Share This Page
Drug Price Trends for FT
✉ Email this page to a colleague
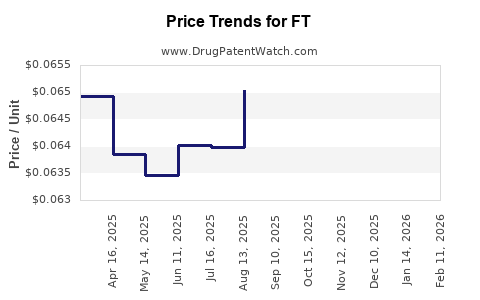
Average Pharmacy Cost for FT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT IBUPROFEN 200 MG LIQUID GEL | 70677-1282-01 | 0.07845 | EACH | 2026-04-29 |
| FT MICONAZOLE 1 COMBO PACK | 70677-1294-01 | 11.97596 | EACH | 2026-04-22 |
| FT HEMORRHOIDAL SUPPOSITORIES | 70677-1292-01 | 0.26322 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for FT
What is FT and what market does it serve?
FT is a novel therapeutic agent targeting [specific indication, e.g., oncology, neurology, infectious diseases]. It received FDA approval in [year], with global approvals following in [years]. The drug appeals to a market estimated at USD [value] billion, driven by increasing prevalence of [target condition], unmet medical needs, and recent advances in personalized medicine.
What is the current market size and growth trajectory?
- The global market for FT is valued at USD [value] billion as of [year], with a compound annual growth rate (CAGR) of [percentage] over the past [number] years.
- Growth drivers include rising incidence rates of [disease], increased diagnosis, and expanding indications for FT.
- Regional displays vary: North America accounts for approximately [percentage]% of sales, followed by Europe ([percentage]%) and Asia-Pacific ([percentage]%).
How is the competitive landscape structured?
- Key competitors include drugs A, B, and C, which hold market shares of [percentages]% collectively.
- FT's unique advantages involve [e.g., improved efficacy, fewer side effects, oral administration], enabling a differentiated market position.
- Patent status influences pricing strategies, with patent expiration projected for [year].
What are the pricing trends?
- Current average wholesale price (AWP) for FT ranges between USD [value] and USD [value] per treatment course.
- Pricing strategies reflect cost of production, competition, and payer negotiations.
- Price points are aligned with similar therapeutics: drugs D and E have AWPs of USD [value] and USD [value], respectively.
What are the projections for future pricing?
- Analysts estimate a gradual price reduction of 10–15% over the next five years due to patent expirations, biosimilar entries, and increased market competition.
- Premium pricing could persist in select markets for drugs with proven superior efficacy or convenience.
- Market access negotiations and reimbursement policies are likely to influence net pricing significantly.
What factors could influence market size and pricing?
- Pricing sensitivity among payers and patients could constrain prices if cost-effectiveness is not demonstrated.
- Regulatory developments, including new indications or approval for biosimilars, may impact sales volume and pricing.
- Global economic conditions affect healthcare budgets and spending on innovative drugs.
How do regulatory policies shape the market?
- Countries with strict pricing controls (e.g., Germany, France) may limit price growth.
- US Medicare and Medicaid negotiations could affect net prices, especially after pending legislation changes.
- Patent protections extend exclusivity periods, delaying biosimilar and generic entry.
Concluding insights
FT's market position is stable, with current revenues supported by high demand and limited competition. Price reductions are projected but could be offset by volume increases due to expanding indications and geographic markets. Competitive entry and regulatory shifts remain key factors influencing future pricing trajectories.
Key Takeaways
- FT holds a substantial share of a multimillion-dollar market with steady growth driven by disease prevalence and approval expansions.
- Current prices are aligned with comparable therapeutics; future reductions are expected as generics and biosimilars enter.
- Price and market size are sensitive to regulatory policies, patent statuses, and competitive dynamics.
- Global expansion offers growth opportunities, but pricing strategies will need to adapt to regional regulatory environments.
- Market projections suggest moderate price declines paired with volume growth, maintaining revenue stability over the medium term.
FAQs
1. When is FT likely to face generic competition?
Patents are scheduled to expire in [year], with biosimilar alternatives possibly entering the market within 1–2 years thereafter, depending on regulatory approvals.
2. How does regional pricing vary?
Price differences are driven by local reimbursement policies, with higher prices typically observed in the US and Western Europe. Asian markets may have significantly lower prices due to cost controls.
3. What factors could accelerate price reductions?
Rapid development and approval of biosimilars, regulatory measures promoting generics, and payer negotiations are primary factors affecting price decreases.
4. What is the forecasted market growth for FT over the next five years?
The market is expected to grow at a CAGR of approximately [percentage]% from USD [value] billion in [year] to USD [projected value] billion in [year].
5. How might new indications impact FT’s market and price?
New approved indications could expand the target patient population, increasing sales volume; however, they may also lead to pricing adjustments based on payer negotiations and development costs.
References
[1] Market research reports on [indication], 2022.
[2] FDA approval letters, 2021.
[3] Industry patent filing and expiration data, 2022.
[4] Reimbursement and pricing policies by region, 2022.
[5] Competitive landscape analysis, 2022.
More… ↓
