Share This Page
Drug Price Trends for EYE ITCH RELIEF
✉ Email this page to a colleague
Average Pharmacy Cost for EYE ITCH RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
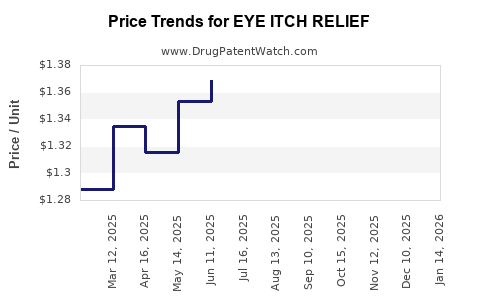
| EYE ITCH RELIEF 0.025% DROPS | 00536-1252-40 | 1.34389 | ML | 2026-03-18 |
| EYE ITCH RELIEF 0.025% DROPS | 70000-0522-01 | 1.34389 | ML | 2026-03-18 |
| EYE ITCH RELIEF 0.025% DROPS | 00536-1252-40 | 1.36999 | ML | 2026-02-18 |
| EYE ITCH RELIEF 0.025% DROPS | 70000-0522-01 | 1.36999 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EYE ITCH RELIEF Market Analysis and Financial Projection
What is Eye Itch Relief?
Eye Itch Relief is a topical medication aimed at alleviating symptoms associated with ocular allergies, such as itching, redness, and irritation. The product typically contains antihistamines or mast cell stabilizers, with popular formulations including ketotifen or olopatadine. These drugs are approved for over-the-counter (OTC) use in many regions and are available in various forms, like eye drops and gels.
What is the Current Market Size and Sales of Eye Itch Relief?
The global market for ophthalmic antihistamines and allergy eye drops, including Eye Itch Relief products, was valued at approximately $2.4 billion in 2022. The market has experienced compound annual growth rates (CAGR) of 4% from 2018 to 2022.
Key regional markets include:
- North America: $1.2 billion (approx. 50% of global sales)
- Europe: $600 million (25%)
- Asia-Pacific: $400 million (16%)
- Rest of World: $200 million (9%)
Leading brands include Zaditor (kiotifen), Alaway (ketotifen), and Visine-A (pheniramine + naphazoline). OTC availability, combination formulations, and increased allergy prevalence drive growth.
What Are Regulatory and Patent Considerations for Eye Itch Relief?
Many formulations are off-patent, allowing generic manufacturers to enter the market, exerting pricing pressure. Branded products typically hold patents for formulation-specific delivery mechanisms or combination therapies, valid until 2025-2030.
The U.S. Food and Drug Administration (FDA) approves OTC versions if they meet efficacy and safety standards. In Europe, the European Medicines Agency (EMA) follows similar procedures.
Patent expirations have led to increased generic competition, driving prices downward. Protecting innovation through formulation patents or delivery mechanisms remains vital.
What Are Price Trends and Projections for Eye Itch Relief?
Current retail prices (average retail prices as of Q1 2023):
| Product Type | Price (per 15 mL bottle) | Key Brands |
|---|---|---|
| Branded OTC | $20 - $30 | Zaditor, Alaway |
| Generic OTC | $8 - $14 | Multiple generics |
| Prescription | $50 - $100 (if prescribed) | Rare, focused on severe cases |
Price trajectory expectations:
-
Short-term (next 1-2 years): Prices to remain stable or slightly decrease due to increased generic competition.
-
Medium-term (3-5 years): Prices may decline by 10-20%, driven by patent expirations and market saturation.
-
Long-term (beyond 5 years): Innovation in sustained-release formulations or combination drugs could command premium pricing, but widespread generic entry will likely lower average prices.
What Factors Influence Market and Price Changes?
- Patent expirations and generics: Expiration of patents on key formulations reduces barriers for generics, decreasing consumer prices.
- Regulatory environment: Easier approval pathways for OTC formulations expedite market entry, increasing competition.
- Product innovation: New delivery systems or combination therapies can justify higher prices and expand market share.
- Consumer awareness: Growing awareness from increased allergy diagnoses expands patient base.
- Distribution channels: Expansion into pharmacy chains, online sales, and international markets influences pricing strategies.
What Are Key Opportunities and Risks in the Market?
Opportunities:
- Development of long-acting formulations reducing dosing frequency.
- Entry into emerging markets with rising allergy prevalence.
- Innovation in preservative-free or preservative-limited drops to appeal to sensitive users.
Risks:
- Price erosion due to patent expirations.
- Regulatory hurdles for new formulations.
- Market saturation in mature markets.
Key Takeaways
- The Eye Itch Relief market is steady at approximately $2.4 billion globally, with North America dominating.
- Patent expirations are creating cost pressures, leading to lower prices and increased generic competition.
- Current retail prices for OTC products range between $8 and $30 per bottle, with further declines expected as generics proliferate.
- Innovation around delivery mechanisms and formulations could command higher prices but face regulatory and development challenges.
- Market growth will be influenced by allergy prevalence, regulatory pathways, and consumer preferences for preservative-free options.
FAQs
1. How does patent expiration impact the price of Eye Itch Relief products? Patent expiration allows generics to enter the market, increasing competition and typically lowering prices.
2. What are the main ingredients in Eye Itch Relief products? Antihistamines such as ketotifen, olopatadine, and pheniramine are common active ingredients.
3. What are the key regions for growth in this market? North America and Asia-Pacific exhibit the highest growth potential due to allergy prevalence and market expansion.
4. Is there potential for new formulations to command higher prices? Yes, sustained-release or preservative-free formulations can justify premium pricing, particularly if they offer improved convenience or tolerability.
5. What factors could hinder the growth of Eye Itch Relief products? Regulatory hurdles, market saturation, and the availability of effective generics could limit growth.
References
[1] MarketWatch, "Global Ophthalmic Allergy Market," 2022.
[2] IQVIA, "Ocular Allergy Market Data," 2023.
[3] FDA, "OTC Eye Drop Drug Approvals," 2022.
[4] European Medicines Agency, "Ocular Allergy Medications," 2022.
More… ↓
