Drug Price Trends for ENALAPRIL
✉ Email this page to a colleague
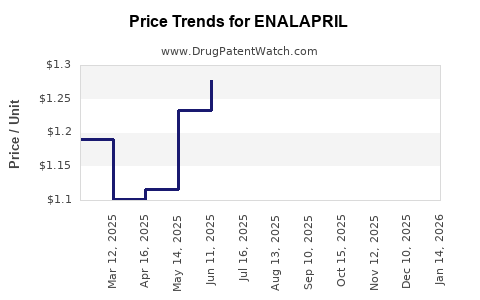
Average Pharmacy Cost for ENALAPRIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ENALAPRIL 1 MG/ML ORAL SOLN | 59651-0529-55 | 0.95754 | ML | 2026-05-20 |
| ENALAPRIL 1 MG/ML ORAL SOLN | 52536-0401-01 | 0.95754 | ML | 2026-05-20 |
| ENALAPRIL-HYDROCHLOROTHIAZIDE 5-12.5 MG TAB | 51672-4045-01 | 0.18641 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ENALAPRIL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ENALAPRIL MALEATE 2.5MG TAB | Golden State Medical Supply, Inc. | 60429-0183-10 | 1000 | 44.30 | 0.04430 | EACH | 2024-04-08 - 2028-06-14 | FSS |
| ENALAPRIL MALEATE 20MG TAB | Golden State Medical Supply, Inc. | 51672-4040-01 | 100 | 12.99 | 0.12990 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| ENALAPRIL MALEATE 20MG TAB | Golden State Medical Supply, Inc. | 60429-0186-90 | 90 | 10.99 | 0.12211 | EACH | 2024-04-08 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
