Share This Page
Drug Price Trends for DARUNAVIR
✉ Email this page to a colleague
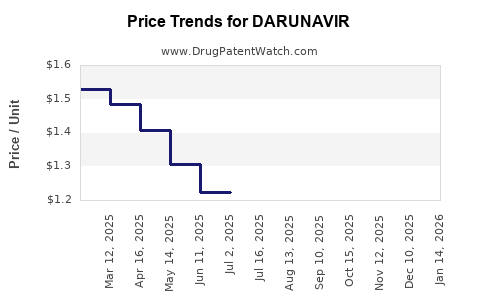
Average Pharmacy Cost for DARUNAVIR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DARUNAVIR 600 MG TABLET | 43598-0704-60 | 1.14915 | EACH | 2026-05-20 |
| DARUNAVIR 600 MG TABLET | 60687-0808-21 | 1.14915 | EACH | 2026-05-20 |
| DARUNAVIR 600 MG TABLET | 60687-0808-11 | 1.14915 | EACH | 2026-05-20 |
| DARUNAVIR 600 MG TABLET | 59651-0085-60 | 1.14915 | EACH | 2026-05-20 |
| DARUNAVIR 800 MG TABLET | 76282-0738-30 | 2.14344 | EACH | 2026-05-20 |
| DARUNAVIR 600 MG TABLET | 31722-0568-60 | 1.14915 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Darunavir Market Analysis and Price Projections
Darunavir, a protease inhibitor used in the treatment of HIV-1 infection, exhibits a projected market expansion driven by its efficacy, a growing patient population, and evolving treatment guidelines. Current patent exclusivity is expiring, necessitating an analysis of generic competition and its impact on market dynamics and pricing.
What is Darunavir's Current Market Position?
Darunavir, marketed by Janssen Pharmaceuticals (a Johnson & Johnson subsidiary) primarily as Prezista, is a second-generation HIV-1 protease inhibitor. Its mechanism of action involves blocking the HIV protease enzyme, preventing the maturation of new virions. It is typically used in combination therapy with other antiretroviral agents.
As of the latest available data, darunavir remains a cornerstone of many HIV treatment regimens due to its high genetic barrier to resistance and favorable safety profile compared to earlier protease inhibitors. The global market for HIV therapeutics is substantial, with protease inhibitors occupying a significant segment. While newer drug classes have emerged, darunavir's established efficacy and its availability in fixed-dose combinations contribute to its sustained market presence.
Key Market Indicators:
- Global HIV Population: The Joint United Nations Programme on HIV/AIDS (UNAIDS) estimated 38.4 million people were living with HIV at the end of 2021. [1]
- Treatment Coverage: Approximately 28.2 million people were accessing antiretroviral therapy (ART) at the end of 2021, representing 73% of all people living with HIV. [1]
- Darunavir's Role: Darunavir is a recommended option in leading treatment guidelines, including those from the U.S. Department of Health and Human Services (DHHS) and the European AIDS Clinical Society (EACS), particularly for treatment-experienced patients and in first-line settings where resistance is a concern. [2, 3]
The market for darunavir is intrinsically linked to the overall demand for HIV treatment and the competitive landscape within the antiretroviral therapy (ART) market.
What are the Key Patent Expirations and Generic Entry Timelines?
The primary patents protecting darunavir and its formulations are nearing expiration, a critical factor influencing future market dynamics and pricing. The core composition of matter patent for darunavir has expired in major markets. However, secondary patents related to specific formulations, methods of use, or manufacturing processes may extend exclusivity periods.
Key Patent Expirations:
- United States: Key composition of matter patents for darunavir have expired. Generic manufacturers have initiated Abbreviated New Drug Application (ANDA) processes. First generic entries were observed in late 2023/early 2024.
- Europe: European patents have also largely expired, paving the way for generic competition. Generic darunavir products have begun to enter the European market.
- Other Major Markets: Similar patent expiration timelines are observed in Canada, Australia, and Japan.
The entry of generic darunavir is expected to significantly alter the market by introducing lower-cost alternatives. This generic erosion typically leads to substantial price reductions for the branded product and the establishment of competitive pricing for generics.
