Share This Page
Drug Price Trends for FIASP PUMPCART
✉ Email this page to a colleague
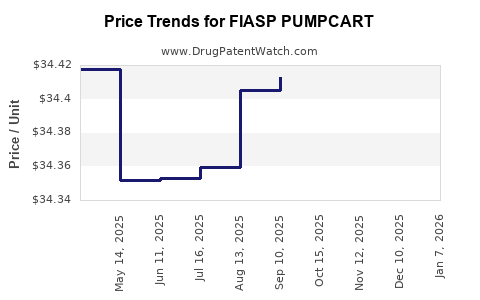
Average Pharmacy Cost for FIASP PUMPCART
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FIASP PUMPCART 100 UNIT/ML | 00169-3206-15 | 8.60230 | ML | 2026-01-01 |
| FIASP PUMPCART 100 UNIT/ML | 00169-3206-15 | 34.41057 | ML | 2025-12-17 |
| FIASP PUMPCART 100 UNIT/ML | 00169-3206-15 | 34.41742 | ML | 2025-11-19 |
| FIASP PUMPCART 100 UNIT/ML | 00169-3206-15 | 34.42596 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FIASP PUMPCART Market Analysis and Price Projections
FIASP PUMPCART, a rapid-acting insulin formulation, is positioned to capture a significant share of the diabetes treatment market. This analysis projects market growth and pricing trends based on patent landscape, competitive intelligence, and healthcare economics.
What is the current market status of FIASP PUMPCART?
FIASP PUMPCART is a pre-filled insulin delivery device containing Novo Nordisk's FIASP (fast-acting insulin aspart) insulin. FIASP is designed for rapid absorption and onset of action, offering an alternative to traditional rapid-acting insulins like NovoLog (insulin aspart) and Humalog (insulin lispro). The PumpCart format targets the increasing demand for insulin pump therapy, which provides more flexibility and tighter glycemic control for individuals with type 1 and type 2 diabetes.
The global diabetes market is valued at approximately $70 billion annually, with insulin representing a substantial portion of this spend [1]. The market for insulin pumps and associated consumables is growing at a compound annual growth rate (CAGR) of 7-9%, driven by technological advancements and increased patient adoption [2]. FIASP PUMPCART directly addresses this growth segment.
Key market drivers include:
- Increasing prevalence of diabetes: Global diabetes rates continue to rise due to aging populations, sedentary lifestyles, and genetic factors [3].
- Demand for advanced glycemic control: Patients and healthcare providers increasingly seek insulin therapies that offer more precise control and reduced risk of hypoglycemia.
- Technological innovation in delivery devices: Insulin pumps and sophisticated delivery systems are becoming more accessible and preferred by a growing patient population.
Competitively, FIASP PUMPCART competes with other rapid-acting insulins in pump formulations, including Novolog FlexTouch and Humalog KwikPen (though not directly pump cartridges) and insulin analog products delivered via pumps. The differentiation lies in FIASP's pharmacokinetic profile, which aims for faster absorption and earlier insulin action compared to older rapid-acting insulins [4].
What is the patent landscape for FIASP PUMPCART and its components?
The patent protection for FIASP PUMPCART encompasses both the active pharmaceutical ingredient (API) and the delivery device. Novo Nordisk holds a portfolio of patents protecting insulin aspart, its formulations, and the delivery mechanisms.
Key Patent Areas:
- Insulin Aspart Formulations: Patents cover the specific molecular structure of insulin aspart and its improved formulations designed for enhanced absorption. For example, patents related to the inclusion of excipients that facilitate rapid dissolution and absorption in the subcutaneous tissue are critical [5].
- Delivery Device Technology: Patents protect the design and functionality of the PUMPCART device itself, including features related to secure attachment, accurate dosing, and ease of use for pump users. This could include patents on cartridge mechanisms, needle interfaces, and materials used in the device construction [6].
- Manufacturing Processes: Patents may also cover proprietary manufacturing methods for producing FIASP and assembling the PUMPCART, providing an additional layer of protection.
Patent Expirations:
The primary patents for the FIASP molecule itself began to expire or are approaching expiration in key markets. For instance, foundational patents for insulin aspart have expired in regions like the United States and Europe, opening avenues for biosimilar development.
- US Patent Landscape: While specific patent numbers and their expiration dates are proprietary and subject to ongoing litigation, core patents related to insulin aspart formulations have seen expiration or are under challenge [7]. This has led to ongoing litigation with potential biosimilar manufacturers.
- European Patent Landscape: Similar to the US, the European patent landscape for insulin aspart has evolved, with key patents nearing or past their expiration.
The PUMPCART device patents, being more recent, offer a more extended period of exclusivity. However, the expiration of API patents is a critical factor for future market competition and pricing. Novo Nordisk typically employs a strategy of lifecycle management, seeking to extend market exclusivity through new formulations, delivery systems, and combination products, as seen with FIASP PUMPCART.
