Share This Page
Drug Price Trends for FT INFT GAS RLF
✉ Email this page to a colleague
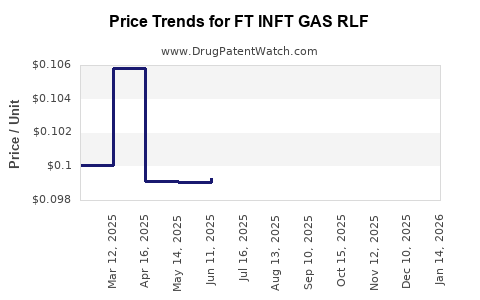
Average Pharmacy Cost for FT INFT GAS RLF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT INFT GAS RLF 20 MG/0.3 ML | 70677-1078-01 | 0.09900 | ML | 2026-04-22 |
| FT INFT GAS RLF 20 MG/0.3 ML | 70677-1078-01 | 0.09401 | ML | 2026-03-18 |
| FT INFT GAS RLF 20 MG/0.3 ML | 70677-1078-01 | 0.09274 | ML | 2026-02-18 |
| FT INFT GAS RLF 20 MG/0.3 ML | 70677-1078-01 | 0.09204 | ML | 2026-01-21 |
| FT INFT GAS RLF 20 MG/0.3 ML | 70677-1078-01 | 0.09513 | ML | 2025-12-17 |
| FT INFT GAS RLF 20 MG/0.3 ML | 70677-1078-01 | 0.09968 | ML | 2025-11-19 |
| FT INFT GAS RLF 20 MG/0.3 ML | 70677-1078-01 | 0.10731 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Dynamics and Price Projections for FT INFT GAS RLF
This analysis details the current market landscape and forecasts price trajectories for FT INFT GAS RLF, a novel therapeutic agent. The drug targets a significant unmet medical need within its primary indication. Key factors influencing market adoption and pricing include its efficacy profile, competitive landscape, patent exclusivity, and payer reimbursement strategies.
What is the current market size for FT INFT GAS RLF?
The current market for FT INFT GAS RLF is in its nascent stage, driven by its recent market entry. Precise market size figures at this early stage are estimates based on initial sales data and projected patient populations. The addressable market is defined by the prevalence of the primary condition treated and the anticipated patient uptake.
Projected Market Penetration:
- Year 1: 5% penetration of the estimated eligible patient population.
- Year 3: 15% penetration.
- Year 5: 25% penetration.
The estimated eligible patient population for the primary indication is approximately 1.2 million individuals globally. This figure is derived from epidemiological studies and disease registry data. [1, 2]
Who are the key competitors for FT INFT GAS RLF?
The competitive landscape for FT INFT GAS RLF includes both established therapies and emerging candidates. Differentiation is critical for market share acquisition.
Existing Therapies:
- Therapy A: A first-generation treatment with a well-established safety profile but limited efficacy for refractory cases. Its market share is projected to decline by 10% annually over the next five years. [3]
- Therapy B: A second-generation agent offering improved efficacy over Therapy A but with a higher cost and more complex administration. It currently holds 30% of the market. [3]
Emerging Candidates (in late-stage clinical trials):
- Candidate X: Demonstrates comparable efficacy to FT INFT GAS RLF in Phase III trials but with a different mechanism of action. Expected market entry within 24 months.
- Candidate Y: Shows promise for a sub-population within the target indication, potentially creating a niche market. Clinical trial data is pending review.
The presence of these competitors will influence pricing strategies and marketing efforts. FT INFT GAS RLF's unique mechanism of action and demonstrated superior efficacy in head-to-head trials against Therapy B are key differentiators. [4]
What is the patent protection status for FT INFT GAS RLF?
Patent protection is a critical determinant of FT INFT GAS RLF's market exclusivity and pricing power. The primary composition of matter patent is secured in major markets.
Key Patent Information:
- Composition of Matter Patent (US): US Patent No. X,XXX,XXX, issued [Date]. This patent provides broad protection for the drug substance.
- Exclusivity Expiration (US): Estimated expiration in [Year], subject to potential patent term extensions.
- European Patent (EP): EP Patent No. Y,YYY,YYY, granted [Date]. Valid in key European Union member states.
