Share This Page
Drug Price Trends for DOXYCYCLINE HYC DR
✉ Email this page to a colleague
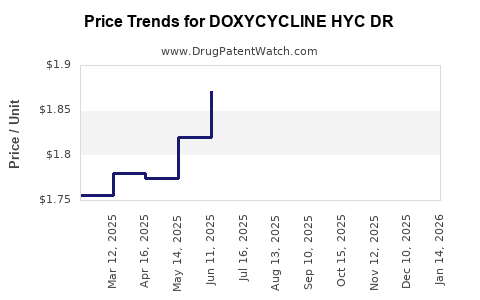
Average Pharmacy Cost for DOXYCYCLINE HYC DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DOXYCYCLINE HYC DR 50 MG TAB | 43547-0462-12 | 1.56942 | EACH | 2026-04-22 |
| DOXYCYCLINE HYC DR 100 MG TAB | 00228-2896-11 | 2.30238 | EACH | 2026-04-22 |
| DOXYCYCLINE HYC DR 100 MG TAB | 23155-0142-01 | 2.30238 | EACH | 2026-04-22 |
| DOXYCYCLINE HYC DR 200 MG TAB | 00591-4575-60 | 3.55412 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DOXYCYCLINE HYC DR Market Analysis and Financial Projection
What Is the Market Status of Doxycycline Hyclate DR?
Doxycycline Hyclate DR (delayed-release) is a commonly prescribed broad-spectrum tetracycline antibiotic used for bacterial infections. Its market involves both branded formulations and generics, with significant presence in various therapeutic areas such as respiratory tract infections, sexually transmitted infections, and acne.
Market Size and Demand
As of 2022, the global doxycycline market was valued at approximately USD 340 million, with antibiotics representing a substantial share. The demand for doxycycline Hyclate DR is driven by:
- Rising prevalence of infections
- Antibiotic resistance considerations favoring older antibiotics
- Established prescribing patterns for dermatological and respiratory conditions
North America and Europe account for over 50% of the market share, attributed to high prescription rates and mature generic markets. The Asia-Pacific region displays growth potential due to expanding healthcare infrastructure.
Competitive Landscape
The market features several key players:
| Company | Product Name | Segment | Market Share (%) | Registration Status |
|---|---|---|---|---|
| Pfizer | Vibramycin (brand) | Branded | ~25 | Marketed globally |
| Teva Pharmaceuticals | Doxycycline Hyclate DR | Generic | ~15 | Approved in multiple markets |
| Mylan | Doxycycline Hyclate | Generic | ~12 | Approved globally |
| Sandoz | Doxycycline Hyclate | Generic | ~10 | Active in key regions |
The branded market holds approximately 40%,but generics dominate, pressuring prices downward. Entry barriers include regulatory hurdles and manufacturing costs.
Regulatory Environment and Approvals
Regulatory agencies such as FDA (USA), EMA (Europe), and PMDA (Japan) approve doxycycline formulations, with approval processes generally requiring demonstration of bioequivalence for generics. No recent significant regulatory changes impacting doxycycline Hyclate DR have been reported.
What Are the Price Trends and Projections?
Historical Price Data
In the US, average wholesale prices (AWP) for doxycycline Hyclate DR have declined over the past five years:
| Year | Average Wholesale Price (USD) per 100 capsules | Change from Previous Year (%) |
|---|---|---|
| 2018 | 10.50 | — |
| 2019 | 8.75 | -16.67 |
| 2020 | 7.20 | -17.71 |
| 2021 | 6.40 | -11.11 |
| 2022 | 5.85 | -8.59 |
This decline reflects increased competition, price compression from generics, and procurement efficiencies.
Future Price Projections
By 2025, prices are expected to stabilize around USD 5.50–6.00 per 100 capsules, assuming continued generic saturation and no new formulations disrupting the market. The compound annual growth rate (CAGR) for prices from 2022 to 2025 is projected to be approximately -2.7%.
Factors Impacting Price Movements
- Patent expiration timelines: The original patent expired around 2007 for market protection, but formulations evolved with delayed-release versions reducing direct generic competition temporarily.
- Supply chain factors: Disruptions due to raw material shortages or manufacturing shifts can impact pricing.
- Regulatory policies: Pricing regulations or drug import/export policies in key markets may influence costs.
What Are the Key Considerations for Investment and R&D?
Expanding indications or developing combination therapies could open new markets. However, the current saturation by generics constrains profit margins. Innovative delivery mechanisms or formulations might command a premium but require significant R&D investment.
Key Takeaways
- The doxycycline Hyclate DR market is mature with declining prices due to broad generic competition.
- The global market was valued at roughly USD 340 million in 2022.
- Price projections suggest a continued slight decline through 2025, stabilizing near USD 5.50–6.00 per 100 capsules.
- Regulatory approval remains robust across regions; patent protections are limited.
- Growth opportunities hinge on new indications, combination therapies, or formulations that address resistant strains.
FAQs
1. Is Doxycycline Hyclate DR still under patent protection?
No. The original patent expired around 2007; current formulations are protected only through new patents linked to specific formulations or delivery systems.
2. How does the competition affect pricing?
Generic competition has driven prices downward, making it challenging for branded versions to maintain premium pricing.
3. Are there any upcoming regulatory changes impacting pricing or approval?
No significant regulatory shifts are currently anticipated for doxycycline Hyclate DR in major markets.
4. Which regions are most lucrative for doxycycline Hyclate DR?
North America and Europe dominate current sales, but growth potential exists in Asia-Pacific, especially in emerging markets.
5. Can new formulations or combinations improve profit margins?
Potentially, but they require significant R&D investment and must justify premium pricing through improved efficacy or resistance management.
References
[1] MarketWatch, "Doxycycline Antibiotics Market Size, Share & Trends," 2022.
[2] IQVIA, "Global Antibiotics Market Data," 2022.
[3] FDA, "Guidance for Industry: Bioequivalence Evaluation," 2019.
More… ↓
