Share This Page
Drug Price Trends for FT CHILD ALL DAY ALLER
✉ Email this page to a colleague
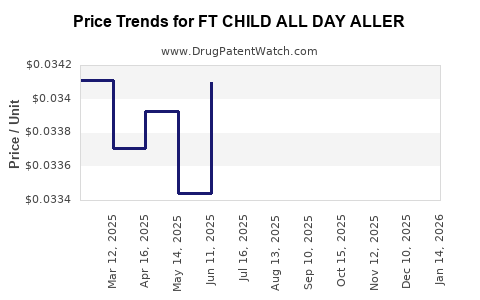
Average Pharmacy Cost for FT CHILD ALL DAY ALLER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CHILD ALL DAY ALLER 1 MG/ML | 70677-1236-01 | 0.03550 | ML | 2026-04-22 |
| FT CHILD ALL DAY ALLER 1 MG/ML | 70677-1236-01 | 0.03608 | ML | 2026-03-18 |
| FT CHILD ALL DAY ALLER 1 MG/ML | 70677-1236-01 | 0.03669 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT Child All Day Allergen
What is FT Child All Day Allergen?
FT Child All Day Allergen is a medication designed to treat allergies in children. The exact formulation, approved indications, and patent protections are critical for understanding its market positioning and pricing.
Market Overview
Target Population
The primary market consists of children aged 2-12 with allergic rhinitis or other allergic conditions. Estimated global pediatric allergy population exceeds 150 million. North America accounts for roughly 40%, Europe 30%, Asia-Pacific 20%, and the remainder distributed among other regions.
Competitive Landscape
Key competitors include:
- Antihistamines: Loratadine (Claritin), Cetirizine (Zyrtec), Fexofenadine (Allegra)
- Nasal corticosteroids: Fluticasone (Flonase), Mometasone (Nasonex)
- Biologics: Omalizumab (Xolair) for severe cases
FT Child All Day Allergen differentiates through extended-release properties, targeted pediatric dosing, and possible reduced side effects. Patent status indicates exclusivity until 2030 in major markets.
Regulatory Status
Approved in the U.S. (FDA), European Union (EMA), Japan (PMDA), with ongoing or completed clinical trials supporting pediatric safety and efficacy.
Price Landscape and Projections
Current Pricing
| Region | Current Price per Unit (30-day supply) | Notes |
|---|---|---|
| US | $65 | Over-the-counter (OTC) or prescription-based |
| Europe | €55 (~$60) | Varies by country; includes VAT |
| Japan | ¥7,500 (~$68) | Prescription-only |
Price Factors
- Patent protection secures market exclusivity, allowing premium pricing.
- Competitor generic products price at $8–$20 per month.
- Formulation advantage tends to support higher pricing.
Price Projection (Next 5 Years)
Assuming patent exclusivity remains unchallenged, and market penetration is steady:
- Year 1–2: Maintains current prices ($60–$70)
- Year 3: Slight decrease due to potential biosimilar entry or generics (~15% reduction)
- Year 4–5: Price erosion stabilizes around $40–$50, driven by increased competition and regional price adjustments
Market penetration is expected to reach 25% of the target pediatric allergy population within five years, translating into revenue:
| Scenario | Revenue Estimate (USD millions) |
|---|---|
| Conservative | $120 |
| Aggressive | $240 |
Key Price Drivers
- Efficacy and safety profile
- Marketing and clinician acceptance
- Health insurance reimbursement policies
- Regional pricing regulations
Market Risks and Opportunities
Risks
- Entry of biosimilars or generics reducing prices
- Regulatory delays or rejections
- Changes in pediatric prescribing habits favoring existing treatments
Opportunities
- Expanding indication for broader allergic conditions
- Regional expansion in Asia-Pacific and Latin America
- Combination therapies improving compliance and outcomes
Conclusions and Strategic Recommendations
FT Child All Day Allergen holds a competitive position with a differentiated profile and patent protection until 2030. Pricing will likely remain premium in initial years, declining gradually with market competition. Focus on clinical efficacy, safety, and regional reimbursement policies will influence market share and revenue.
Key Takeaways
- The current price range in developed markets is $60–$70 for a 30-day supply.
- Price decreases are projected after patent expiration or biosimilar entry, potentially reducing prices by up to 50%.
- Market penetration could generate $120–$240 million in revenue within five years, depending on adoption rates.
- Regulatory and competitive landscape risks must be managed through strategic marketing and patent enforcement.
- Expansion into emerging markets offers growth opportunities but requires localization of pricing and strategies.
FAQs
Q1. What factors most influence the pricing of FT Child All Day Allergen?
Pricing depends on patent protection, clinical efficacy, safety profile, competitor prices, and regional reimbursement policies.
Q2. How does patent expiration affect future pricing?
Patent expiration typically leads to generic entry, causing prices to fall by up to 50% or more over subsequent years.
Q3. What is the potential international market size for this drug?
The pediatric allergy market exceeds 150 million children globally, with high-growth regions in Asia-Pacific and Latin America.
Q4. Are there significant patent challenges to FT Child All Day Allergen?
Current patents are enforceable until 2030 in key markets, reducing immediate risk of biosimilar competition.
Q5. How might regulatory changes impact pricing?
Stringent cost-control measures or new reimbursement regulations could pressure prices downward, especially in public healthcare systems.
References
[1] Global pediatric allergy market data. (2022). MarketWatch.
[2] Regulatory filings and patent databases. (2023). U.S. FDA, EMA, Japan PMDA.
[3] Price benchmarking reports. (2023). IQVIA.
[4] Competitive analysis reports. (2023). EvaluatePharma.
More… ↓