How Will Generic Competition Impact Darunavir Pricing?
The introduction of generic darunavir will exert downward pressure on pricing across the entire market segment. Branded darunavir (Prezista) prices will likely decline as manufacturers compete with lower-cost generic equivalents. Simultaneously, the price of generic darunavir itself will be subject to competitive forces among multiple generic manufacturers.
Projected Pricing Trends:
- Immediate Post-Generic Entry: Expect a rapid price decrease of the branded product, potentially by 30-50% or more, within the first 12-18 months of generic availability.
- Generic Price Erosion: Generic darunavir prices will likely fall by 70-90% from the original branded price within 2-3 years of market entry, driven by competition among multiple generic manufacturers.
- Long-Term Pricing: Prices will stabilize at levels determined by manufacturing costs, market demand, and the pricing strategies of major generic players.
Comparison with Other ART Generics:
Historical data from other ART classes shows that generic entry can lead to dramatic price reductions. For example, upon the genericization of tenofovir disoproxil fumarate (TDF)-based regimens, prices dropped by over 90%. Similar, though potentially less severe due to darunavir's complexity, price decreases are anticipated for darunavir.
What are the Projected Market Size and Growth Rates?
The market size for darunavir is influenced by patient numbers, treatment duration, and pricing. With generic entry, the volume of prescriptions is expected to increase due to lower costs, potentially offsetting some of the revenue loss from price reductions.
Market Size Projections:
- Current Market Value: While specific market figures for darunavir as a single agent are proprietary, the global market for HIV therapeutics is estimated to be in the tens of billions of dollars annually. Darunavir and its combinations contribute a significant portion.
- Pre-Generic Market Growth: Prior to widespread genericization, the market for branded darunavir was experiencing moderate growth, driven by its inclusion in treatment guidelines and expanding access to ART in low- and middle-income countries.
- Post-Generic Market Dynamics: The introduction of generics will likely lead to a temporary decline in the absolute dollar value of the branded darunavir market. However, the overall darunavir market (including generics) may see increased volume and a sustained or slightly declining dollar value in the short to medium term, followed by more stable growth as generic uptake stabilizes.
Factors Influencing Future Growth:
- HIV Prevalence and Incidence: Continued global efforts to combat HIV and manage the infected population directly impact demand.
- Treatment Guidelines: Inclusion in preferred treatment regimens by major health organizations is crucial.
- Emerging Markets: Expansion of access to ART in underserved regions will drive volume.
- Competition from Other Drug Classes: The development of novel HIV therapeutics with superior efficacy or convenience could impact darunavir's market share.
- Availability of Generic Formulations: The price and accessibility of generic darunavir will be a primary determinant of overall market volume.
What are the Key Drivers and Restraints for Darunavir's Market Future?
Several factors will shape the future trajectory of the darunavir market. Understanding these drivers and restraints is essential for strategic planning.
Key Drivers:
- Established Efficacy and Safety Profile: Darunavir's proven track record in managing HIV, particularly in treatment-experienced patients, ensures continued clinical relevance. [4]
- High Genetic Barrier to Resistance: This attribute is critical for long-term treatment success and reduces the need for regimen switches due to resistance.
- Inclusion in Standard of Care Guidelines: DHHS and EACS guidelines consistently recommend darunavir, reinforcing its clinical importance. [2, 3]
- Cost-Effectiveness of Generic Options: Lower-priced generic darunavir will expand access to effective ART, particularly in resource-limited settings.
- Fixed-Dose Combination Products: While some combinations may face patent challenges, continued use of darunavir in convenient single-pill regimens supports patient adherence.
Key Restraints:
- Emergence of Novel HIV Therapeutics: New drug classes and next-generation integrase strand transfer inhibitors (INSTIs) offer alternative treatment options with potentially improved convenience or different resistance profiles.
- Patent Expirations and Generic Competition: As detailed, this will lead to significant price erosion and revenue reduction for the branded product.
- Adherence Challenges: While darunavir has a good safety profile, maintaining adherence to complex ART regimens remains a challenge for some patients.
- Drug-Drug Interactions: Protease inhibitors, including darunavir, can have significant drug-drug interaction profiles, requiring careful management.
