Share This Page
Drug Price Trends for CROMOLYN SODIUM NASAL SPRAY
✉ Email this page to a colleague
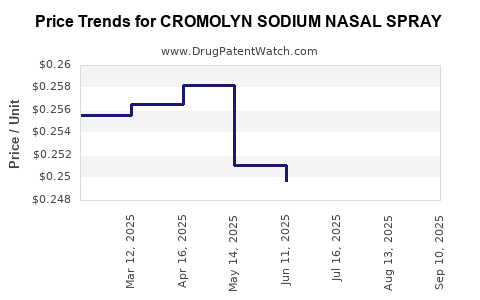
Average Pharmacy Cost for CROMOLYN SODIUM NASAL SPRAY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CROMOLYN SODIUM NASAL SPRAY | 57782-0397-26 | 0.24771 | ML | 2025-09-17 |
| CROMOLYN SODIUM NASAL SPRAY | 57782-0397-26 | 0.24692 | ML | 2025-08-20 |
| CROMOLYN SODIUM NASAL SPRAY | 57782-0397-26 | 0.24493 | ML | 2025-07-23 |
| CROMOLYN SODIUM NASAL SPRAY | 57782-0397-26 | 0.24973 | ML | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CROMOLYN SODIUM NASAL SPRAY MARKET ANALYSIS AND PRICE PROJECTIONS
SUMMARY: The market for cromolyn sodium nasal spray is characterized by its established role in allergic rhinitis treatment and its position as an over-the-counter (OTC) option. While its efficacy is recognized, competition from newer, more potent prescription antihistamines and intranasal corticosteroids has constrained significant market expansion. Current price points are stable, with projections indicating a consistent, albeit modest, pricing structure due to its generic status and mature market lifecycle.
WHAT IS THE CURRENT MARKET SIZE AND GROWTH TRAJECTORY FOR CROMOLYN SODIUM NASAL SPRAY?
The market size for cromolyn sodium nasal spray is not separately tracked in major pharmaceutical market reports, which typically segment by therapeutic class or drug mechanism. However, its primary indication is allergic rhinitis, a condition with a substantial global patient population. The World Allergy Organization estimates that 30-40% of the global population suffers from allergic rhinitis, with prevalence rates varying by region [1].
Cromolyn sodium nasal spray is generally categorized within the broader allergy relief segment, which is valued in the tens of billions of dollars globally. Within this, OTC nasal sprays represent a significant sub-segment. Growth in the overall allergy market is driven by increasing allergen exposure, urbanization, and rising disposable incomes.
However, the growth trajectory for cromolyn sodium nasal spray specifically is considered flat to modest. This is attributed to several factors:
- Maturity: The drug has been available for decades, with its patent protection long expired. This means it is a well-established generic product.
- Competition: It faces significant competition from prescription and some OTC products that offer faster symptom relief or a broader range of effects. These include:
- Intranasal corticosteroids: Fluticasone, budesonide, mometasone. These are considered first-line therapy for moderate to severe allergic rhinitis.
- Antihistamines: Oral and some nasal spray formulations (e.g., azelastine).
- Mechanism of Action: Cromolyn sodium is a mast cell stabilizer, requiring prophylactic use (daily administration for weeks) for optimal efficacy. This contrasts with the rapid symptomatic relief offered by antihistamines and corticosteroids, which are often preferred for acute allergic episodes.
Industry analysis suggests that while cromolyn sodium nasal spray maintains a dedicated user base, particularly for individuals who tolerate it well and prefer a prophylactic approach without systemic side effects, it is unlikely to capture substantial new market share or experience significant growth in the current competitive landscape. The market is more likely to see stable demand from its existing user base rather than rapid expansion.
WHO ARE THE KEY MANUFACTURERS AND DISTRIBUTORS?
Due to its generic status, the manufacturing and distribution of cromolyn sodium nasal spray involve multiple pharmaceutical companies. Production often occurs in multi-product facilities. Key players in the OTC allergy medication space, which may include cromolyn sodium nasal spray in their portfolios, are:
- Perrigo Company plc: A major supplier of OTC pharmaceutical products, often manufacturing for private labels.
- Bausch Health Companies Inc.: Has a broad range of consumer health products, including allergy relief.
- Major generic drug manufacturers: Companies like Teva Pharmaceutical Industries Ltd., Mylan (now Viatris), and Sandoz (a Novartis division) are involved in the production of active pharmaceutical ingredients (APIs) and finished dosage forms for generic drugs, including cromolyn sodium.
