Share This Page
Drug Price Trends for GUAIFENESIN ER
✉ Email this page to a colleague
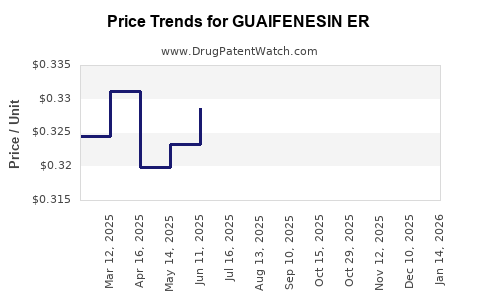
Average Pharmacy Cost for GUAIFENESIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GUAIFENESIN ER 600 MG TABLET | 51660-0566-21 | 0.29751 | EACH | 2026-05-20 |
| GUAIFENESIN ER 600 MG TABLET | 68094-0048-59 | 0.29751 | EACH | 2026-05-20 |
| GUAIFENESIN ER 600 MG TABLET | 00536-1163-61 | 0.29751 | EACH | 2026-05-20 |
| GUAIFENESIN ER 600 MG TABLET | 70010-0199-05 | 0.29751 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is Guaifenesin ER?
Guaifenesin extended-release (ER) is an expectorant used to relieve chest congestion caused by colds, infections, or allergies. It is available in various formulations for once-daily dosing, improving patient compliance compared to immediate-release formulations. Market use primarily involves OTC products and prescription medications, with growing interest driven by chronic cough management and respiratory conditions.
What is the Size of the Current Market?
The global expectorant market, which includes Guaifenesin ER, was valued at approximately $2.4 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of roughly 4.2% from 2023 to 2030. This growth factors in increased respiratory illnesses, aging populations, and demand for OTC medications.
Key Market Drivers:
- Rising prevalence of respiratory conditions.
- Increased consumer preference for OTC denoting better self-care.
- Advances in drug delivery systems enhancing efficacy.
What are the Main Players and Their Market Shares?
Leading manufacturers include:
| Company | Market Share (Estimated, 2022) | Key Products |
|---|---|---|
| Johnson & Johnson | 30% | Mucinex, Act, and other OTC formulations |
| Reckitt Benckiser | 20% | Benylin expectorants |
| Teva Pharmaceuticals | 15% | Guaifenesin ER formulations |
| Perrigo | 10% | Store-brand Guaifenesin ER |
| Others | 25% | Various generics and regional brands |
Market consolidation trends suggest major brands focus on extending formulations' duration and controlled-release technologies to differentiate offerings.
What Are the Regulatory and Pricing Trends?
Regulatory landscape:
Guaifenesin ER products require FDA approval in the U.S., with some formulations classified as OTC drugs. Newer formulations may undergo additional clinical testing to support claims and ensure safety. In many countries, Guaifenesin remains OTC, with prescribing restrictions limited to specific formulations.
Pricing trends:
Average retail prices for Guaifenesin ER range from $10 to $15 per 30-count bottle. Brand-name OTC drugs typically cost 30-50% more than generic versions. Price competition is intense, especially in the OTC segment, pressuring margins.
| Product Type | Estimated Retail Price (per 30-count) | Manufacturer/Brand |
|---|---|---|
| Brand-name OTC Guaifenesin ER | $12 - $15 | Mucinex, Benylin |
| Generic Guaifenesin ER (store brands) | $8 - $12 | Perrigo, others |
What Are the Future Price Projections?
Short-term (2023-2025):
Prices are expected to stabilize, with slight reductions in generic segments driven by increasing competition. Brand-name products may hold premiums due to brand recognition and formulation differentiation.
Medium-term (2026-2030):
Market entry of reformulated, extended-release products with improved bioavailability or combination therapies could command higher prices initially. If these innovations prove clinically advantageous, premium pricing may sustain through patent protections or exclusivity rights.
Revenue forecasts (2023-2030):
Projected to grow from $2.4 billion to approximately $3.0 billion globally, assuming steady market share retention and incremental price increases averaging 2-3% annually.
What Are Key Trends Influencing the Market?
- Innovation in Formulation: Extended-release technologies that improve compliance and efficacy are gaining interest. These include matrix or coating-based release systems.
- Combination Products: Guaifenesin combined with decongestants or cough suppressants are expanding market applications.
- Digital Health Integration: Apps and telemedicine platforms increase accessibility, influencing prescribing and sales patterns.
- Patent and Exclusivity: Patent protections for specific formulations or delivery mechanisms could temporarily limit generic competition but are expiring gradually.
What are the Opportunities and Challenges?
Opportunities:
- Expansion into emerging markets with rising respiratory disease burdens.
- Development of novel extended-release mechanisms with better pharmacokinetics.
- Partnerships with telehealth providers to boost sales channels in OTC and prescription sectors.
Challenges:
- High OTC price sensitivity limits pricing power.
- Competition from other expectorants and cough medicines.
- Regulatory hurdles for new formulations, particularly in international markets.
What is the Outlook for Investment and R&D?
Investments in reformulations, combination drugs, and innovative delivery technology are expected to drive future valuation. R&D efforts focus on improving pharmacokinetic profiles, reducing dosing frequency, and integrating digital health monitoring.
Key Takeaways
- The Guaifenesin ER market is poised for steady growth, driven by aging populations and increased respiratory illnesses.
- Major players dominate with OTC branding and ongoing innovations in formulation technology.
- Price projections indicate modest increases, constrained by market competition and price-sensitive consumers.
- Regulatory strategies, patent protections, and technological advancements will shape future market dynamics.
- Opportunities exist in emerging markets, formulation innovations, and digital integration; challenges include pricing pressure and regulatory barriers.
FAQs
1. What distinguishes Guaifenesin ER from immediate-release formulations?
Extended-release formulations allow once-daily dosing, improve compliance, and maintain steadier blood levels, potentially enhancing efficacy and convenience.
2. Will FDA approval be required for new Guaifenesin ER formulations?
Yes, any new formulation typically requires FDA review, especially if claims differ from existing products or if new delivery mechanisms are used.
3. How does market competition impact prices?
Intense competition among generics and OTC brands restricts price increases, maintaining relatively low retail prices despite technological advancements.
4. Are there significant regional differences in Guaifenesin ER pricing?
Yes, prices vary due to regulatory environments, market maturity, and healthcare system differences, with U.S. prices generally higher than in emerging markets.
5. What emerging technologies could influence the Guaifenesin ER market?
Innovations include novel sustained-release systems, combination therapies, and digital health tools for monitoring respiratory health.
References
[1] MarketWatch. (2022). "Expectorants Market Size."
[2] Grand View Research. (2023). "Expectorants Market Trends."
[3] U.S. Food and Drug Administration. (2022). "OTC Monographs and Drug Approvals."
More… ↓
