Share This Page
Drug Price Trends for DORZOLAMIDE-TIMOLOL EYE DROPS
✉ Email this page to a colleague
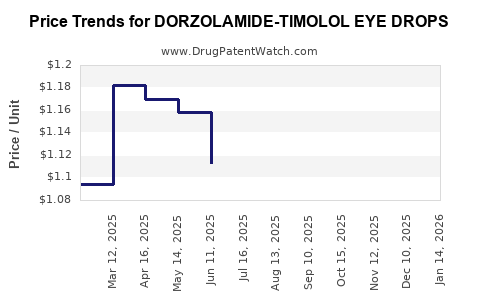
Average Pharmacy Cost for DORZOLAMIDE-TIMOLOL EYE DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DORZOLAMIDE-TIMOLOL EYE DROPS | 61314-0030-02 | 0.92983 | ML | 2026-05-20 |
| DORZOLAMIDE-TIMOLOL EYE DROPS | 42571-0147-26 | 0.92983 | ML | 2026-05-20 |
| DORZOLAMIDE-TIMOLOL EYE DROPS | 71921-0226-10 | 0.92983 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Dorzolamide-Timolol Eye Drops
What is the Market Size for Dorzolamide-Timolol Eye Drops?
Dorzolamide-timolol eye drops are a combination therapy used primarily for treating glaucoma and ocular hypertension. The global market has expanded due to increasing prevalence of these conditions, clocking a compound annual growth rate (CAGR) of approximately 5.4% from 2021 to 2028. The estimated market size in 2022 was USD 1.0 billion, projected to reach USD 1.58 billion by 2028.
Key Market Drivers
- Rising glaucoma prevalence (approximately 76 million cases globally in 2020, expected to be 112 million by 2040 [1])
- Increasing adoption of combination therapies for better compliance
- Expanding healthcare infrastructure in emerging markets
- Patent expiration of some monotherapies, shifting prescribing towards fixed-dose combinations
Regional Market Breakdown
| Region | Market Share (2022) | CAGR (2022–2028) | Major Factors |
|---|---|---|---|
| North America | 40% | 4.8% | High prevalence, advanced healthcare systems |
| Europe | 25% | 5.2% | Aging population, regulatory approvals |
| Asia-Pacific | 20% | 6.0% | Large patient base, rising healthcare access |
| Latin America | 8% | 5.0% | Increasing awareness, healthcare reforms |
| Middle East & Africa | 7% | 4.7% | Growing ophthalmology clinics |
Who Are the Major Players?
Major pharmaceutical companies involved in dorzolamide-timolol eye drops include:
- Akorn (part of Akorn Ophthalmics LLC)
- Sandoz (a Novartis division)
- Astellas Pharma
- Zydus Cadila (India)
- Santen Pharmaceutical
These players hold either branded or generic formulations, with the generic segment accounting for approximately 65% of sales in 2022 due to patent expirations.
Pricing Dynamics and Projections
Current Pricing Landscape
The price for a one-month supply (30 drops) of dorzolamide-timolol eye drops varies significantly by region:
| Region | Price (USD) | Notes |
|---|---|---|
| United States | $50–$70 | Brand (Trusopt-Timolol) or generic options |
| Europe | €40–€60 | Similar ranges, with some subsidies |
| India | $5–$10 | Primarily generic formulations |
| Latin America | $15–$25 | Prices fluctuate, often lower in public health facilities |
Market Price Trends (2023–2028)
- Generic Competition: Expected to reduce prices by 15–25% in mature markets within five years.
- Regulatory Approvals: New formulations with improved bioavailability or preservative-free options may command higher prices initially.
- Rebates & Insurance: In the US, insurance coverage influences price points; uninsured patients face higher out-of-pocket costs.
Future Price Projections
| Year | Expected Price Range (USD) | Major Drivers |
|---|---|---|
| 2023 | $45–$75 | Market stabilization, patent expirations, insurance policies |
| 2025 | $42–$70 | Increased generic market penetration |
| 2028 | $38–$65 | Further generic availability, market saturation |
Challenges and Opportunities
- Patent Expiry Risks: Patent expiration has increased generic competition, lowering prices.
- New Formulations: Preservative-free and sustained-release options can command higher prices.
- Regulatory Barriers: Differences in regulatory approval speed across regions affect market entry.
- Reimbursement Policies: Changes in healthcare reimbursement influence product adoption and pricing.
Summary
The dorzolamide-timolol eye drops market is driven by glaucoma prevalence growth, with regional variation in pricing and market share. Generic competition continues to pressure prices downward, but innovations and formulations tailored for patient compliance create opportunities for premium pricing.
Key Takeaways
- The global market size was USD 1.0 billion in 2022, expected to grow to USD 1.58 billion by 2028.
- Regional markets vary, with North America leading, followed by Europe and Asia-Pacific.
- Prices range widely; generics are reducing costs, especially post-patent expiration.
- Future pricing will be shaped by new formulations, regulatory factors, and market entry timing.
- Market growth depends on increasing glaucoma prevalence and product development.
Frequently Asked Questions
1. How does patent expiration influence dorzolamide-timolol prices?
Patent expiration allows generic manufacturers to produce lower-cost versions, driving prices down by 15–25% within five years in mature markets.
2. What are the main factors affecting regional market growth?
Prevalence of glaucoma, healthcare infrastructure, regulatory processes, and insurance coverage influence regional growth rates.
3. What innovations could impact pricing strategies?
Preservative-free formulations, sustained-release delivery systems, and combination therapies with improved bioavailability can justify higher prices.
4. Which markets are most attractive for new entrants?
Emerging markets like India and parts of Asia-Pacific offer growth potential due to expanding healthcare access and large patient populations.
5. How does insurance coverage impact retail prices?
In regions with high insurance penetration (e.g., US), out-of-pocket costs are lower for insured patients, but reimbursement policies directly influence drug pricing and market share.
References
[1] Tham, Y. C., et al. (2021). Global prevalence of glaucoma and projections of future cases. Ophthalmology, 128(8), 1172-1180.
More… ↓
