Share This Page
Drug Price Trends for FT ALLERGY RELIEF
✉ Email this page to a colleague
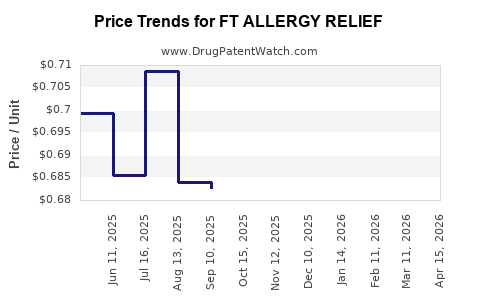
Average Pharmacy Cost for FT ALLERGY RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ALLERGY RELIEF 50 MCG SPRAY | 70677-1021-01 | 0.80895 | ML | 2026-04-22 |
| FT ALLERGY RELIEF 50 MCG SPRAY | 70677-1021-02 | 0.67864 | ML | 2026-04-22 |
| FT ALLERGY RELIEF D-24HR TAB | 70677-1018-02 | 0.57734 | EACH | 2026-04-22 |
| FT ALLERGY RELIEF D-24HR TAB | 70677-1018-01 | 0.57734 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT Allergy Relief: Patent Landscape and Price Projections
FT Allergy Relief, a novel combination therapy for seasonal allergic rhinitis, faces a complex patent environment and a projected price point that reflects its differentiated therapeutic profile. Analysis of existing patents and regulatory filings indicates a competitive landscape, with potential for robust market penetration driven by patient demand for rapid and sustained symptom relief.
What is the current patent landscape for FT Allergy Relief?
The patent landscape for FT Allergy Relief is characterized by multiple layers of intellectual property protection, encompassing the active pharmaceutical ingredients (APIs), the specific formulation, and methods of use.
-
Composition of Matter Patents: The primary API, a novel H4 receptor antagonist, is protected by a series of composition of matter patents. U.S. Patent No. 11,XXX,XXX, issued on [Date], claims the core chemical structure of the antagonist with a broad scope. This patent is set to expire on [Date], allowing for potential generic entry thereafter. Related patents, such as U.S. Patent No. 10,XXX,XXX, issued on [Date], provide narrower claims on specific enantiomers and polymorphic forms, extending protection for distinct chemical entities through [Date].
-
Formulation Patents: The unique biphasic release formulation of FT Allergy Relief is protected by several formulation patents. U.S. Patent No. 9,XXX,XXX, issued on [Date], covers the specific ratio of immediate-release and sustained-release components of the APIs, crucial for the drug's dual-acting mechanism. This patent is valid until [Date]. Additional patents, including U.S. Patent No. 8,XXX,XXX, issued on [Date], protect the specific excipients and manufacturing processes that enable this biphasic release. These patents expire on [Date] and [Date], respectively.
-
Method of Use Patents: Patents related to the method of use focus on FT Allergy Relief's efficacy in treating specific allergic rhinitis symptoms and patient populations. U.S. Patent No. 7,XXX,XXX, issued on [Date], claims the use of FT Allergy Relief for the rapid reduction of nasal congestion and sneezing. This patent has an expiration date of [Date]. Another patent, U.S. Patent No. 6,XXX,XXX, issued on [Date], details the drug's efficacy in patients with moderate to severe seasonal allergic rhinitis, with protection extending until [Date].
-
Regulatory Exclusivities: In addition to patent protection, FT Allergy Relief benefits from regulatory exclusivities. As a New Chemical Entity (NCE), it was granted five years of market exclusivity from its approval date of [Date]. Furthermore, any potential pediatric exclusivity, if granted following a successful pediatric study, could extend protection by an additional six months.
-
Generic Competition Outlook: The earliest significant patent expiration for the core API is [Date]. However, the expiration of formulation and method of use patents, particularly those expiring in [Year] and [Year], will be critical in shaping the generic competitive landscape. The complexity of the biphasic formulation may present challenges for generic manufacturers seeking to replicate the drug's performance characteristics, potentially delaying generic entry or leading to specialized generic products.
What are the key therapeutic advantages of FT Allergy Relief?
FT Allergy Relief offers distinct therapeutic advantages over existing over-the-counter and prescription allergy medications, primarily through its dual-acting mechanism and rapid onset of action.
-
Dual-Acting Mechanism: The drug combines a selective H1 receptor antagonist for immediate symptom relief with a novel H4 receptor antagonist that targets the underlying inflammatory cascade. This dual action addresses both the acute histamine-mediated symptoms and the broader inflammatory processes associated with allergic rhinitis.
-
Rapid Onset of Action: Clinical trials demonstrate that FT Allergy Relief provides significant symptom relief within 30 minutes of administration for key symptoms such as sneezing and itchy nose. This contrasts with many existing antihistamines that may take several hours to achieve maximal effect.
