Drug Price Trends for GS SLEEP TIME
✉ Email this page to a colleague
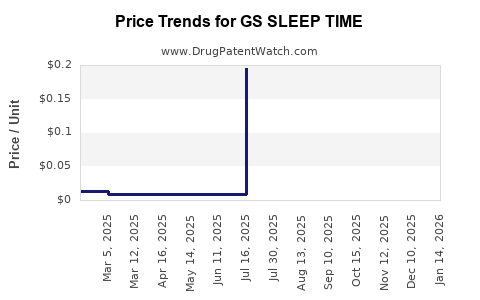
Average Pharmacy Cost for GS SLEEP TIME
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS SLEEP TIME 50 MG/30 ML LIQ | 00113-0186-40 | 0.00836 | ML | 2024-11-20 |
| GS SLEEP TIME 25 MG SOFTGEL | 00113-0052-67 | 0.17600 | EACH | 2024-11-20 |
| GS SLEEP TIME 50 MG/30 ML LIQ | 00113-0186-30 | 0.01363 | ML | 2024-11-20 |
| GS SLEEP TIME 25 MG SOFTGEL | 00113-0052-53 | 0.17600 | EACH | 2024-11-20 |
| GS SLEEP TIME 25 MG SOFTGEL | 00113-0052-67 | 0.19708 | EACH | 2024-10-23 |
| GS SLEEP TIME 50 MG/30 ML LIQ | 00113-0186-40 | 0.00767 | ML | 2024-10-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |


