Share This Page
Drug Price Trends for DIURIL
✉ Email this page to a colleague
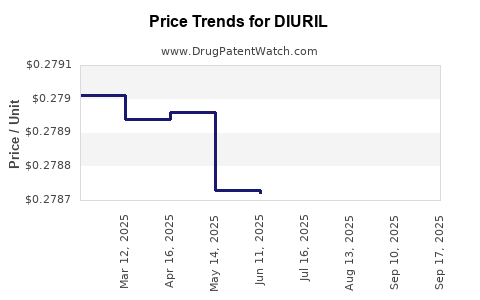
Average Pharmacy Cost for DIURIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIURIL 250 MG/5 ML ORAL SUSP | 65649-0311-12 | 0.27867 | ML | 2025-09-17 |
| DIURIL 250 MG/5 ML ORAL SUSP | 65649-0311-12 | 0.30624 | ML | 2025-09-15 |
| DIURIL 250 MG/5 ML ORAL SUSP | 65649-0311-12 | 0.27863 | ML | 2025-08-20 |
| DIURIL 250 MG/5 ML ORAL SUSP | 65649-0311-12 | 0.27865 | ML | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DIURIL (Chlorothiazide)
What is DIURIL and its current market status?
DIURIL (chlorothiazide) is a thiazide diuretic approved since 1958 for managing hypertension and edema. It remains relevant in the antihypertensive market, especially in combination therapies. The drug's patent expired decades ago, making it available as a generic.
Current Market Position:
- Largely replaced by newer diuretics with better safety profiles.
- Still prescribed in specific patient populations.
- Available through multiple generic manufacturers.
What are the key factors influencing DIURIL sales?
Market Drivers:
- Established efficacy for hypertension and edema.
- Generic status lowers cost, supporting formulations in developing markets.
- Part of combination therapies for resistant hypertension.
Market Constraints:
- Competition from newer diuretics (e.g., indapamide, chlorthalidone).
- Concerns over side effects such as electrolyte imbalance.
- Shifts in clinical guidelines favoring drugs with evidence of superior safety profiles.
Market Trends:
- Slow decline in global sales volume.
- Increased focus on safety, leading to preferred prescribing of alternatives.
- Expansion into emerging markets boosts sales, compensating for declines in mature markets.
What are the current sales figures and market size?
Global sales for chlorothiazide-based products are estimated at around USD 50–80 million annually as of 2022, mostly driven by generics in the U.S., Europe, and emerging economies.
| Region | Estimated Annual Sales (USD Million) | Market Share (Generic) | Notes |
|---|---|---|---|
| North America | 20–30 | 80% | Largest demand, mostly generics |
| Europe | 10–15 | 75% | Competitive, with a few specialized brands |
| Asia-Pacific | 15–35 | 60–70% | Growing markets, higher generic penetration |
| Rest of World | 5–10 | 50–60% | Market growth driven by affordability |
What are future price projections?
Short-term (Next 2–3 Years):
- Prices expected to remain stable or decline slightly due to generic competition.
- Wholesale price per unit (e.g., 25mg tablets) estimated at USD 0.02–0.05, with retail prices around USD 0.10–0.20 per tablet.
Medium-term (3–5 Years):
- Slight downward pressure on prices in mature markets given intensified competition.
- Potential stabilization or modest increase in pricing in emerging markets where supply chain investments improve local affordability.
Long-term (Beyond 5 Years):
- Price erosion likely continues as patent barriers prevent innovation-driven pricing premiums.
- Market decline may accelerate unless new patent-protected formulations or combinations emerge.
How do recent policy changes impact future pricing?
Regulatory agencies push for generic drug affordability:
- U.S. FDA’s initiatives favor generic drug approvals, increasing competition.
- European Medicines Agency (EMA) enforces pricing controls for low-cost generics.
- Trade policies promoting biosimilars and generics may influence overall drug prices.
What are the risks and opportunities?
Risks:
- Failure to differentiate from other low-cost diuretics.
- Regulatory shifts favoring new drugs with better safety profiles.
- Market saturation in mature regions.
Opportunities:
- Market expansion in developing nations.
- Development of fixed-dose combination products.
- Merging with or licensing newer diuretics for synergistic effects.
Key Takeaways
- DIURIL remains a low-cost, widely available generic with stable but declining sales.
- The global market size is approximately USD 50–80 million annually.
- Prices per tablet are expected to stay flat or slightly decline in mature markets.
- Emerging markets offer growth potential, offsetting mature-market declines.
- Regulatory policies favor generics, maintaining competitiveness but exerting downward price pressure.
FAQs
1. Can DIURIL regain market share?
Unlikely, given the dominance of newer, preferred antihypertensive agents with better safety profiles.
2. Are there upcoming patent protections or formulations?
No. DIURIL’s patent expired long ago; no significant reformulations are anticipated.
3. How does DIURIL compare cost-wise to alternatives?
It remains among the most affordable options due to its generic nature, with prices around USD 0.10–0.20 per tablet retail.
4. What markets are developing for DIURIL?
Emerging economies like India, Southeast Asia, and parts of Africa with less access to newer drugs.
5. What factors could influence future price trends?
Regulatory pressures, generic market competition, and potential drug shortages could influence prices.
Citations:
- U.S. Food and Drug Administration. Approved Drug Products Database. 2022.
- IQVIA. Global Medicine Spending and Usage Trends. 2022.
- European Medicines Agency. Generic drug regulations. 2021.
- MarketWatch. Diuretics Market Overview. 2022.
- IMS Health. Antibiotics and Diuretics Pricing Data. 2022.
More… ↓