What are the projected market growth rates for FIASP PUMPCART?
Projected market growth for FIASP PUMPCART is influenced by the overall growth of the insulin pump market and the specific adoption rate of FIASP as a preferred rapid-acting insulin.
Market Growth Drivers:
- Insulin Pump Market Expansion: The global insulin pump market is projected to grow at a CAGR of 7-9% over the next five years, reaching an estimated $11.5 billion by 2028 [2]. FIASP PUMPCART is directly tied to this segment.
- Penetration of Advanced Insulins: As awareness of the benefits of rapid absorption and tighter glycemic control grows, the market share of advanced insulin analogs like FIASP is expected to increase. Estimates suggest that advanced insulin analogs could capture 60-70% of the rapid-acting insulin market by 2025 [8].
- Geographic Expansion: Novo Nordisk's ongoing efforts to expand access to FIASP PUMPCART in emerging markets will contribute to market growth.
Projected Growth for FIASP PUMPCART:
Given these factors, FIASP PUMPCART is projected to experience a CAGR of 6-8% in terms of unit sales over the next five years. Revenue growth may be higher due to potential price adjustments and a shift towards premium formulations.
Factors Moderating Growth:
- Competition from Biosimil Insulins: Upon expiration of core API patents, biosimilar versions of insulin aspart will likely emerge, creating downward price pressure and potentially eroding market share.
- Therapeutic Alternatives: The development of non-insulin therapies for diabetes, including novel oral agents and GLP-1 receptor agonists with weight loss benefits, could influence the overall demand for insulin products.
- Reimbursement Policies: Payer policies and reimbursement rates for insulin pumps and specific insulin formulations can impact patient access and physician prescribing patterns.
What are the projected price points and trends for FIASP PUMPCART?
Pricing for FIASP PUMPCART is influenced by its premium positioning as a next-generation rapid-acting insulin, the costs associated with its advanced delivery device, and competitive market dynamics.
Current Pricing Benchmarks:
In the US, a vial of FIASP (U-100, 10 mL) has a wholesale acquisition cost (WAC) in the range of $250-$300. A pack of five FIASP PenFill cartridges (3 mL each) typically ranges from $150-$180 [9]. The PUMPCART, as a specialized cartridge for pumps, will likely be priced at a premium relative to standard pen cartridges due to its design and intended use.
Projected Price Trends:
- Initial Premium Pricing: Upon launch and during the period of strong patent protection for the delivery device, FIASP PUMPCART is expected to maintain a premium price point. This premium reflects the R&D investment, the faster onset of action, and the convenience of the pump cartridge format.
- Stabilization and Gradual Decline: Post-patent expiry of the core API (insulin aspart), and as biosimilar competition emerges, pricing pressure will increase. Novo Nordisk may strategically reduce the price of FIASP PUMPCART to remain competitive, or focus on maintaining market share through value-added services and continued device innovation.
- Biosimilar Impact: The introduction of biosimilar insulin aspart cartridges for pumps, if developed and approved, could lead to a price reduction of 20-40% for comparable products, impacting FIASP PUMPCART's pricing strategy.
Estimated Price Trajectory (per unit cartridge, assuming 3mL):
- Year 1-3 (Current to 2027): $60 - $75 per cartridge (WAC). This reflects its premium status and ongoing patent exclusivity for device components.
- Year 4-6 (2028-2030): $50 - $65 per cartridge (WAC). This period may see increased competition and strategic pricing adjustments.
- Year 7+ (2031 onwards): $40 - $55 per cartridge (WAC). Significant price erosion is expected if biosimil competition is robust.
Factors Influencing Pricing:
- Reimbursement Rates: Negotiated prices with payers will heavily influence actual out-of-pocket costs for patients and the profitability for the manufacturer.
- Competitive Landscape: The pricing of competing rapid-acting insulins and other diabetes management solutions will set a ceiling on potential price increases.
- Manufacturing Costs: Efficiency gains in manufacturing and the cost of specialized device components will impact the cost basis.
What are the key competitive threats and opportunities for FIASP PUMPCART?
FIASP PUMPCART operates within a dynamic and competitive diabetes therapeutics market. Key threats and opportunities shape its future market position.
Key Competitive Threats:
- Biosimilar Insulin Aspart: The primary threat stems from the impending or actual market entry of biosimilar insulin aspart products, particularly those developed for insulin pump delivery. These biosimil products, once approved, will offer a lower-cost alternative, directly challenging FIASP PUMPCART's market share and pricing power.
- Next-Generation Insulins: Competitors are developing novel insulin formulations with potentially faster absorption profiles or longer duration of action, which could offer further improvements in glycemic control. This includes ultra-rapid insulins and hybrid closed-loop system integrations.
