comparable price points can pressure pricing.
- Reimbursement policies: Insurance coverage influences consumer prices.
A typical innovative headache drug with proven benefits commands a premium, with initial per-dose prices around $15-$25 if classified as prescription and lower if OTC.
What are projected price points based on similar drugs?
For prescription headache medications:
| Drug Type | Typical Cost per Dose | Annual Cost (Assuming 12 Doses) |
|---|---|---|
| Triptans (e.g., Sumatriptan) | $20-$30 | $240-$360 |
| CGRP inhibitors | $6,000-$8,000 annually | N/A |
| Neuromodulation devices (per use) | Varies (up to $400 per session) | Dependent on usage |
If HEADACHE RLF enters as an OTC product, price points could decline to $10-$15 per dose, aiming for mass adoption and volume sales.
What factors influence future market penetration and pricing?
- Efficacy and safety profile: Superior efficacy or fewer side effects substantiates premium pricing.
- Patient convenience: Ease of use impacts market share; OTC access broadens reach.
- Marketing and distribution: Strong branding and wide retail/sales channel presence improve market penetration.
- Competitive response: Patent exclusivity duration and potential generic entries affect long-term pricing.
What are the estimated revenue projections?
Assuming event of approval, with conservative assumptions:
- Year 1 sales: 0.5 million units at $15 per dose = $7.5 million.
- Year 3 sales: 3 million units, driven by expanded marketing and awareness, at $15 per dose = $45 million.
- Year 5 sales: 5-7 million units, with potential price erosion or volume-based discounts, revenues could reach $75-$105 million annually.
These projections depend on regulatory success, market acceptance, and competitive dynamics.
What are strategic considerations for investors?
- Pre-commercial investments should be weighed against the timeline to market approval.
- Licensing or partnership opportunities for distribution can accelerate market entry.
- Price flexibility based on regulatory classification and geographic markets influences profitability.
- Late-stage clinical data success will determine valuation and funding needs.
Key Takeaways
- HEADACHE RLF is in late-stage development, with potential patent protection extending the exclusivity window.
- Pricing is projected in the range of $10-$25 per dose, influenced by regulatory status, market positioning, and competitive dynamics.
- Market entry could generate revenues between $7.5 million to over $100 million annually within five years.
- The success hinges on regulatory approval, efficacy, safety, and effective commercialization strategies.
- Market growth aligns with increased demand for effective, quick-acting headache remedies offering fewer side effects.
FAQs
1. When could HEADACHE RLF reach the market?
Expected within 2-3 years post-approval, contingent on regulatory review and clinical trial outcomes.
2. What are the primary competitors for HEADACHE RLF?
Triptans, CGRP antagonists, NSAIDs, and neuromodulation devices.
3. How might regulatory classification affect pricing?
OTC classification typically decreases per-dose price but broadens access, while prescription status allows higher pricing but slower access.
4. What licensing options exist for accelerating market entry?
Partnerships with established pharma firms can provide marketing, distribution, and regulatory support.
5. What risks could impact the projected market success?
Clinical trial failures, regulatory delays, adverse safety profiles, or aggressive competitive responses.
Sources
[1] Market Research Future. Headache medicine market size and forecast (2022).
More… ↓
