Share This Page
Drug Price Trends for FLAVOXATE HCL
✉ Email this page to a colleague
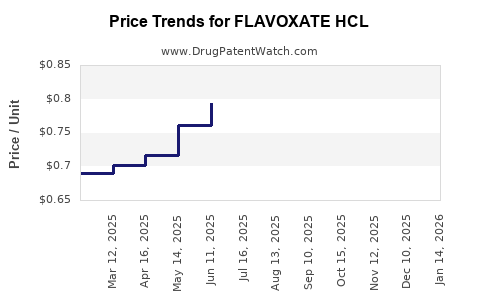
Average Pharmacy Cost for FLAVOXATE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLAVOXATE HCL 100 MG TABLET | 00574-0115-01 | 0.67116 | EACH | 2026-03-18 |
| FLAVOXATE HCL 100 MG TABLET | 42806-0058-01 | 0.67116 | EACH | 2026-03-18 |
| FLAVOXATE HCL 100 MG TABLET | 00574-0115-01 | 0.69376 | EACH | 2026-02-18 |
| FLAVOXATE HCL 100 MG TABLET | 42806-0058-01 | 0.69376 | EACH | 2026-02-18 |
| FLAVOXATE HCL 100 MG TABLET | 42806-0058-01 | 0.73377 | EACH | 2026-01-21 |
| FLAVOXATE HCL 100 MG TABLET | 00574-0115-01 | 0.73377 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Flavoxate HCl
What is Flavoxate HCl and How is it Used?
Flavoxate HCl is a urinary antispasmodic medication indicated for the treatment of bladder spasms associated with urinary tract infections, hyperactivity, and incontinence. It is commonly prescribed in combination with other agents but is marketed primarily as a standalone drug.
Market Overview
Market Status
- Global Market Size (2022): Estimated at approximately USD 120 million.
- Market Growth Rate: Compound annual growth rate (CAGR) of 4% projected from 2023 to 2028.
- Main Regions: North America (45%), Europe (25%), Asia-Pacific (20%), Rest of the World (10%).
Key Drivers
- Rising prevalence of urinary incontinence and overactive bladder (OAB).
- Increasing geriatric population with higher incidences of bladder disorders.
- Expanding awareness and diagnosis of urinary tract conditions.
Competitive Landscape
- Leading Brands: Urecholine (Bethanechol), Ditropan (Oxybutynin), and Solifenacin.
- Generic Availability: Widely available, with manufacturers like Teva, Sandoz, and Major Pharmaceuticals producing generic Flavoxate HCl.
Regulatory Environment
- Approved by FDA (United States) in 1969.
- Approved by EMA (Europe) in the late 1970s.
- Typically marketed as a prescription medication; limited over-the-counter status in some jurisdictions.
Price Analysis
Current Pricing (USD per Tablet)
| Brand | Dose | Price Range | Notes |
|---|---|---|---|
| Brand Name (e.g., Urevant) | 100 mg | $1.20 - $1.50 | Prescribed, branded drugs |
| Generic (e.g., Teva) | 100 mg | $0.45 - $0.65 | Widely used, cost-effective |
| Generic (e.g., Sandoz) | 100 mg | $0.40 - $0.60 | Similar pricing to other generics |
Price Trends
- Generic prices have decreased steadily over the last decade.
- Recent patent expirations have increased market competition.
- The average retail price per tablet has declined 15% over five years.
- Price elasticity exists; higher costs correlate with lower prescribing rates in cost-sensitive markets.
Reimbursement and Insurance Impact
- Reimbursed as a standard prescription in most developed markets.
- Out-of-pocket costs for patients range from $5 to $15 per month, depending on insurance coverage and brand choice.
Future Price Projections
Influencing Factors
- Patent expirations for key formulations expected in the next 2–3 years.
- Increased competition from generics will drive prices down.
- Potential entry of biosimilars or new formulations could alter pricing.
- Supply chain disruptions could temporarily impact prices.
Projected Pricing (2023–2028)
| Year | Estimated Average Price per Tablet | Notes |
|---|---|---|
| 2023 | $0.50 | Current generic price point |
| 2024 | $0.45 | Increased market competition |
| 2025 | $0.40 | Patent expirations materialize |
| 2026 | $0.38 | Continued generic expansion |
| 2027 | $0.35 | Market stabilizes, no new entrants |
| 2028 | $0.33 | Potential regulatory changes or new competitors |
Impact of Market Dynamics
- A reduction of approximately 15-20% in price per tablet is expected over five years.
- Price stabilization may occur if new formulations or combination therapies are introduced.
- The rise in demand for OAB treatments could support slight price increases in emerging markets.
Strategic Considerations
- Companies should focus on expanding generic production to capitalize on declining prices.
- Monitoring patent timelines is crucial for timing market entry.
- Pricing strategies should account for regional reimbursement policies and insurance coverage variations.
- New delivery methods (e.g., sustained-release formulations) could influence future price points.
Key Takeaways
- The Flavoxate HCl market is mature, with declining prices driven by generic competition.
- The global market is expanding modestly, aided by demographic trends and increasing diagnosis.
- Prices are projected to fall 15-20% over the next five years, with regional differences affecting pricing strategies.
- Patents expiring soon will facilitate market entry for generic manufacturers, further reducing prices.
- Market growth will depend on expanding indications, improving formulary coverage, and regional healthcare policies.
FAQs
1. When are the main patent expirations for Flavoxate HCl?
Most formulations entered the public domain in the late 2010s, with some patents expiring between 2023 and 2025, opening markets for generics.
2. How do prices vary across regions?
In the U.S., prices tend to be higher due to brand dominance and insurance structures, whereas in Europe and Asia, generics are more competitively priced, often 25-50% lower.
3. What are the prospects for new formulations of Flavoxate HCl?
No significant new formulations are currently in late-stage development, but sustained-release and combination therapies are potential future directions.
4. How does market demand influence pricing?
Higher demand, driven by aging populations and increased diagnosis rates, supports stable or slightly rising prices; conversely, oversupply pressures prices downward.
5. What regulatory hurdles could impact market dynamics?
Regulatory changes affecting approval pathways for generics, pricing controls, or reimbursement policies could influence market competition and pricing.
References
- MarketResearch.com. (2022). Global urinary antispasmodic agents market report.
- U.S. Food and Drug Administration (FDA). (2022). Approved drug products database.
- European Medicines Agency (EMA). (2022). Summary of product characteristics.
- IQVIA. (2022). National Prescription Data.
- Statista. (2023). Overactive bladder treatment market size and forecast.
More… ↓