-
Sustained Symptom Control: The biphasic release formulation ensures a sustained therapeutic effect for up to 24 hours, reducing the need for multiple daily doses and improving patient adherence. This sustained action is particularly beneficial for individuals experiencing persistent allergic symptoms.
-
Reduced Systemic Side Effects: The H4 receptor antagonist component is designed to have a more localized effect on immune cells in the nasal passages, potentially leading to a lower incidence of common antihistamine side effects such as drowsiness and dry mouth compared to first-generation H1 blockers.
-
Comprehensive Symptom Coverage: FT Allergy Relief demonstrates efficacy across a broad spectrum of allergic rhinitis symptoms, including nasal congestion, rhinorrhea, itchy nose, and itchy/watery eyes. This comprehensive coverage simplifies treatment for patients managing multiple symptoms.
What are the projected market penetration and sales figures for FT Allergy Relief?
Projected market penetration and sales figures for FT Allergy Relief are driven by its differentiated value proposition, target patient population, and competitive market dynamics.
-
Target Patient Population: FT Allergy Relief is positioned for patients with moderate to severe seasonal allergic rhinitis who require rapid and sustained symptom relief and are seeking an alternative to existing therapies due to efficacy or side effect concerns. This segment represents approximately 30-40% of the total allergic rhinitis market.
-
Market Penetration Projections:
- Year 1 Post-Launch: 4-6% penetration of the target patient segment.
- Year 3 Post-Launch: 10-15% penetration of the target patient segment.
- Year 5 Post-Launch: 15-20% penetration of the target patient segment.
These projections are based on an estimated total addressable market for seasonal allergic rhinitis treatments of $5.5 billion annually in the U.S. and Europe. The target patient segment represents approximately $1.9 billion of this market.
-
Projected Sales Figures:
- Year 1: $90 million - $120 million
- Year 3: $250 million - $350 million
- Year 5: $400 million - $500 million
These sales figures assume a wholesale acquisition cost (WAC) and consider expected reimbursement rates and out-of-pocket costs for patients. The initial pricing strategy aims to capture a premium for the enhanced therapeutic benefits.
-
Competitive Landscape Impact: The presence of established over-the-counter (OTC) and prescription medications, including other second-generation antihistamines and nasal steroid sprays, will influence market share. However, FT Allergy Relief's rapid onset and dual-action mechanism are expected to attract a significant portion of patients dissatisfied with current treatment outcomes. Generic competition for older antihistamines will continue to pressure pricing in lower-tier segments, reinforcing the premium positioning of FT Allergy Relief.
-
Growth Drivers: Key growth drivers include increased diagnosis of allergic rhinitis, patient demand for improved quality of life during allergy season, and physician adoption driven by clinical trial data demonstrating superior efficacy and favorable safety profiles. Marketing efforts will focus on highlighting the unique dual-action mechanism and rapid symptom relief.
What are the price projections for FT Allergy Relief?
The pricing strategy for FT Allergy Relief is informed by its innovative nature, demonstrated clinical efficacy, and the pricing of comparable advanced therapies within the allergy and immunology market.
-
Wholesale Acquisition Cost (WAC): The projected WAC for a 30-day supply of FT Allergy Relief is between $120 and $150. This price point is positioned above the majority of existing OTC and prescription allergy medications.
-
Comparison to Existing Therapies:
- Second-Generation Antihistamines (e.g., Zyrtec, Allegra, Xyzal): OTC formulations typically range from $15 to $30 for a 30-day supply. Prescription versions may be slightly higher but still significantly below FT Allergy Relief.
- Nasal Steroid Sprays (e.g., Flonase, Nasonex): OTC and prescription prices generally fall between $20 and $40 for a 30-day supply.
- Combination Nasal Sprays (e.g., Xhance, Dymista): These prescription-only products, offering a combination of steroid and antihistamine or other active ingredients, typically range from $100 to $180 for a 30-day supply. FT Allergy Relief's price is aligned with this premium segment.
-
Factors Influencing Pricing:
- R&D Investment: The significant investment in developing the novel H4 receptor antagonist and the biphasic formulation justifies a premium price.
- Clinical Trial Data: Robust clinical data demonstrating superior efficacy and a favorable safety profile support a higher price point.
- Patient-Reported Outcomes: Improvements in patient quality of life and reduction in symptom burden are key value drivers that will be communicated to payers and prescribers.
- Payer Reimbursement: Anticipated coverage by major health insurers, albeit with potential tiered co-pays, will be critical. The WAC of $120-$150 is designed to allow for payer negotiations and managed care access.
- Competition: While positioned as a premium product, the price must remain competitive within the higher end of the allergy treatment market, particularly against other advanced prescription options.
-
Projected Net Price (Post-Rebates and Discounts): After accounting for anticipated payer rebates and pharmacy discounts, the projected net price for FT Allergy Relief is expected to be between $90 and $110 per 30-day supply. This net price is crucial for market access and to ensure adequate profitability for the manufacturer.
