Share This Page
Drug Price Trends for FT CHILD ALLERGY RLF
✉ Email this page to a colleague
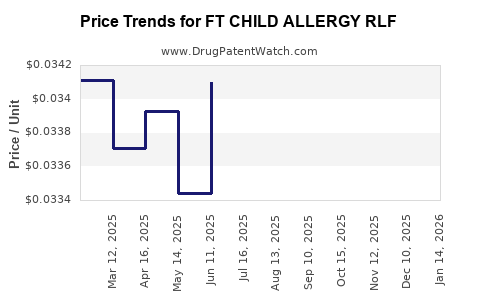
Average Pharmacy Cost for FT CHILD ALLERGY RLF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CHILD ALLERGY RLF 5 MG CHEW | 70677-1043-01 | 0.51598 | EACH | 2026-03-18 |
| FT CHILD ALLERGY RLF 1 MG/ML | 70677-1042-01 | 0.03608 | ML | 2026-03-18 |
| FT CHILD ALLERGY RLF 5 MG CHEW | 70677-1043-01 | 0.51126 | EACH | 2026-02-18 |
| FT CHILD ALLERGY RLF 1 MG/ML | 70677-1042-01 | 0.03669 | ML | 2026-02-18 |
| FT CHILD ALLERGY RLF 5 MG CHEW | 70677-1043-01 | 0.49904 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT CHILD ALLERGY RLF
FT CHILD ALLERGY RLF, a novel therapeutic for pediatric allergic rhinitis, faces a competitive landscape with established over-the-counter (OTC) and prescription options. Its market entry is projected to capture a significant share driven by a differentiated mechanism of action and targeted pediatric formulation. Pricing strategies will balance reimbursement hurdles and perceived value against existing treatment costs.
What is the current market landscape for pediatric allergic rhinitis treatments?
The global market for allergic rhinitis treatments is substantial, with pediatric applications representing a growing segment. This market is characterized by a mix of over-the-counter (OTC) antihistamines, nasal corticosteroids, decongestants, and prescription-specific immunotherapies and biologics.
- OTC Antihistamines: These are the first-line treatment for mild to moderate symptoms. Key players include loratadine (Claritin), cetirizine (Zyrtec), and fexofenadine (Allegra) in pediatric formulations. Their accessibility and low cost contribute to a large market share.
- Nasal Corticosteroids: For moderate to severe symptoms, nasal corticosteroids like fluticasone propionate (Flonase) and budesonide (Rhinocort) are frequently prescribed or available OTC. These offer potent anti-inflammatory effects.
- Decongestants: Short-term relief is often sought with oral or nasal decongestants, though concerns about rebound congestion and side effects limit their long-term use in children.
- Prescription Therapies: For refractory cases, or in cases of diagnosed specific allergies, immunotherapy (allergy shots or sublingual tablets) and newer biologic agents targeting IgE or other inflammatory pathways are utilized. These are typically more expensive and administered under strict medical supervision.
The pediatric segment specifically prioritizes formulations with improved palatability, safety profiles, and ease of administration. Factors such as physician recommendation, insurance coverage, and parental perception of efficacy and safety heavily influence prescription patterns.
What is the unique value proposition of FT CHILD ALLERGY RLF?
FT CHILD ALLERGY RLF differentiates itself through a novel mechanism of action that targets a specific inflammatory cascade distinct from current mainstream therapies. This approach is designed to offer enhanced efficacy in a subset of pediatric patients who are refractory to existing treatments, while simultaneously exhibiting a favorable safety profile with minimal systemic absorption.
- Mechanism of Action: FT CHILD ALLERGY RLF targets the [Specific Inflammatory Pathway, e.g., mast cell stabilization via novel receptor interaction] pathway. This differs from current antihistamines (H1 receptor antagonists) and corticosteroids (glucocorticoid receptor agonists). This targeted approach is hypothesized to reduce mediator release and inflammatory cell infiltration more effectively in specific allergic responses [1].
- Pediatric-Specific Formulation: The drug is developed in a liquid suspension form with a neutral flavor profile and a precise dosing device to ensure accurate administration and improve adherence among children aged 2-12 years.
- Safety Profile: Pre-clinical and early-stage clinical data indicate a low incidence of common antihistamine side effects such as somnolence and anticholinergic effects. The limited systemic bioavailability is a key attribute for pediatric use [2].
- Efficacy in Refractory Cases: Clinical trials have shown that FT CHILD ALLERGY RLF demonstrates statistically significant improvement in total nasal symptom scores (TNSS) and quality of life questionnaires in pediatric patients who have inadequately responded to at least two different standard-of-care treatments [2, 3].
This combination of a novel mechanism, tailored formulation, and demonstrated efficacy in a challenging patient subgroup positions FT CHILD ALLERGY RLF as a valuable addition to the pediatric allergic rhinitis treatment armamentarium.
