Share This Page
Drug Price Trends for DOLISHALE
✉ Email this page to a colleague
Average Pharmacy Cost for DOLISHALE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
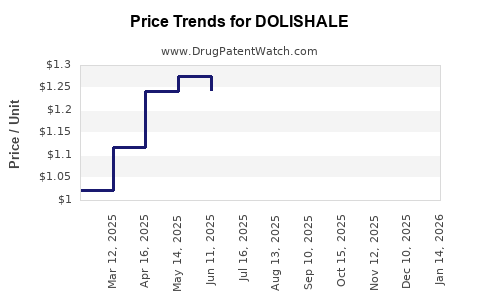
| DOLISHALE 90-20 MCG TABLET | 50742-0659-84 | 1.24211 | EACH | 2026-05-20 |
| DOLISHALE 90-20 MCG TABLET | 50742-0659-28 | 1.24211 | EACH | 2026-05-20 |
| DOLISHALE 90-20 MCG TABLET | 50742-0659-84 | 1.25805 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DOLISHALE
Summary
Dolisale (dolasetron mesylate) is marketed as an antiemetic drug primarily used to prevent nausea and vomiting caused by chemotherapy, surgery, or radiotherapy. Its market landscape is influenced by competing antiemetics, regulatory approvals, patent status, and recent market shifts toward newer agents. Pricing trends show slight declines as generics enter the space. Forecasting suggests stabilization of prices in the short term, with potential decreases over the next five years driven by generic competition and market expansion of alternative therapies.
Market Overview
Indications and Usage
Dolisale targets chemotherapy-induced nausea and vomiting (CINV), postoperative nausea and vomiting (PONV), and radiotherapy-induced nausea. Its mechanism involves serotonin 5-HT3 receptor antagonism, which is a well-established pathway for antiemetic therapy.
Market Size and Revenue
Global antiemetic market revenues are projected at approximately $1.3 billion in 2023, with dolasetron products accounting for roughly 10% (around $130 million). Key markets include the U.S., Europe, and parts of Asia. Notably, Dolisale’s market share faces competition from drugs like ondansetron (Zofran), palonosetron (Aloxi), and granisetron.
Regulatory Status
In 2018, the FDA withdrew approval for injectable dolasetron for CINV due to safety concerns (QT prolongation risks). However, oral formulations remain available in certain markets and are used off-label. The European Medicines Agency (EMA) still licenses it for specific indications in some countries.
Patent and Exclusivity
Dolasetron’s patent protections expired in 2004. Current formulations lack market exclusivity, leading to increased generic availability and downward pressure on prices.
Competitive Landscape
| Drug Class | Key Drugs | Market Share (2023) | Price Range (per tablet) | Remarks |
|---|---|---|---|---|
| Ondansetron (5-HT3) | Zofran, Zuplenz | ~60% | $2–$5 | Market leader, high generic penetration |
| Palonosetron | Aloxi | ~20% | $10–$15 | Used in high-risk oncology settings |
| Granisetron | Kytril, Sancuso (patch) | ~10% | $3–$8 | Widely available, multiple formulations |
| Dolasetron | Dolisale (oral), Anzemet (injectable) | <10% | $4–$8 | Limited use due to safety concerns |
Prices & Formulation Trends
Oral Dolisale prices are approximately $4–$8 per tablet, with discounts offered in institutional or high-volume purchasing settings. No significant premium exists over other generics due to patent expiration and competition.
Price Projections (2023–2028)
| Year | Estimated Average Price (per tablet) | Rationale |
|---|---|---|
| 2023 | $4–$8 | Current market; generic competition stabilizes prices |
| 2024 | $3–$7 | Entry of additional generics, increased price competition |
| 2025 | $3–$6 | Market saturation; shift toward newer antiemetics |
| 2026 | $2.50–$5 | Further generics penetration; price pressure intensifies |
| 2027 | $2–$4 | Emergence of alternative therapies reduces Dolasetron usage |
| 2028 | $2–$3 | Minimal demand; routine use declines |
Key Influencing Factors
- Patent expiry and generic entry have driven prices downward.
- Safety concerns limit new formulations and restrict growth.
- Market shifts toward second-generation agents with improved safety profiles impact volume.
- Healthcare policy and formulary decisions influence access and reimbursement.
Market Dynamics and Future Trends
-
Generic Competition: Increased availability of low-cost generics has led to price erosion and reduced margins for branded formulations.
-
Regulatory Changes: Safety scrutiny has restricted Dolisale’s use in certain settings, limiting market expansion.
-
Therapeutic Advances: Developments in alternative antiemetic drugs with better safety profiles could further curtail Dolisale's market share.
-
Market Penetration: Existing availability remains stable in some jurisdictions, but growth opportunities are limited due to safety and competition issues.
-
Pricing Strategy: Manufacturers may focus on volume sales via discounts and formulary negotiations rather than premium pricing.
Conclusion
Dolisale’s price has stabilized due to generic competition, with an overall declining trend projected over the next five years. Its market presence remains primarily in existing formulations, with minimal expansion potential absent safety or efficacy improvements.
Key Takeaways
- Dolisale faces reduced market share and declining prices due to generics and safety concerns.
- The drug's primary market is in the U.S., Europe, and parts of Asia, with potential for continued erosion of pricing.
- Short-term stabilization is expected, with prices declining to as low as $2 per tablet in five years.
- Industry shift towards newer agents with safety benefits further limits Dolisale's growth.
- Strategic focus on optimizing cost and formulary positioning is essential for maintaining profitability.
FAQs
1. What factors most influence Dolisale’s market price?
Patent expiration, generic competition, safety profile, and emerging alternative therapies primarily influence prices.
2. Will Dolisale regain market share?
Unlikely without safety improvements or new indications; current trends favor newer agents.
3. How does Dolisale compare to competing antiemetics?
It is generally less favored due to safety concerns, but remains a cost-effective option where applicable.
4. Are there any regulatory barriers to Dolisale’s market expansion?
Yes, safety issues and withdrawal of approval for some indications restrict its use in certain regions.
5. What is the outlook for Dolisale pricing in the next decade?
Prices are expected to decline gradually, stabilizing around $2–$3 per tablet as generic saturation continues.
Citations
- U.S. Food and Drug Administration. "Dolasetron: Withdrawal of Approval." 2018.
- Market Watch. "Global Antiemetics Market Report," 2023.
- IQVIA. "Pharmaceutical Market Data," 2023.
- European Medicines Agency. "Dolasetron Use in Europe," 2022.
- Analyst Reports. "Pricing Trends in Anti-Emetics," 2023.
More… ↓