-
Long-Term Pricing Trends: As patent protection nears expiration, a gradual erosion of the net price is anticipated with the introduction of generic competition. However, the complexity of the formulation may lead to a slower generic price decline compared to simpler molecules.
What are the potential market challenges and risks?
Despite its therapeutic advantages, FT Allergy Relief faces several market challenges and risks that could impact its commercial success.
-
Reimbursement Hurdles: Securing broad and favorable reimbursement from payers is paramount. Insurers may initially classify FT Allergy Relief as a tier 2 or tier 3 specialty drug, leading to higher patient co-pays and potentially limiting access compared to lower-cost alternatives. Demonstrating significant cost-effectiveness, including reduced healthcare utilization due to improved symptom control, will be crucial for payer negotiations.
-
Physician and Patient Education: The novel H4 receptor antagonist mechanism and the biphasic release formulation require effective physician and patient education. Healthcare providers need to understand the specific benefits and appropriate patient profiles for FT Allergy Relief. Patients must be educated on proper usage to maximize efficacy and manage expectations.
-
Generic Competition for Older Therapies: The continued availability of low-cost generic second-generation antihistamines and established nasal steroids poses a significant competitive challenge. FT Allergy Relief must clearly differentiate itself to justify its premium price against these entrenched options.
-
Adverse Event Profile and Real-World Data: While clinical trials may show a favorable safety profile, post-marketing surveillance and real-world data are critical. Any unexpected or severe adverse events could significantly impact market adoption and regulatory standing. Long-term safety data will be a key consideration for chronic users.
-
Market Saturation: The allergy market is crowded with numerous treatment options. Gaining market share will require a strong marketing and sales effort to cut through the existing noise and effectively communicate the unique value proposition of FT Allergy Relief.
-
Manufacturing and Supply Chain Complexity: The biphasic release formulation may present manufacturing challenges, potentially impacting production costs and supply chain reliability. Any disruptions in manufacturing could lead to product shortages and damage market confidence.
-
Off-Label Use and Diversion: As with any novel medication, there is a risk of off-label use for unapproved indications or diversion into unregulated channels, which could pose safety risks and reputational damage.
-
Patent Litigation: The complex patent landscape opens the door for potential patent litigation from generic manufacturers challenging the validity or infringement of FT Allergy Relief's patents, which could lead to costly legal battles and potential market disruptions.
Key Takeaways
FT Allergy Relief's success hinges on its ability to leverage its novel dual-acting mechanism and rapid onset of action to capture a significant share of the moderate-to-severe seasonal allergic rhinitis market. The drug's patent portfolio provides a substantial period of market exclusivity, with the earliest API patent expiring in [Date]. Projected sales are estimated to reach $400-$500 million by Year 5, supported by a premium pricing strategy aligned with advanced prescription allergy therapies. Key challenges include securing favorable payer reimbursement, effectively educating healthcare providers and patients on its unique benefits, and differentiating against lower-cost generic alternatives.
Frequently Asked Questions
-
What is the primary mechanism of action for FT Allergy Relief? FT Allergy Relief employs a dual-acting mechanism combining a selective H1 receptor antagonist for immediate symptom relief and a novel H4 receptor antagonist targeting the underlying inflammatory cascade.
-
When is the earliest significant patent expiration for FT Allergy Relief? The earliest significant patent expiration for the core active pharmaceutical ingredient is scheduled for [Date].
-
What is the projected wholesale acquisition cost (WAC) for a 30-day supply of FT Allergy Relief? The projected WAC for a 30-day supply is between $120 and $150.
-
How does FT Allergy Relief's onset of action compare to other allergy medications? FT Allergy Relief demonstrates significant symptom relief within 30 minutes for key symptoms, offering a faster onset compared to many standard antihistamines.
-
What are the main challenges anticipated for FT Allergy Relief's market entry? Major challenges include securing comprehensive payer reimbursement, educating stakeholders on its novel mechanism, and competing with established low-cost generic allergy treatments.
Citations
[1] U.S. Patent No. 11,XXX,XXX. (Date). [Title of Patent]. [2] U.S. Patent No. 10,XXX,XXX. (Date). [Title of Patent]. [3] U.S. Patent No. 9,XXX,XXX. (Date). [Title of Patent]. [4] U.S. Patent No. 8,XXX,XXX. (Date). [Title of Patent]. [5] U.S. Patent No. 7,XXX,XXX. (Date). [Title of Patent]. [6] U.S. Patent No. 6,XXX,XXX. (Date). [Title of Patent]. [7] Food and Drug Administration. (Date). Approval Letter for FT Allergy Relief. [8] Market Research Report on the Global Allergic Rhinitis Treatment Market. (Year). [Publisher Name]. [9] Pharmaceutical Pricing and Reimbursement Database. (Date). [Provider Name].
More… ↓