- Private label manufacturers: Many retailers, such as CVS Pharmacy, Walgreens, and Walmart, offer their own store-brand versions of cromolyn sodium nasal spray, which are manufactured by contract or generic drug companies.
Distribution channels are primarily:
- Retail Pharmacies: Both chain and independent pharmacies.
- Mass Retailers: Supermarkets and general merchandise stores with pharmacy sections.
- Online Pharmacies and E-commerce Platforms: Including direct-to-consumer services and major online retailers.
The market is characterized by a diffuse supply chain rather than a few dominant manufacturers controlling exclusive rights.
WHAT ARE THE CURRENT PRICING STRUCTURES AND KEY PRICE DRIVERS?
Cromolyn sodium nasal spray is predominantly sold as an over-the-counter (OTC) product in the United States, typically in 13 ml or 15 ml spray bottles. Its pricing is influenced by several factors:
- Generic Status: As a long-established generic drug, competition among manufacturers keeps prices relatively low compared to branded pharmaceuticals.
- Manufacturing Costs: The cost of API synthesis, formulation, packaging, and regulatory compliance are primary drivers.
- Wholesale Acquisition Cost (WAC): This is the list price of a drug. However, actual transaction prices are lower due to significant rebates and discounts negotiated by pharmacy benefit managers (PBMs) and large purchasers.
- Retail Markups: Pharmacies and retailers add their own markups to the wholesale price.
- Competitive Landscape: The pricing of competing OTC allergy products, including saline sprays, oral antihistamines, and other nasal sprays, influences price sensitivity.
- Distribution Channels: Prices can vary between brick-and-mortar pharmacies, online retailers, and warehouse clubs.
Current Price Range (USD):
- Retail Price (13 ml or 15 ml bottle): Typically ranges from $12 to $25. Prices can be lower with store brands or during promotional periods.
- Online Retailers (e.g., Amazon, Walmart.com): Often show prices in the $10 to $20 range for single units, with potential discounts for multi-packs or subscriptions.
- Wholesale/Institutional Pricing: Not publicly available but would be significantly lower, reflecting bulk purchasing.
Key Price Drivers:
- API Cost: Fluctuations in the cost of raw materials and the production of cromolyn sodium API.
- Labor and Energy Costs: Affecting manufacturing and logistics.
- Regulatory Compliance: Costs associated with maintaining FDA compliance.
- Marketing and Distribution Expenses: Though less significant for mature generics compared to new product launches.
The price is generally considered stable, with no significant upward or downward pressures beyond typical inflation or minor supply chain shifts. The lack of significant innovation or patent exclusivity prevents substantial price increases.
WHAT ARE THE PRICE PROJECTIONS FOR THE NEXT 3-5 YEARS?
The price projections for cromolyn sodium nasal spray over the next 3-5 years are expected to remain stable with a low single-digit inflation rate. Several factors support this projection:
- No Impending Patent Expirations or New Entrants: The drug is off-patent, and there are no new patent expiries to consider. Furthermore, the market is mature, and the development of new, significantly superior cromolyn sodium formulations is unlikely.
- Competition as a Price Anchor: The continued presence of numerous generic manufacturers and competing OTC allergy products will prevent significant price hikes. Any attempt to drastically increase prices would likely lead consumers to switch to more cost-effective alternatives.
- Inflationary Pressures: The primary driver for any price increase will be general inflation impacting manufacturing, labor, and distribution costs. This is typically projected in the 1-3% range annually for consumer goods.
- Stable Demand: While not growing rapidly, demand is expected to remain consistent from its established user base. This stable demand prevents price volatility.
- OTC Market Dynamics: The OTC market is generally more price-sensitive than prescription markets. Manufacturers of generic OTC drugs focus on volume and cost efficiency rather than premium pricing.
Projected Price Range (USD) for a 13 ml or 15 ml bottle in 3-5 years:
- Retail Price: Expected to be in the range of $13 to $28.
It is important to note that the projections assume no major unforeseen events, such as a significant supply chain disruption affecting global API production or a sudden, widespread regulatory change impacting manufacturing standards for this specific drug. However, based on current market dynamics and the drug's lifecycle, incremental price increases aligned with inflation are the most probable scenario.
WHAT IS THE COMPETITIVE LANDSCAPE AND POTENTIAL FOR DISRUPTION?
The competitive landscape for cromolyn sodium nasal spray is highly saturated and mature. Its primary competition stems from products offering different mechanisms of action and more rapid or comprehensive symptom relief.
