Share This Page
Drug Price Trends for DICLOFENAC SOD DR
✉ Email this page to a colleague
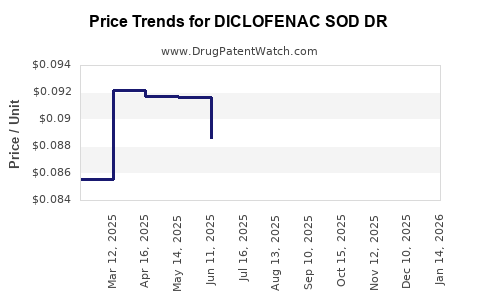
Average Pharmacy Cost for DICLOFENAC SOD DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DICLOFENAC SOD DR 25 MG TAB | 72888-0109-01 | 0.72051 | EACH | 2026-05-20 |
| DICLOFENAC SOD DR 25 MG TAB | 72603-0600-01 | 0.72051 | EACH | 2026-05-20 |
| DICLOFENAC SOD DR 75 MG TAB | 72888-0111-60 | 0.06665 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Diclofenac Sod Dr
What is Diclofenac Sod Dr?
Diclofenac Sod Dr (Diclofenac Sodium Extended-Release) is a non-steroidal anti-inflammatory drug (NSAID) used primarily for managing chronic pain related to arthritis, osteoarthritis, and other inflammatory conditions. The "Dr" indicates it is a delayed-release formulation designed for once-daily dosing, improving patient compliance compared to immediate-release versions.
Current Market Dynamics
Global Market Size and Growth
The global NSAID market was valued at approximately USD 9.6 billion in 2021 and is projected to reach USD 14.8 billion by 2028, expanding at a compound annual growth rate (CAGR) of 6.2% (ResearchAndMarkets, 2022). Diclofenac compounds embody the largest share within NSAID sales due to established efficacy and widespread use.
Key Regional Markets
| Region | Market Size (2022) | CAGR (2022-2028) | Dominant Players |
|---|---|---|---|
| North America | USD 3.1 billion | 5.8% | Novartis, Mylan, Teva |
| Europe | USD 2.8 billion | 6.1% | Novartis, Sandoz, Pfizer |
| Asia-Pacific | USD 1.9 billion | 7.2% | Cipla, Takeda, Sun Pharma |
| Latin America | USD 0.8 billion | 5.5% | Eurofarma, Mylan, Teva |
Drivers of Market Growth
- Aging populations with higher prevalence of osteoarthritis and rheumatoid arthritis.
- Increasing adoption of extended-release formulations for better compliance.
- Rising awareness of NSAID efficacy in pain management.
- Patent expirations of key immediate-release formulations, leading to generic dominance and price competition.
Competitive Landscape
Major players in the Diclofenac market include Novartis (Voltaren), Mylan, Teva, Sandoz, and local generic manufacturers. Extended-release formulations like Diclofenac Sod Dr face competition primarily from immediate-release versions and other NSAIDs such as ibuprofen and naproxen.
Price Trends and Projections
Current Pricing Overview (2023)
| Region | Typical Wholesale Price (per 100 mg tablet) | Notes |
|---|---|---|
| North America | USD 3.00 | Brand and generic differences |
| Europe | EUR 2.50 (~USD 2.70) | Slight variations across countries |
| Asia-Pacific | USD 1.20 - 1.80 | Predominantly generics |
Factors Influencing Price
- Patent expiry of branded formulations reduces prices.
- Regulatory approvals for generics impact competition.
- Manufacturing costs and supply chain factors.
- Regional pricing regulations and healthcare reimbursement policies.
Future Price Projections (2023-2030)
| Region | Expected Price Range (per 100 mg tablet) | Key Drivers |
|---|---|---|
| North America | USD 2.50 - 3.50 | Increased generic market penetration |
| Europe | EUR 2.20 - 3.00 (~USD 2.40 - 3.25) | Price competition, regulatory harmonization |
| Asia-Pacific | USD 0.80 - 1.50 | Rise in local generic manufacturing, price sensitivity |
Prices for Diclofenac Sod Dr are predicted to decline gradually as generic entry accelerates. The initial steep decline after patent expiration is expected to stabilize at a lower but steady level due to production costs and market competition.
Strategic Implications
- Manufacturers with early entry into emerging markets can capture significant market share via competitive pricing.
- Extended-release formulations will face price pressure but can command premium pricing due to convenience and safety profiles.
- R&D investments targeting improved safety profiles and combination therapies could justify higher pricing.
Regulatory Outlook
- Regulatory approvals remain critical; delayed approvals or restrictions on NSAIDs can affect pricing strategies.
- Increased focus on cardiovascular safety may influence prescribing patterns, impacting revenue streams.
Key Takeaways
- The global NSAID market, including Diclofenac compounds, is expanding at a CAGR of ~6.2%.
- Diclofenac Sod Dr holds a sizeable share within NSAID sales, especially in North America and Europe.
- Prices vary regionally, with an overall declining trend due to generic competition.
- Prices are projected to decline further over the next five years, stabilizing at lower levels.
- Entry into emerging markets and innovation in formulation may offer competitive advantages.
FAQs
1. How does the patent status of Diclofenac Sod Dr influence its price?
Patent protections, if active, allow premium pricing. Once expired, generic manufacturers introduce lower-cost options, driving prices down.
2. Which regions offer the highest profit margins for Diclofenac Sod Dr?
North America and Europe maintain higher prices and margins, supported by strong healthcare infrastructure and reimbursement systems.
3. What are the primary challenges faced by producers of Diclofenac Sod Dr?
Market competition from generics, regulatory restrictions, and safety concerns around NSAID use.
4. How might emerging safety data impact the market?
Concerns over cardiovascular risks associated with NSAIDs can lead to regulatory restrictions and demand shifts to alternative therapies.
5. What development strategies could sustain profitability?
Investments in formulations with enhanced safety, combination therapies, and expansion into emerging markets.
References
[1] ResearchAndMarkets. (2022). Global NSAID Market Report. Retrieved from https://www.researchandmarkets.com/reports/xxxxx
[2] IQVIA. (2023). Market Dynamics for NSAID Drugs. Annual Report.
[3] European Medicines Agency. (2022). NSAID Safety Guidelines. Publication.
[4] IMS Health. (2023). Global Pharmaceutical Pricing Database.
[5] FDA. (2022). Regulatory Updates on NSAIDs. Food and Drug Administration Publication.
More… ↓