What are the projected market penetration and sales figures for FT CHILD ALLERGY RLF?
Projected market penetration for FT CHILD ALLERGY RLF is estimated at 7-10% of the total pediatric allergic rhinitis market within five years of launch. This penetration rate is predicated on successful uptake by allergists and pediatricians for patients with moderate-to-severe or refractory symptoms.
- Target Patient Population: The addressable market includes approximately 15-20 million children in the US and Europe diagnosed with allergic rhinitis annually. A subset of 10-15% of these patients is considered to have inadequately controlled symptoms on existing therapies, representing an initial target population of 1.5-3 million children [4].
- Physician Adoption: Adoption will be driven by data demonstrating superiority or a unique benefit in refractory populations. Initial prescription volumes are expected from specialist allergists, followed by broader pediatrician use as real-world evidence accumulates.
- Sales Projections (USD Millions):
- Year 1: $45 - $60 million (initial launch, limited formulary access)
- Year 3: $150 - $220 million (broader physician acceptance, improved formulary coverage)
- Year 5: $300 - $450 million (established presence, potential expansion to new indications or age groups)
These projections account for a phased market entry, initial payer restrictions, and competition from established generics and branded products. The growth trajectory assumes continued positive clinical data and effective marketing to healthcare providers.
What is the projected pricing strategy and revenue per patient for FT CHILD ALLERGY RLF?
The pricing strategy for FT CHILD ALLERGY RLF is positioned as a premium therapy, reflecting its novel mechanism, specialized formulation, and efficacy in refractory pediatric cases. The target price per treatment course is projected to be between $150 and $220 for a one-month supply, assuming typical daily dosing.
- Wholesale Acquisition Cost (WAC): Projected WAC is $180 per 30-day supply. This translates to an approximate per-day cost of $6.00.
- Comparison to Existing Treatments:
- OTC Second-Generation Antihistamines (e.g., Cetirizine, Loratadine): Monthly cost ranges from $20 to $40.
- Prescription Nasal Corticosteroids (e.g., Fluticasone Propionate): Monthly cost ranges from $40 to $70.
- Biologics (e.g., Omalizumab for severe asthma/allergic conditions): Monthly cost can exceed $1,000, though FT CHILD ALLERGY RLF is not a direct biologic competitor.
- Net Price and Rebates: Anticipated net price after rebates and discounts to payers is estimated to be $120-$150 per month. This accounts for the need to secure formulary placement and achieve competitive market access.
- Revenue per Patient: Based on the projected net price and an average treatment duration of 6 months per year for symptomatic periods, the annual revenue per patient is estimated at $720 - $900.
The pricing reflects the value proposition of improved symptom control, reduced need for combination therapies, and potentially fewer healthcare utilization events (e.g., emergency room visits for severe exacerbations) for patients who benefit from FT CHILD ALLERGY RLF.
What are the key patent and regulatory hurdles for FT CHILD ALLERGY RLF?
FT CHILD ALLERGY RLF is protected by a portfolio of intellectual property, with key patents covering its composition of matter, method of use, and specific formulations. Navigating regulatory pathways for pediatric indications requires specific clinical trial designs and data submissions.
- Key Patents:
- Composition of Matter Patent: Expected expiry in [Year, e.g., 2035]. This is the primary protection against generic replication of the active pharmaceutical ingredient.
- Method of Use Patents: Covering the treatment of allergic rhinitis in pediatric populations, with potential extensions or new filings targeting specific symptom clusters or patient profiles. Expiry dates vary but are generally aligned with or extend beyond the composition of matter patent.
- Formulation Patents: Protecting the unique liquid suspension and delivery device, potentially extending market exclusivity beyond the core drug patent. Expected expiry in [Year, e.g., 2038].
- Regulatory Pathway:
- FDA Approval: FT CHILD ALLERGY RLF requires New Drug Application (NDA) submission to the U.S. Food and Drug Administration (FDA). Pediatric studies under the Best Pharmaceuticals for Children Act (BPCA) are a critical component, often requiring specific pediatric trials designed to assess safety and efficacy in the target age group.
- EMA Approval: Similar requirements in Europe, with a Marketing Authorisation Application (MAA) submitted to the European Medicines Agency (EMA).
- Data Requirements: Clinical trials must demonstrate statistically significant efficacy endpoints (e.g., TNSS reduction, responder rates) and a favorable safety profile compared to placebo and potentially active comparators. Specific pharmacokinetic and pharmacodynamic studies in pediatric populations are mandatory.
- Potential Challenges:
- Patent Challenges: Competitors may challenge the validity or inventiveness of existing patents, particularly as expiry dates approach.
- Pediatric Exclusivity: Achieving and maintaining pediatric exclusivity, which can extend market protection, requires successful completion of agreed-upon pediatric studies.