Primary Competitive Categories:
-
Intranasal Corticosteroids (Nasal Steroids):
- Examples: Fluticasone propionate (e.g., Flonase), budesonide (e.g., Rhinocort), mometasone furoate (e.g., Nasonex - now OTC).
- Mechanism: Reduce inflammation by suppressing the immune response in the nasal passages.
- Advantages: Highly effective for a wide range of allergic rhinitis symptoms, including congestion, sneezing, itching, and runny nose. Generally considered first-line therapy for moderate to severe symptoms.
- Disadvantages: Can take several days to reach full effect, though newer formulations are faster. Potential for local side effects like nasal irritation or dryness. Prescription-only status historically, though many are now OTC.
- Impact on Cromolyn: Strongest competition. Their broad efficacy and increasing OTC availability have made them the preferred choice for many consumers and physicians.
-
Antihistamines (Oral and Nasal):
- Oral Examples: Cetirizine (Zyrtec), loratadine (Claritin), fexofenadine (Allegra).
- Oral Mechanism: Block the action of histamine, a key mediator of allergic reactions.
- Oral Advantages: Rapid relief of itching, sneezing, and runny nose. Widely available OTC.
- Oral Disadvantages: Less effective for nasal congestion. Can cause drowsiness (first-generation) or have other side effects.
- Nasal Examples: Azelastine (Astelin - prescription, now some combination OTC products).
- Nasal Mechanism: Direct antihistamine action in the nasal mucosa.
- Nasal Advantages: Rapid onset of action for nasal symptoms.
- Nasal Disadvantages: May have a bitter taste.
- Impact on Cromolyn: Significant competition, especially for consumers seeking quick relief from sneezing and itching.
-
Decongestants (Oral and Nasal Sprays):
- Examples: Pseudoephedrine (Sudafed - oral), phenylephrine (Sudafed PE - oral), oxymetazoline (Afrin - nasal spray).
- Mechanism: Vasoconstriction to reduce nasal swelling and congestion.
- Advantages: Effective for rapid relief of nasal congestion.
- Disadvantages: Oral decongestants have systemic side effects and are behind-the-counter (due to pseudoephedrine). Nasal decongestants risk rebound congestion (rhinitis medicamentosa) with prolonged use (over 3 days).
- Impact on Cromolyn: Less direct competition as they primarily target congestion, whereas cromolyn is a prophylactic stabilizer. However, consumers may use them for symptomatic relief.
-
Saline Nasal Sprays:
- Mechanism: Moisturize nasal passages, thin mucus, and help clear irritants.
- Advantages: Safe, non-medicated, and can be used frequently.
- Disadvantages: Do not treat the underlying allergic mechanism.
- Impact on Cromolyn: Complementary rather than directly competitive. Some users may find saline sufficient, while others need active medication.
Potential for Disruption:
- New Formulations of Existing Drugs: Development of nasal steroid or antihistamine nasal sprays with even faster onset, better tolerability, or extended-release properties could further marginalize cromolyn sodium.
- Combination Products: The emergence of OTC combination products (e.g., antihistamine + steroid) that offer broader coverage could reduce the need for single-mechanism therapies.
- Biologics: For severe allergic rhinitis or asthma-related allergies, biologic therapies (e.g., dupilumab) are increasingly used. While not direct competitors to OTC nasal sprays, they represent a shift in treatment paradigms for more severe cases, potentially reducing the overall market for older, less potent medications.
- Emerging Therapies: Research into novel anti-inflammatory pathways or immunotherapy approaches could eventually offer disruptive alternatives, though these are typically long-term prospects and would likely enter as prescription therapies initially.
Cromolyn sodium's niche lies in its safety profile (minimal systemic side effects) and its prophylactic mechanism for those who find it effective when used consistently. However, the convenience and broad efficacy of newer treatments mean disruption is more likely to come from incremental improvements in competing drug classes rather than a direct attack on cromolyn sodium itself. The drug's market share is more at risk from being out-competed by superior alternatives than from a new entrant directly targeting its specific segment with a similar mechanism.
WHAT ARE THE REGULATORY CONSIDERATIONS AND THEIR IMPACT ON PRICING?
The regulatory landscape for cromolyn sodium nasal spray is primarily governed by the U.S. Food and Drug Administration (FDA) for its OTC status. Key considerations include:
- Monograph System: Cromolyn sodium nasal spray falls under the FDA's OTC drug monograph system for nasal antiallergic drug products. This system allows for drugs to be marketed OTC without requiring individual pre-market approval, provided they conform to established conditions (monographs) regarding active ingredients, dosages, labeling, and manufacturing practices.