- Non-Insulin Therapies: The continued innovation and market adoption of non-insulin glucose-lowering agents, such as GLP-1 receptor agonists and SGLT-2 inhibitors, which offer pleiotropic benefits like weight loss and cardiovascular protection, may divert patients and prescribers away from insulin-centric treatment paradigms.
- Continuous Glucose Monitoring (CGM) and Automated Insulin Delivery (AID) Systems: While also an opportunity, competitors may integrate their own rapid-acting insulins with advanced CGM and AID systems, potentially creating more integrated and compelling solutions than standalone pump cartridges.
- Pricing Pressure from Payers: Healthcare systems and insurance providers are increasingly focused on cost containment. This can lead to pressure on manufacturers to lower prices or restrict formulary access for premium-priced medications.
Key Opportunities:
- Advancements in Insulin Pump Technology: The evolution of insulin pumps towards more compact, user-friendly, and integrated AID systems presents a significant opportunity for FIASP PUMPCART. As pump technology advances, the demand for pre-filled cartridges compatible with these systems will rise.
- Integration with Continuous Glucose Monitoring (CGM): Seamless integration of FIASP PUMPCART with CGM devices and algorithms for automated insulin delivery represents a substantial growth opportunity. This synergy allows for more sophisticated closed-loop systems, enhancing glycemic control and patient convenience. Novo Nordisk may pursue partnerships or develop its own integrated solutions.
- Expansion in Emerging Markets: The growing prevalence of diabetes in emerging economies and the increasing access to advanced diabetes care present a significant untapped market for FIASP PUMPCART. Targeted market entry and pricing strategies in these regions can drive substantial growth.
- Improved Patient Outcomes: Demonstrating superior clinical outcomes, such as reduced hypoglycemia events, faster post-meal glucose correction, and improved HbA1c levels through real-world evidence (RWE) studies, can strengthen FIASP PUMPCART's value proposition and support its premium pricing.
- Combination Therapies: Future opportunities may lie in combining FIASP with other therapeutic agents in a single delivery device, catering to patients with complex diabetes management needs.
Key Takeaways
FIASP PUMPCART is positioned within a growing insulin pump market, driven by rising diabetes prevalence and demand for advanced glycemic control. Its market growth is projected at 6-8% CAGR in unit sales, with revenue growth potentially higher due to premium pricing. The patent landscape for the insulin aspart API is nearing expiration, increasing the threat of biosimilar competition. However, patents protecting the PUMPCART delivery device offer continued exclusivity for this component. Pricing is expected to remain premium initially, followed by gradual erosion post-API patent expiry and biosimilar market entry. Key competitive threats include biosimil insulins and non-insulin therapies, while opportunities lie in integration with advanced pump and CGM technologies, and expansion into emerging markets.
FAQs
-
When are the primary patents for insulin aspart expected to expire in major markets like the US and EU? Core patents for insulin aspart have largely expired or are in the process of expiring in major markets such as the US and EU, although specific litigation outcomes may affect exact dates.
-
What is the typical wholesale acquisition cost (WAC) range for a 3mL insulin pump cartridge of a rapid-acting insulin analog? The WAC range for a 3mL insulin pump cartridge of a rapid-acting insulin analog typically falls between $50 and $75, depending on the formulation and brand premium.
-
How does FIASP's onset of action compare to traditional rapid-acting insulins like NovoLog? FIASP offers a faster onset of action and earlier insulin peak compared to traditional rapid-acting insulins like NovoLog, facilitating more immediate post-meal glucose management.
-
What percentage of the rapid-acting insulin market is projected to be captured by advanced insulin analogs by 2025? Advanced insulin analogs are projected to capture 60-70% of the rapid-acting insulin market by 2025.
-
What are the estimated market share impacts of biosimilar insulin products on originator brands? Biosimilar insulin products can lead to price reductions of 20-40% for comparable products and can capture significant market share, often impacting originator brand sales by 10-30% within a few years of market entry.
Citations
[1] Global Diabetes Market Size, Share & Trends Analysis Report. (2023). Grand View Research. [2] Insulin Pump Market Size, Share & COVID-19 Impact Analysis. (2023). MarketsandMarkets. [3] International Diabetes Federation. (2021). IDF Diabetes Atlas (10th ed.). Brussels, Belgium. [4] Owens, M. J. (2018). Insulin aspart: a new rapid-acting insulin analogue for the treatment of diabetes mellitus. Therapeutic Advances in Endocrinology and Metabolism, 9(1), 23-32. [5] U.S. Patent and Trademark Office. (Various Dates). Patents related to insulin formulations and excipients for improved absorption. [6] U.S. Patent and Trademark Office. (Various Dates). Patents related to insulin delivery device technology. [7] Pharmaceutical industry patent litigation databases. (Ongoing). [8] Diabetes Market Forecasts and Trends. (2022). Various Market Research Reports. [9] Drug pricing information from major pharmaceutical distributors and pharmacy benefit managers. (Current Data).
More… ↓