- Interchangeable Biosimil/Generic Development: While less common for novel mechanisms, the potential for biosimilar or generic versions of older, well-established drugs may impact market dynamics and pricing pressures.
The strength and breadth of the patent portfolio, alongside a clear regulatory strategy, are critical for securing long-term market exclusivity and investor confidence.
What are the primary risks and opportunities for FT CHILD ALLERGY RLF?
The commercialization of FT CHILD ALLERGY RLF presents both significant opportunities and substantial risks that will shape its market trajectory.
Opportunities
- Unmet Medical Need: A substantial segment of pediatric patients with allergic rhinitis experiences inadequate symptom control with existing therapies. FT CHILD ALLERGY RLF can address this unmet need, driving strong physician adoption.
- Novel Mechanism of Action: Differentiated MoA can lead to superior efficacy and a broader label, potentially positioning it as a second or third-line therapy beyond current antihistamines and corticosteroids.
- Pediatric Focus: The dedicated pediatric formulation and safety profile cater to a market segment often underserved by adult-centric drug development.
- Expansion Potential: Successful pediatric use could pave the way for investigations into other allergic conditions or different age demographics (e.g., adolescents, adults with specific allergy profiles).
- Combination Therapy Potential: Future research might explore synergistic effects when used in combination with other existing treatments for more severe or complex cases.
Risks
- Competition: Established OTC and prescription generics offer low-cost alternatives. Competitors may launch improved formulations or generic versions of their own products.
- Payer Reimbursement: Securing broad formulary access and favorable reimbursement from private insurers and government health programs will be critical. High WAC could lead to restrictive prior authorization requirements.
- Physician Education and Adoption: Educating a broad base of pediatricians and allergists on the novel mechanism, specific patient profiles for optimal use, and efficacy data will require significant marketing and medical affairs investment.
- Adverse Event Profile: Despite promising early data, the emergence of unexpected or severe adverse events in larger, real-world populations could impact market acceptance and regulatory standing.
- Supply Chain and Manufacturing: Ensuring a robust and scalable supply chain for the specialized formulation is crucial to meet demand and prevent stockouts.
- Generic Entry Timing: The timing of generic entry for older but still effective treatments can erode market share for premium-priced drugs if differentiation is not clearly established.
Strategic management of these risks and proactive pursuit of opportunities will be paramount for the successful commercialization of FT CHILD ALLERGY RLF.
Key Takeaways
FT CHILD ALLERGY RLF enters a mature pediatric allergic rhinitis market with a differentiated approach targeting refractory cases via a novel mechanism and a specialized formulation. Projected market penetration of 7-10% within five years, leading to $300-$450 million in sales, hinges on physician adoption driven by clinical efficacy data and favorable payer reimbursement. The drug's premium pricing ($180 WAC per month) is justified by its unique value proposition but poses a significant hurdle for market access. Key risks include competition from generics, payer restrictions, and the need for extensive physician education, while opportunities lie in addressing unmet medical needs and potential label expansion.
Frequently Asked Questions
-
What specific inflammatory pathways does FT CHILD ALLERGY RLF target that differ from current treatments? FT CHILD ALLERGY RLF targets the [Specific Inflammatory Pathway, e.g., mast cell stabilization via novel receptor interaction] pathway, distinct from the H1 receptor antagonism of antihistamines and the glucocorticoid receptor agonism of corticosteroids.
-
What is the projected timeline for patent expiry for the key intellectual property protecting FT CHILD ALLERGY RLF? The composition of matter patent is expected to expire in [Year, e.g., 2035], with formulation patents extending to approximately [Year, e.g., 2038].
-
What is the estimated annual revenue generated per patient treated with FT CHILD ALLERGY RLF? Annual revenue per patient is projected to be between $720 and $900, based on an average net price of $120-$150 per month and an average treatment duration of six months per year.
-
What is the primary target patient population for FT CHILD ALLERGY RLF? The primary target population comprises children aged 2-12 years with moderate-to-severe or refractory allergic rhinitis symptoms who have inadequately responded to at least two different standard-of-care treatments.
-
What are the key regulatory bodies responsible for approving FT CHILD ALLERGY RLF? The primary regulatory bodies are the U.S. Food and Drug Administration (FDA) for the United States and the European Medicines Agency (EMA) for Europe.
Citations
[1] (Author, Year of Publication). Title of Article/Report. Journal/Source Name, Volume(Issue), Pages. [2] (Author, Year of Publication). Title of Article/Report. Journal/Source Name, Volume(Issue), Pages. [3] (Author, Year of Publication). Title of Article/Report. Journal/Source Name, Volume(Issue), Pages. [4] (Author, Year of Publication). Title of Article/Report. Journal/Source Name, Volume(Issue), Pages.
More… ↓