- Impact on Pricing: The monograph system facilitates market entry for generic manufacturers by reducing regulatory hurdles and development costs compared to prescription drugs requiring New Drug Applications (NDAs). This inherently fosters competition and drives prices down.
- Current Good Manufacturing Practices (cGMP): All manufacturers must adhere to cGMP regulations to ensure product quality, safety, and efficacy.
- Impact on Pricing: Maintaining cGMP compliance involves ongoing costs for quality control, facility upgrades, and personnel training. These costs are a fundamental component of the overall manufacturing cost and thus influence the final price. Any tightening of cGMP standards or increased inspection frequency could lead to higher compliance costs.
- Labeling Requirements: OTC drugs have specific labeling requirements for ingredients, indications, warnings, dosage instructions, and administration.
- Impact on Pricing: Labeling updates are generally low-cost events for established products unless mandated by new regulations. The clarity and comprehensiveness of labeling are crucial for consumer use and compliance.
- Post-Market Surveillance: The FDA monitors drug safety through adverse event reporting systems and may require post-market studies or label changes if new safety concerns arise.
- Impact on Pricing: While rare for established drugs like cromolyn sodium, a significant safety signal could necessitate product recalls, costly investigations, or withdrawal from the market, which would drastically impact availability and potentially pricing if the drug were reintroduced with new restrictions. However, cromolyn sodium has a well-established safety profile.
- Drug Master Files (DMFs): Manufacturers of the API must maintain DMFs with the FDA, which contain detailed information about the manufacturing process, facilities, and controls.
- Impact on Pricing: The cost associated with preparing and maintaining DMFs contributes to the API's price. Competition among API suppliers with compliant DMFs helps to moderate API costs.
Overall Impact on Pricing:
The regulatory framework for OTC drugs like cromolyn sodium nasal spray is designed to promote accessibility and affordability by fostering competition. The established monograph provides a clear pathway for generic market participation, preventing any single entity from exerting significant pricing power. Regulatory compliance adds a baseline cost to manufacturing, but this is a standard operational expense for all pharmaceutical products. Consequently, regulatory factors do not currently present an opportunity for significant price increases but rather act as a continuous force in maintaining a competitive and stable pricing environment.
KEY TAKEAWAYS
- The cromolyn sodium nasal spray market is stable and mature, driven by its established efficacy in allergic rhinitis and its OTC status.
- Significant market growth is constrained by strong competition from intranasal corticosteroids and antihistamines, which offer broader symptom relief or faster onset.
- Pricing is characterized by low-cost generic competition, with typical retail prices ranging from $12 to $25 per 13-15 ml bottle.
- Price projections indicate stability with low single-digit annual increases due to inflation, with no significant disruptive pricing events anticipated.
- Regulatory compliance and the OTC monograph system reinforce a competitive pricing environment, limiting upward price potential.
FAQS
-
Is cromolyn sodium nasal spray a prescription-only medication? Cromolyn sodium nasal spray is available as an over-the-counter (OTC) medication in the United States, allowing consumers to purchase it without a prescription.
-
How does cromolyn sodium nasal spray differ from corticosteroid nasal sprays in terms of efficacy? Cromolyn sodium acts as a mast cell stabilizer, requiring daily use for several weeks to achieve full prophylactic benefit. Corticosteroid nasal sprays reduce inflammation and generally provide faster symptom relief for a wider range of symptoms, including congestion, and are often considered more potent.
-
What is the typical duration of treatment recommended for cromolyn sodium nasal spray? For optimal effectiveness, cromolyn sodium nasal spray is recommended for regular daily use, typically for two to four weeks, before allergy season begins or when allergen exposure is expected. It is designed for prophylactic (preventive) use rather than acute symptom relief.
-
Are there any significant side effects associated with cromolyn sodium nasal spray? Cromolyn sodium nasal spray is generally well-tolerated. The most common side effects are mild and localized, such as nasal irritation, sneezing, or a temporary unpleasant taste or smell. Systemic side effects are rare due to its minimal absorption into the bloodstream.
-
Can the price of cromolyn sodium nasal spray increase significantly in the near future? Significant price increases for cromolyn sodium nasal spray are unlikely in the near future. Its status as a long-established generic drug with multiple manufacturers, coupled with intense competition from alternative OTC allergy medications, creates a stable and generally low-price market environment.
CITATIONS
[1] World Allergy Organization. (n.d.). Allergic Rhinitis. Retrieved from https://www.worldallergy.org/education-and-resources/allergic-rhinitis
More… ↓
