Drug Price Trends for GAS RELIEF
✉ Email this page to a colleague
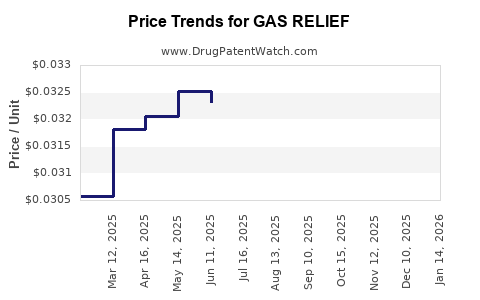
Average Pharmacy Cost for GAS RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GAS RELIEF 125 MG CHEW TABLET | 24385-0307-89 | 0.09984 | EACH | 2026-05-20 |
| GAS RELIEF 125 MG CHEW TABLET | 00536-1223-08 | 0.09984 | EACH | 2026-05-20 |
| GAS RELIEF 180 MG SOFTGEL | 70000-0566-01 | 0.04862 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for GAS RELIEF
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| GAS RELIEF EXTRA STRENGTH | Geri-Care Pharmaceutical Corp | 57896-0694-03 | 30 | 1.34 | 0.04467 | EACH | 2024-03-06 - 2026-06-14 | FSS |
| GAS RELIEF | Geri-Care Pharmaceutical Corp | 57896-0791-01 | 100 | 1.65 | 0.01650 | EACH | 2022-11-15 - 2026-06-14 | FSS |
| GAS RELIEF | Geri-Care Pharmaceutical Corp | 57896-0799-01 | 100 | 1.65 | 0.01650 | EACH | 2021-09-18 - 2026-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
