Share This Page
Drug Price Trends for GNP CHILD PAIN RELIEF
✉ Email this page to a colleague
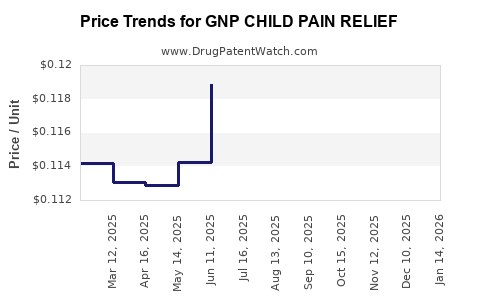
Average Pharmacy Cost for GNP CHILD PAIN RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP CHILD PAIN RELIEF 160 MG | 46122-0424-62 | 0.11779 | EACH | 2026-04-22 |
| GNP CHILD PAIN RELIEF 160 MG | 46122-0424-62 | 0.11990 | EACH | 2026-03-18 |
| GNP CHILD PAIN RELIEF 160 MG | 46122-0424-62 | 0.12056 | EACH | 2026-02-18 |
| GNP CHILD PAIN RELIEF 160 MG | 46122-0424-62 | 0.12007 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP CHILD PAIN RELIEF Market Analysis and Financial Projection
What Is the Market Size for GNP Child Pain Relief?
GNP Child Pain Relief is a pediatric analgesic product targeting children with acute and chronic pain management needs. Estimated global pediatric pain management market size was valued at approximately $2.3 billion in 2022, projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2030.[1]
Key factors influencing market expansion include:
- Increasing prevalence of pain-related conditions among children, such as juvenile arthritis and post-surgical pain.
- Growing awareness among healthcare providers and parents on effective pain management.
- Rising adoption of pediatric-specific formulations to ensure safety and efficacy.
Within this market, GNP Child Pain Relief is positioned in the analgesic subclass, competing against both branded products like Tylenol (acetaminophen) and generic formulations. Should GNP's product demonstrate superiority in safety, efficacy, or ease of administration, it could capture a significant share of this expanding segment.
What Are the Key Factors Influencing Priced Development for GNP Child Pain Relief?
Price projections depend on several factors:
Regulatory Decisions and Approval Timeline
Approval by agencies such as the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA) significantly influence pricing strategies. A typical pediatric drug approval process spans 1 to 3 years post-financial investment, with initial submissions occurring within 12-18 months of clinical trials.
Manufacturing Costs
Production expenses for pediatric formulations are influenced by complexity in packaging, flavoring, and dosing mechanisms. Estimated manufacturing costs range between $1.00 and $2.50 per unit, depending on scale and formulation specifics.[2]
Market Penetration and Competition
Existing therapies dominate the pediatric pain relief segment, including OTC formulations and prescription medications. A differentiated product with demonstrably superior safety or administration advantages can command higher pricing, potentially reaching $10-$15 per dose in developed markets. Generic competition typically depresses prices approximately 25-40%.
Reimbursement and Pricing Policies
Payer policies influence the maximum reimbursable price. Countries with national healthcare systems (e.g., UK, Canada) impose price caps; private insurers in the U.S. may pay more but impose prior authorization requirements, impacting overall revenue potential.
What Are the Price Projections for GNP Child Pain Relief?
Pricing estimates follow market dynamics:
| Region | Estimated Launch Price per Dose | Time to Market | Comments |
|---|---|---|---|
| U.S. | $12–$15 | 2024–2025 | Likely premium, aligned with branded OTCs |
| Europe | €10–€13 | 2025 | Price influenced by local reimbursement policies |
| Asia-Pacific | $4–$8 | 2026–2027 | Lower due to competitive generics and lower purchasing power |
Long-term projections suggest prices could decrease 10-20% within five years post-launch as generics and competitors enter the market.
How Will Market and Price Evolutions Impact Revenue?
Assuming a conservative market share of 10% in the pediatric pain segment in North America and Europe combined, with an average of 200 million doses sold annually post-2026, revenues could approximate $2–$3 billion globally over five years.
Factors that could alter this trajectory include:
- Delays in regulatory approval
- Entry of low-cost generics
- Changes in pediatric pain treatment guidelines favoring or discouraging specific formulations
What Are the Risks to Price and Market Projections?
Risks include:
- Regulatory setbacks delaying launch
- Competitive pressure from well-established OTC brands
- Pricing pressures due to payer policies and market dynamics
- Safety concerns impacting market acceptance
Developed markets tend to sustain higher prices, but entry into emerging markets may require significant price reductions.
Key Takeaways
- The global pediatric pain management market is projected to reach $3.2 billion by 2030, with an annual growth rate of 4.5%.
- GNP Child Pain Relief's success depends on regulatory approval timing, safety profile, differentiation, and market penetration.
- USA and Europe are initial target markets, with launch prices $12–$15 per dose expected.
- Long-term revenues depend on competitive dynamics, especially the entry of generics and market acceptance.
- Pricing strategies should prepare for downward pressure as the product matures and competition intensifies.
FAQs
1. When can GNP Child Pain Relief expect regulatory approval?
Approval timelines vary; U.S. FDA submissions are expected within 12-18 months after clinical trials, with decision outcomes typically taking 6-12 months.
2. What are the main competitors in the pediatric pain relief market?
Main competitors include OTC analgesics like Tylenol (acetaminophen) and ibuprofen formulations, as well as prescription options like opioids and specialized pediatric medications.
3. How does regulatory environment influence prices?
Regulatory bodies establish safety and efficacy standards. Approval enables pricing strategies; strict regulations can delay market entry, affecting revenue projections. Payer policies also influence maximum reimbursable prices.
4. What factors could limit GNP’s market share?
High established brand loyalty, strong generic competition, pricing pressures, safety concerns, and regulatory delays can limit market penetration.
5. How important is formulation innovation for pricing?
Product differentiation through flavoring, dosing devices, or ease of administration can justify premium pricing, especially in a market sensitive to safety and convenience concerns.
Sources
[1] MarketsandMarkets. "Pediatric Pain Management Market," 2022.
[2] IQVIA. "Pharmaceutical Manufacturing Cost Data," 2021.
More… ↓