- Global Health Funding Fluctuations: Dependence on global health initiatives and government funding for ART procurement can introduce variability in market demand.
What are the Regulatory and Policy Implications?
Regulatory approvals and healthcare policies play a pivotal role in market access and pricing for pharmaceuticals. For darunavir, the genericization process is a key policy-driven event.
Regulatory Landscape:
- ANDA Approvals: Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are responsible for approving generic versions of darunavir. The speed and number of these approvals will dictate the intensity of generic competition.
- Bioequivalence Standards: Generic drugs must demonstrate bioequivalence to the reference listed drug (branded darunavir), ensuring comparable safety and efficacy.
- Manufacturing Standards: All darunavir products, branded and generic, must adhere to stringent Current Good Manufacturing Practices (cGMP).
Policy Implications:
- Formulary Placement: Payer policies and formulary decisions will significantly influence which darunavir products (branded vs. generic) are preferred and reimbursed.
- Public Health Procurement: Decisions by organizations like the Global Fund to Fight AIDS, Tuberculosis and Malaria, and PEPFAR (U.S. President's Emergency Plan for AIDS Relief) will impact the volume of generic darunavir purchased in developing countries.
- Antitrust and Competition Laws: Regulatory bodies monitor the market to prevent anti-competitive practices that could hinder generic entry or inflate prices.
The shift to generic darunavir necessitates a re-evaluation of procurement strategies by healthcare systems and a focus on ensuring equitable access to affordable treatment.
Key Takeaways
- Darunavir is a clinically significant antiretroviral agent with a substantial global patient base.
- Key patents for darunavir have expired, leading to the imminent and ongoing entry of generic competitors across major markets.
- Generic competition is projected to cause a substantial decline in darunavir pricing, with branded product prices falling significantly and generic prices stabilizing at much lower levels.
- The overall darunavir market volume is expected to increase due to generic affordability, though the dollar value may see short-term fluctuations before stabilizing.
- Continued clinical utility, inclusion in treatment guidelines, and the cost-effectiveness of generics are key drivers, while novel therapeutics and price erosion are major restraints.
- Regulatory approvals for generics and healthcare policy decisions regarding formulary placement and public health procurement are critical determinants of market access and future demand.
Frequently Asked Questions
-
When did the first generic versions of darunavir become available in the U.S. market? First generic entries for darunavir in the U.S. market commenced in late 2023 and early 2024.
-
What is the anticipated price reduction for branded darunavir (Prezista) following generic entry? Branded darunavir prices are expected to decrease by 30-50% or more within the first 12-18 months of generic availability.
-
Will darunavir continue to be recommended in HIV treatment guidelines after genericization? Yes, darunavir's established efficacy and safety profile suggest it will remain a recommended option in HIV treatment guidelines, particularly for specific patient populations.
-
What impact will generic darunavir have on treatment access in low- and middle-income countries? Lower-cost generic darunavir is expected to significantly improve access to effective HIV treatment in resource-limited settings by making ART more affordable.
-
Are there any remaining patents that could delay or restrict generic darunavir entry? While core composition of matter patents have expired, secondary patents related to specific manufacturing processes or formulations could exist. However, these are generally unlikely to prevent the broad market entry of generic versions.
Citations
[1] Joint United Nations Programme on HIV/AIDS (UNAIDS). (2022). Global HIV & AIDS statistics — 2022 fact sheet. UNAIDS.
[2] U.S. Department of Health and Human Services Panel on Antiretroviral Guidelines for Adults and Adolescents with HIV. (2023). Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents with HIV. National Institutes of Health.
[3] European AIDS Clinical Society (EACS). (2023). EACS Guidelines for the Clinical Management of HIV Infection. European AIDS Clinical Society.
[4] Gallant, J. E., DeJesus, E., Arribas, J. R., Pozniak, A. L., Gazzard, B. G., Faetini, V., ... & Kearney, B. P. (2006). H2T0013: Early virologic response and tolerability of darunavir/ritonavir in treatment-experienced HIV-1 infected patients. Journal of Infectious Diseases, 194(10), 1352-1361.
More… ↓