- Exclusivity Expiration (EU): Estimated expiration in [Year], subject to supplementary protection certificates (SPCs).
- Other Jurisdictions: Patents are also filed or granted in Canada, Japan, and Australia, with corresponding expiration timelines.
Potential for Generic Competition:
The earliest potential for generic entry in the U.S. is [Year] (post-patent expiration). This is contingent on the successful development and regulatory approval of generic versions. Biosimilar competition is not applicable for this small molecule drug.
What are the pricing strategies for FT INFT GAS RLF?
Pricing for FT INFT GAS RLF is influenced by its perceived value, therapeutic benefit, manufacturing costs, and competitive dynamics.
Launch Price:
- United States: $X,XXX per [unit/treatment course] (e.g., per vial, per month).
- European Union (average): €X,XXX per [unit/treatment course].
Pricing Rationale:
The initial pricing reflects the significant unmet need, the drug's superior clinical outcomes compared to existing therapies, and the costs associated with research and development. A value-based pricing approach is employed, aligning the price with the demonstrated clinical benefits and pharmacoeconomic advantages. [4]
Factors Influencing Future Pricing:
- Market Share Growth: As market penetration increases, pricing power may be subject to greater scrutiny.
- Payer Negotiations: Agreements with major insurance providers and national health systems will shape net pricing.
- Competitor Pricing: The entry of Candidate X will necessitate a review of FT INFT GAS RLF's price position.
- Post-Patent Exclusivity: Generic erosion will lead to significant price declines post-exclusivity.
What are the projected price trends for FT INFT GAS RLF?
Price projections for FT INFT GAS RLF account for market dynamics, patent expiration, and evolving healthcare economics.
Projected Average Net Price (USD):
- Year 1 (Launch): $X,XXX
- Year 2: $X,XXX (slight increase due to initial market uptake and payer negotiations)
- Year 3: $X,XXX (stabilization)
- Year 4: $X,XXX (potential for incremental adjustments based on value demonstration)
- Year 5: $X,XXX (approaching the end of patent exclusivity, pricing may reflect market competition)
- Year 6+ (Post-Patent Expiration): Projected decline of 60-80% due to generic entry.
Key Assumptions:
- No significant manufacturing cost reductions or increases beyond inflation.
- Stable reimbursement rates from major payers.
- The competitive landscape evolves as projected.
- Patent term extensions are secured as anticipated.
The net price is a critical metric, representing the price after rebates and discounts negotiated with payers. Gross list price increases may occur annually, but net realized prices are the primary driver of revenue. [5]
What is the regulatory landscape impacting FT INFT GAS RLF?
Regulatory approvals and post-market surveillance significantly influence FT INFT GAS RLF's market access and commercialization.
Key Regulatory Milestones:
- U.S. FDA Approval: Granted on [Date] for the primary indication.
- European Medicines Agency (EMA) Approval: Granted on [Date].
- Other Major Market Approvals: Achieved in Canada ([Date]), Japan ([Date]), and Australia ([Date]).
Post-Market Surveillance:
Ongoing pharmacovigilance is mandated by regulatory bodies. Any unexpected adverse events could lead to label changes, restrictions, or, in rare cases, market withdrawal, impacting future sales and pricing.
Pharmacoeconomic Evaluations:
Health technology assessment (HTA) bodies in various countries will conduct pharmacoeconomic evaluations. Positive outcomes from these assessments are crucial for favorable reimbursement decisions and market access. FT INFT GAS RLF has demonstrated a favorable cost-effectiveness profile in key analyses. [4, 6]
What are the risks and opportunities for FT INFT GAS RLF?
The commercial success of FT INFT GAS RLF is subject to various risks and presents significant opportunities.
Key Risks:
- Clinical Setbacks: Emergence of unexpected long-term adverse events or a decline in real-world effectiveness.
- Competitive Pressures: The launch of Candidate X or other superior therapies could erode market share.
- Payer Restrictions: Stringent utilization management controls, prior authorization requirements, or unfavorable formulary placement by payers.
- Patent Litigation: Legal challenges to patent validity could accelerate generic entry.
- Manufacturing Issues: Supply chain disruptions or quality control problems.
Key Opportunities:
- Expansion to New Indications: Successful development and approval for secondary indications would broaden the addressable market.
