Drug Price Trends for DEXTROAMP-AMPHETAM
✉ Email this page to a colleague
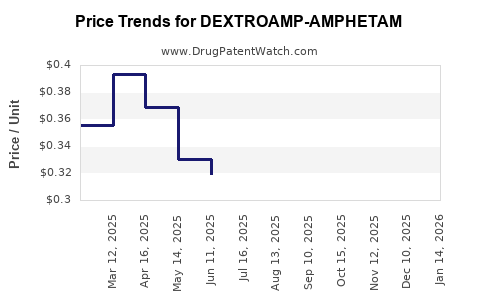
Average Pharmacy Cost for DEXTROAMP-AMPHETAM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 42858-0724-01 | 0.28617 | EACH | 2024-11-20 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 11534-0193-01 | 0.28617 | EACH | 2024-11-20 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 31722-0158-01 | 0.28617 | EACH | 2024-11-20 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 00527-0763-37 | 0.28617 | EACH | 2024-11-20 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 16714-0805-01 | 0.28617 | EACH | 2024-11-20 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 00527-1503-37 | 0.28617 | EACH | 2024-11-20 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 00555-0776-02 | 0.28617 | EACH | 2024-11-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |


