Share This Page
Drug Price Trends for GRALISE ER
✉ Email this page to a colleague
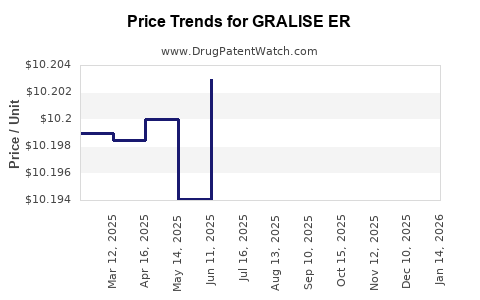
Average Pharmacy Cost for GRALISE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GRALISE ER 900 MG TABLET | 52427-0890-60 | 15.30527 | EACH | 2026-03-18 |
| GRALISE ER 600 MG TABLET | 52427-0806-90 | 10.16196 | EACH | 2026-03-18 |
| GRALISE ER 300 MG TABLET | 52427-0803-90 | 10.19808 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GRALISE ER
What is the Current Market Position of GRALISE ER?
GRALISE ER (gabapentin extended-release) is approved by the FDA for postherpetic neuralgia (PHN). It is positioned as a treatment option for nerve pain, often competing with generic gabapentin and other nerve pain medications such as pregabalin. Despite background competition, its extended-release formulation aims to provide improved compliance and symptom management.
Who Are the Key Competitors and Market Landscape?
The nerve pain treatment sector is highly competitive. Major competitors include:
- Generic gabapentin: Market share dominates due to low cost, with prices as low as $0.10 per capsule.
- Lyrica (pregabalin): A branded alternative with annual U.S. sales exceeding $4 billion (2019 data). Priced at approximately $10–$15 per capsule.
- Neurontin (gabapentin): Previously branded, now mainly generic, with similar low prices.
Market share for GRALISE ER remains limited, mainly targeted at patients intolerant to immediate-release formulations or requiring once-daily dosing.
How Has the Market for Nerve Pain Drugs Evolved?
Postherpetic neuralgia drugs have seen stable demand, driven by an aging population and increased awareness of nerve pain management. However, the market faces pressures from:
- Increased availability of generics reducing branded drug prices.
- Competition from other mechanisms (e.g., antidepressants, opioids).
- Strict prescribing guidelines to limit opioid use.
FDA approvals of new formulations provide limited incremental volume, mainly maintaining existing patient segments.
What Are the Estimated Revenue and Market Opportunities for GRALISE ER?
In 2022, clinical sales estimates for GRALISE ER ranged between $80 million and $150 million in the U.S. The drug's market share as of Q2 2022 is estimated at 4-6%.
Market growth primarily depends on:
- Insurance coverage policies.
- Physician prescribing patterns.
- Patient acceptance of extended-release formulations.
Key factors influencing sales include:
- Lack of patent protection: As of 2017, the patent for GRALISE ER expired, increasing generic competition.
- Pricing strategy: Current wholesale acquisition cost (WAC) for GRALISE ER is approximately $10–$13 per capsule, compared to $0.10–$0.50 for generics.
- Market penetration: Limited by generic substitution policies and cost sensitivity.
What Are the Price Projections for the Coming Years?
Considering market dynamics, price projections assume:
- Short-term (next 1–2 years): Maintains current WAC with minor fluctuations. A price of $10–$13 per capsule is maintained, barring aggressive discounts or formulary negotiations.
- Medium-term (3–5 years): As competition intensifies and generics capture a larger share, branded prices may decline by 20–40%. Expected average price: $6–$8 per capsule.
- Long-term (beyond 5 years): With patent expiration and generic penetration, price reductions to as low as $0.20–$0.50 per capsule are likely, similar to existing generic gabapentin.
How Do Pricing Trends Compare Internationally?
In Europe, branded gabapentin formulations tend to be priced 20–30% higher than in the U.S. Due to different reimbursement models, direct price comparisons vary, but generic prices are similarly low.
What Are the Regulatory and Policy Factors Affecting Pricing?
- Patent expirations: Reduce barriers to generic entry.
- Reimbursement policies: Favor generics; rebates and tiered formularies limit branded drug use.
- Prescribing guidelines: Encourage alternatives that are often cheaper.
What Will Impact Market Potential?
Key factors include:
- Patent status: Patent expiry increases generic competition, pressuring prices.
- Uptake by physicians: Willingness to prescribe extended-release formulations over generics.
- Insurance policies: Coverage of branded formulations influences patient access.
Conclusion: Market Outlook Summary
GRALISE ER faces significant generic competition, affecting its price stability and market share. Current prices are close to $10–$13 per capsule in the U.S. With increasing generics, pricing is expected to decline to the $1–$2 range within 5–7 years. The drug's growth opportunities will largely depend on its differentiation in efficacy, safety, and patient adherence advantages.
Key Takeaways
- Assets face intense price erosion due to generic gabapentin.
- Sales are expected to decline in the medium term as generics dominate.
- Price projections suggest a halving or more of current WAC within five years.
- Market growth depends on prescriber preference, insurance coverage, and patent status.
- International prices follow similar trends but vary by healthcare system.
FAQs
-
What is the main driver of GRALISE ER's market share?
Its advantage lies in once-daily dosing and potentially better tolerability, but generic competition limits market penetration. -
How soon will generic gabapentin dominate the market?
Generic gabapentin has been available since 2004; its dominance in pricing and prescribing is strong, expected to continue expanding. -
Is there a premium pricing opportunity for GRALISE ER?
Limited, unless recent clinical data show superior efficacy or safety profile, which could justify higher reimbursement. -
What regulatory protections does GRALISE ER have now?
Patent protections expired in 2017; no new exclusivity periods are current. -
What strategies could extend GRALISE ER's revenue?
Focus on niche indications, patient adherence benefits, or combination therapies might sustain higher prices.
References
[1] IQVIA. (2022). Pharmaceutical Market Reports.
[2] U.S. Food and Drug Administration. (2017). Patent expiration dates for GRALISE ER.
[3] Statista. (2022). Annual sales of gabapentin medications in the U.S.
[4] EvaluatePharma. (2022). Global neurology drug market projections.
More… ↓