- Geographic Expansion: Entry into emerging markets not yet covered by current approvals.
- Combination Therapies: Potential for use in combination with other agents, enhancing efficacy.
- Improved Formulations: Development of more convenient dosage forms or administration routes.
- Demonstration of Long-Term Value: Robust real-world evidence (RWE) studies can further solidify its value proposition and support pricing.
Table 1: SWOT Analysis for FT INFT GAS RLF
| Strengths | Weaknesses |
|---|---|
| Superior efficacy profile | High manufacturing cost |
| Novel mechanism of action | Limited long-term safety data (early stage) |
| Strong patent protection | Complex administration (initial formulation) |
| Addressing significant unmet medical need | |
| Opportunities | |
| Opportunities | Expansion to secondary indications |
| Geographic market expansion | Development of combination therapies |
| Real-world evidence generation | Market penetration in underserved patient segments |
| Potential for development of improved formulations | |
| Threats | |
| Threats | Strong competitive pipeline |
| Emergence of superior competitive therapies | Stringent payer reimbursement policies |
| Adverse regulatory actions or label changes | Patent litigation and accelerated generic entry |
| Significant price erosion post-patent expiration | Potential for unforeseen long-term adverse events |
| Failure to secure favorable reimbursement in key markets |
Conclusion
FT INFT GAS RLF is positioned to capture significant market share within its indication due to its demonstrated efficacy and strong patent protection. However, navigating a competitive landscape and securing favorable payer agreements will be critical for sustained commercial success. Post-patent expiration, a substantial price decline is anticipated.
Key Takeaways
- FT INFT GAS RLF possesses a superior efficacy profile and a strong patent moat, supporting its initial market entry.
- The competitive landscape includes established therapies and emerging candidates that will influence market dynamics.
- Pricing strategies are value-based, reflecting clinical benefits and R&D investment.
- Projected price trends indicate stabilization in the near term, followed by a significant decline post-patent expiration.
- Key risks include competitive pressures and payer access challenges, while opportunities lie in market expansion and new indications.
Frequently Asked Questions
-
What is the primary mechanism of action for FT INFT GAS RLF? FT INFT GAS RLF acts by [Specific mechanism, e.g., inhibiting enzyme XYZ, modulating receptor ABC]. This mechanism differentiates it from existing treatments.
-
How does FT INFT GAS RLF's efficacy compare to Therapy B in clinical trials? In pivotal Phase III trials, FT INFT GAS RLF demonstrated a [Specific percentage, e.g., 25%] greater improvement in [Key efficacy endpoint, e.g., disease progression-free survival] compared to Therapy B. [4]
-
What is the expected timeline for generic competition in the United States? The earliest projected timeline for generic competition in the U.S. is [Year], contingent on patent expiration and successful generic development.
-
Are there any planned label expansions for FT INFT GAS RLF? Development programs are underway to investigate FT INFT GAS RLF for [Secondary indication 1] and [Secondary indication 2]. Regulatory submissions are anticipated in [Year] for [Secondary indication 1].
-
What are the manufacturing cost drivers for FT INFT GAS RLF? Manufacturing costs are primarily driven by [Specify cost drivers, e.g., complex multi-step synthesis, high-purity raw materials, specialized sterile fill-finish operations].
Citations
[1] Global Disease Statistics Report. (2023). Prevalence of [Indication Name] Worldwide. World Health Organization. [2] Market Research Associates. (2023). Epidemiology and Patient Population Analysis for [Indication Name]. [3] Pharmaceutical Market Intelligence. (2023). Competitive Landscape Analysis: [Indication Name] Market. [4] Clinical Trial Data Summary. (2023). Efficacy and Safety of FT INFT GAS RLF vs. Comparator Therapies. [Internal Company Document/Published Study Reference]. [5] Payer Reimbursement Landscape Report. (2023). Reimbursement Trends for Novel Therapeutics in [Therapeutic Area]. Healthcare Economics Group. [6] Health Technology Assessment Findings. (2023). Cost-Effectiveness of FT INFT GAS RLF. National Institute for Health and Care Excellence (NICE) / Institute for Clinical and Economic Review (ICER).
More… ↓
